Introduction
Metabolism is the set of enzyme-catalyzed reactions that occur in the body to produce energy and building blocks for biosynthesis.
Because metabolic reactions occur inside cells, they are compartmentalized across cellular and subcellular membranes.
Understanding metabolism therefore requires knowledge of membrane structure, transport systems, and transmembrane electrical potentials.
Why this matters
Metabolic reactions are tightly regulated according to the needs of the organism. Regulation occurs at both the enzyme and gene level and depends heavily on cell signaling (for example hormones and neurotransmitters).
Most diseases involve altered metabolism and/or altered metabolic regulation.
This includes classic metabolic diseases (for example diabetes), but also cardiovascular disease, neurodegenerative disease, psychiatric disorders, and cancer with major metabolic components.
- Degenerative: Alzheimer’s disease is linked to impaired cerebral glucose metabolism
- Psychiatric: Bipolar disorder is linked to mitochondrial dysfunction
- Cancer: Colorectal cancer is strongly linked to hyperinsulinemia
Core Topics
- Metabolic pathways
- Catabolism and anabolism
- General bioenergetics
- Coupled reactions
- Metabolic regulation
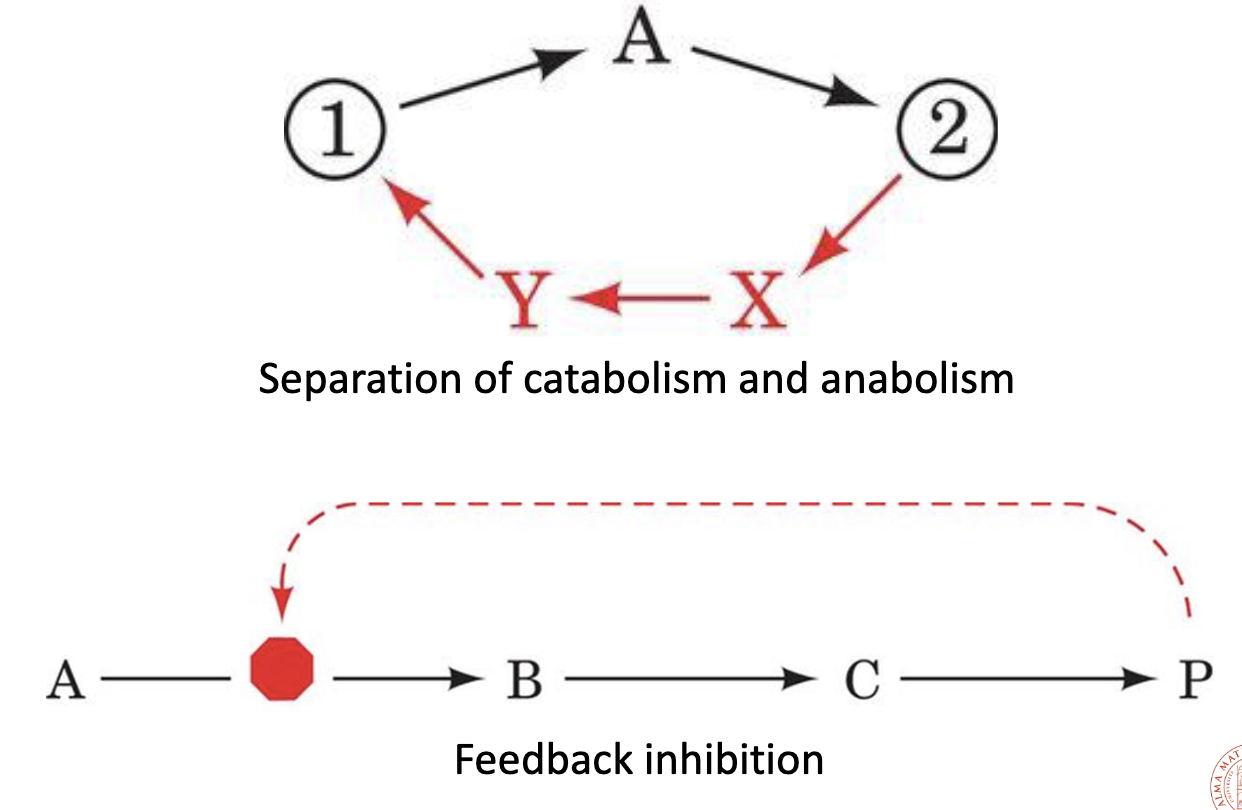
Catabolism and Anabolism
Important
Catabolism:
- is oxidative
- and releases energy.
- the number of C–O bonds increases.
Anabolism is:
- reductive
- and requires energy.
Palmitate oxidation ← Catabolism
Palmitate -> 8 acetate
It requires 14 oxidation steps and generates 28 ATP.
Palmitate synthesis ← Anabolism
8 acetate -> palmitate
It requires 14 reductions and 7 ATP.
Anki cloze
Catabolism is {1:oxidative} and {1:releases} energy.
Anabolism is {1:reductive} and {1:requires} energy.
Extra: Palmitate -> 8 acetate needs 14 oxidations and yields 28 ATP. Palmitate synthesis needs 14 reductions} and 7 ATP.
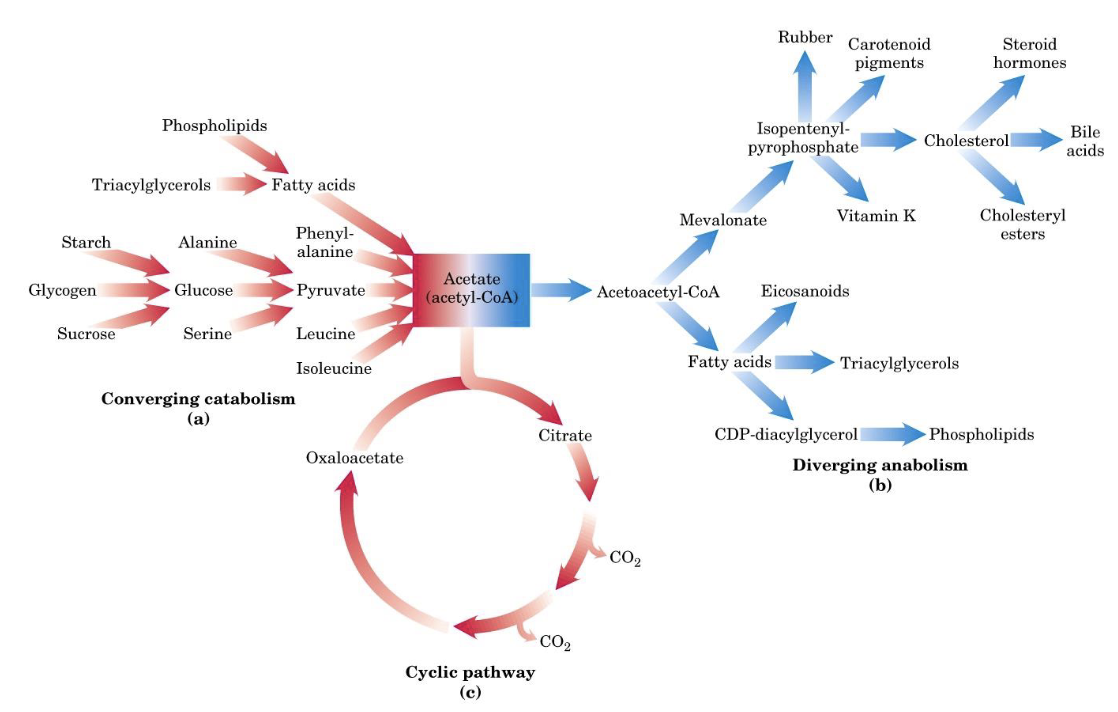
Metabolic Pathways
- Linear (for example glycolysis)
- Branched
- Converging (for example toward pyruvate)
- Diverging (for example from isopentenyl pyrophosphate)
- Cyclic (for example TCA cycle, ornithine cycle)
Anki cloze
Metabolic pathways can be {1:linear}, {1:branched} (either {2:converging} or {2:diverging}), or {1:cyclic}.
Examples: {2:glycolysis} is linear, pathways to {2:pyruvate} can be converging, and {2:TCA cycle} is cyclic.
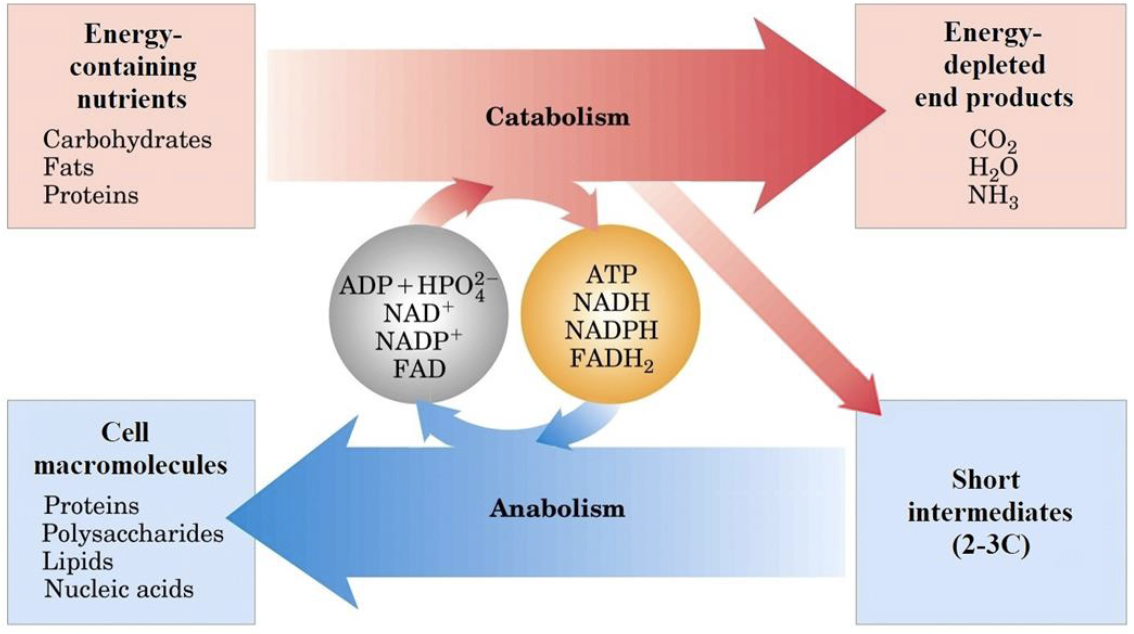
Overview of Catabolic Processes
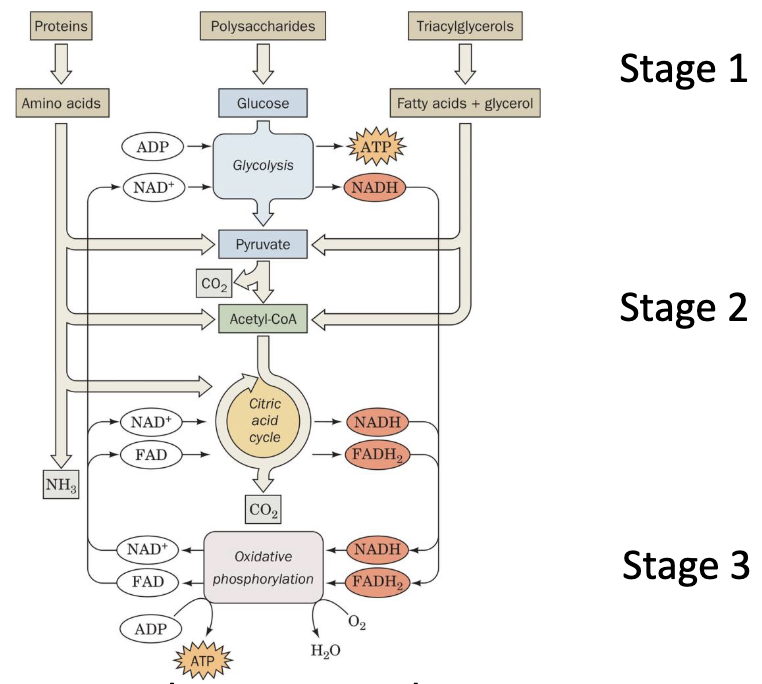
- Polymers (macromolecules) are broken down to monomers and small molecules.
- Redox coenzymes are generated from simple molecules.
- ATP is produced.
Anabolic pathways proceed in the opposite direction
Stage 2 requires ATP and reducing power. Stage 1 also requires ATP.
Major Concepts in Metabolism
- Metabolic flux
- Steady state
- Direction (thermodynamics)
- Rate (kinetics)
- Quantitative transformation
- Cell compartmentalization
- Metabolic control
Steady State
For a pathway:
At steady state, concentrations of intermediates remain constant because formation rate equals consumption rate.
For example, is constant when .
Thermodynamics and Kinetics
Thermodynamics determines reaction direction:
- Net flow proceeds toward equilibrium.
- In living systems, true equilibrium is usually not reached; pathways run at steady state.
- At equilibrium, — meaning no net energy is released. Equilibrium is therefore incompatible with sustained energy production.
Why steady state ≠ equilibrium
At steady state, the system is stable but not at equilibrium. Reactions continue at a constant rate. Equilibrium would mean no further net transformation — no energy output.
Kinetics determines reaction rate:
- Metabolic rate is the amount of matter transformed per unit time.
- Enzymes establish and regulate this rate.
- Only proteins have the versatility to fine-tune chemical reactions.
On standard conditions and
Standard values assume physiological pH (). Standard conditions are necessary for comparison between reactions, even though cells never operate under true standard conditions.
Why and are common end products
and are always present as end products because they contain the most stable bonds. More stable = less free energy available to release.
Energy Flow During Metabolism
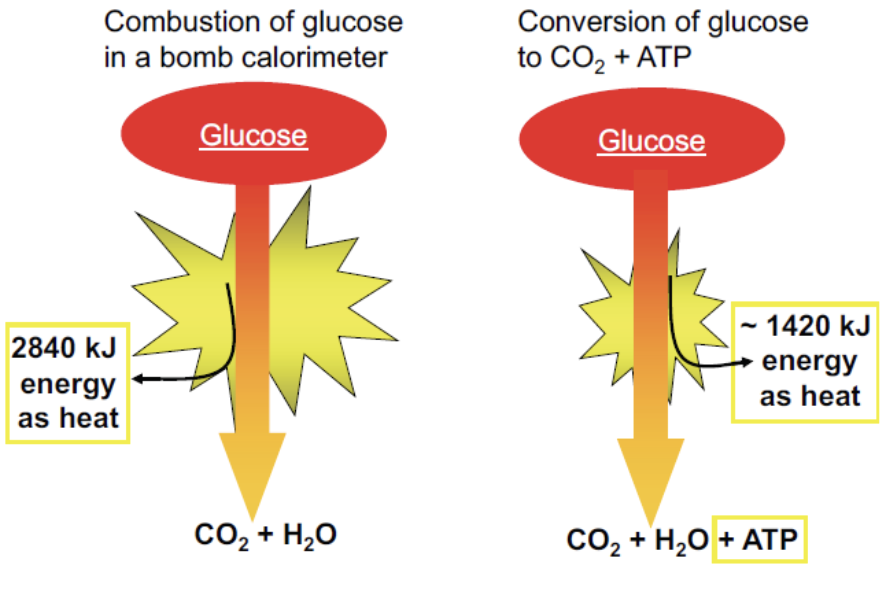
Cells obtain most usable energy through oxidation reactions, but not by direct reaction with oxygen in a single step.
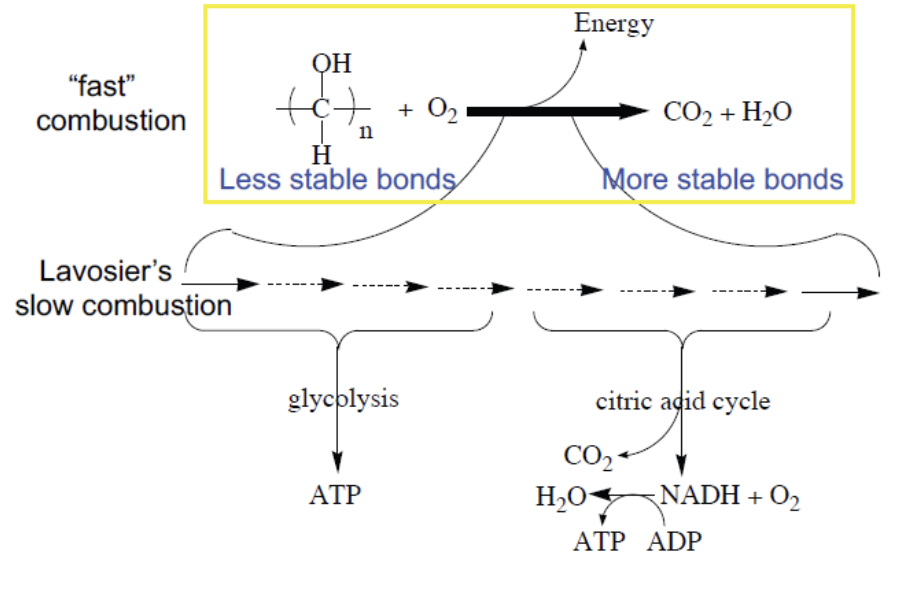
Part of oxidation energy is conserved as ATP.
Net direction and equilibrium
For , equilibrium position is given by:
“Reactant” and “product” labels are based on equation position, not on net direction under specific concentrations.
Gibbs Free Energy
For :
Using base-10 logs at physiological temperature (approximation):
At equilibrium:
Practice Problems (Solved)
1 → ATP hydrolysis under cellular concentrations
Given: , , ,
2 → FBP cleavage under cellular concentrations
Given: , , ,
Redox Reactions
Basic half-reaction:
Oxidations are often dehydrogenations:
Reduction partner:
Overall redox reaction:
Redox Potential and Nernst Equation
Approximation:
Relations to thermodynamics:
In non-standard conditions:
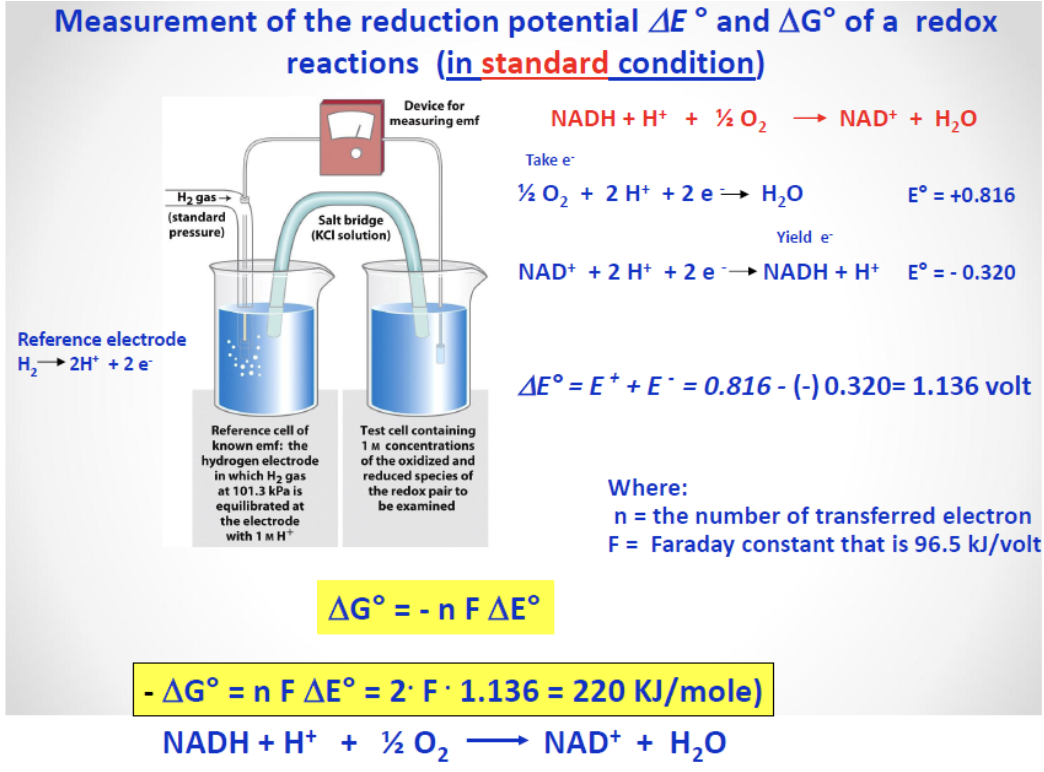
Coupled Reactions
An unfavorable reaction () can proceed if it is coupled to a favorable reaction (), as long as total free energy is negative:
Coupling often occurs through a common intermediate:
- ()
- ()
- Net: with
ATP coupling
Amino acid polymerization is endergonic.
ATP hydrolysis is exergonic.
Coupling the two makes overall protein synthesis thermodynamically favorable.
Glucose phosphorylation
has
has
Sum:
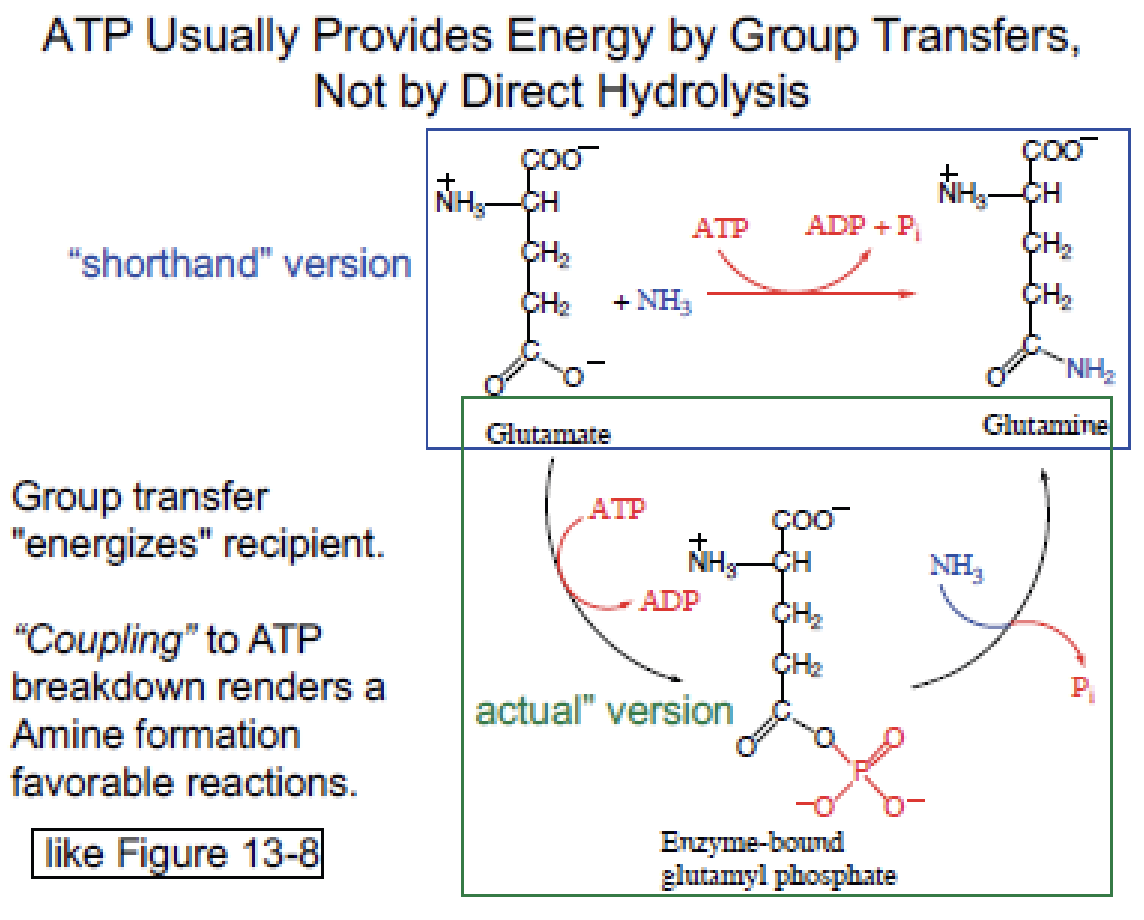
Glutamine synthesis
has
Coupled ATP hydrolysis gives:
Net
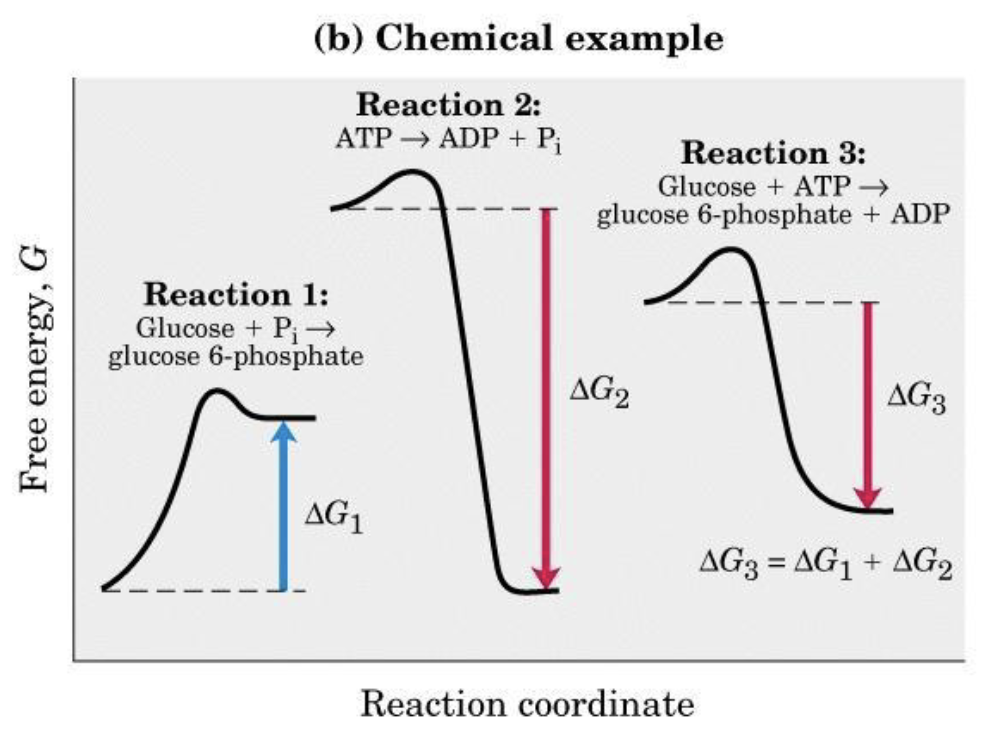
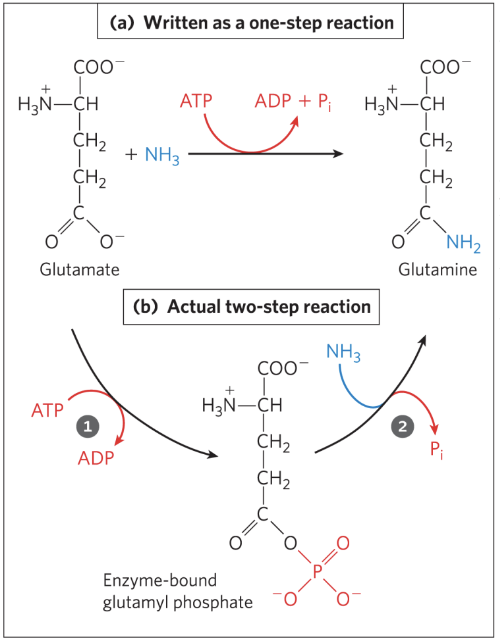
ATP Synthesis and ATP Utilization
ATP Synthesis (Phosphorylation of ADP)
Mechanisms:
- Substrate-level phosphorylation
- Oxidative phosphorylation
ATP Utilization
Important
ATP hydrolysis in metabolism is functionally a two-step process through enzyme-bound intermediates.
ATP typically provides energy by group transfer (transfer of a phosphoryl group to a substrate), not by direct hydrolysis. “Hydrolysis” is a simplification — the phosphoryl group is transferred either to a substrate or to water.
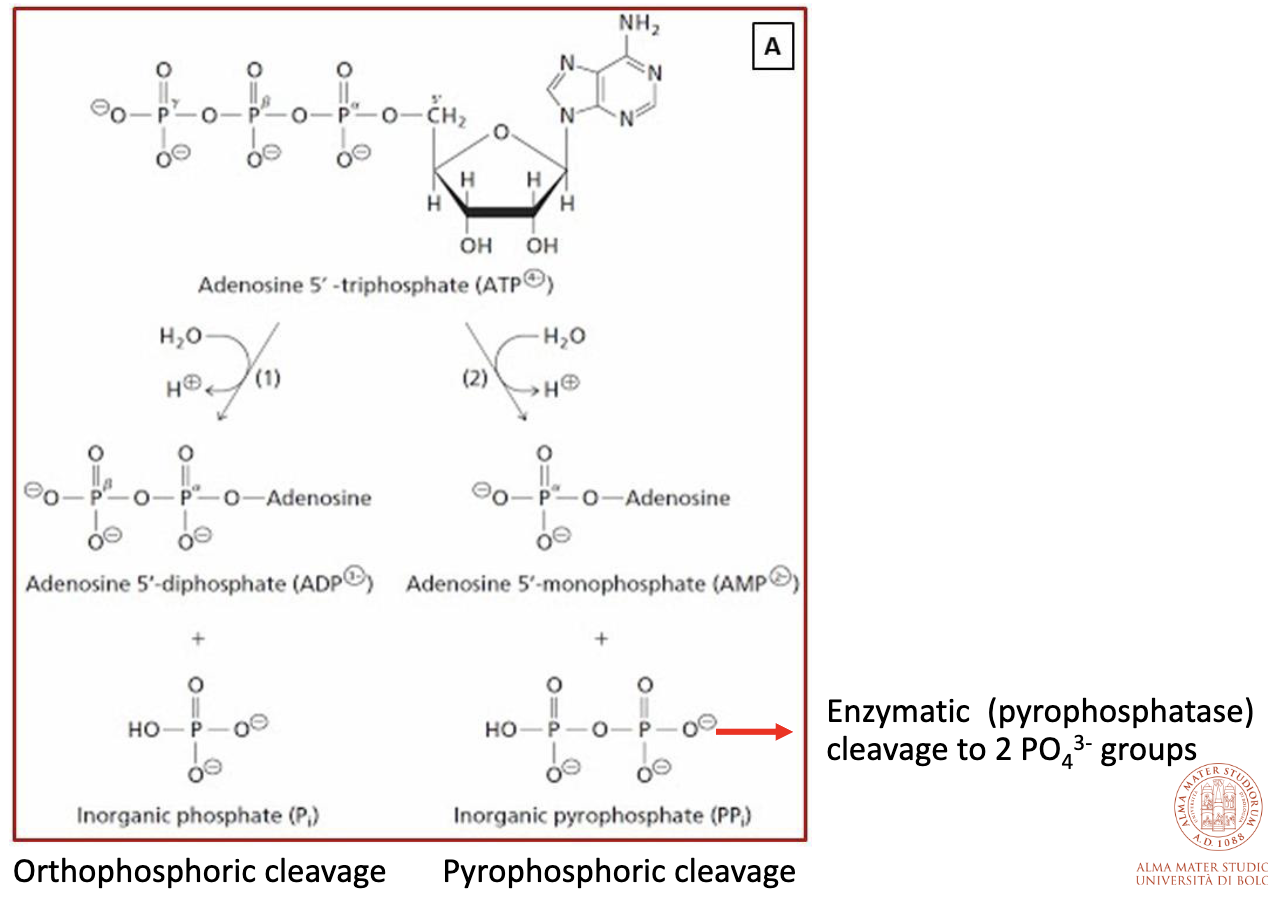
Orthophosphoric cleavage:
Example: glucose + ATP -> glucose-6-phosphate + ADP
Pyrophosphoric cleavage:
Example: amino acid activation in protein synthesis
Reaction is strongly driven forward by:
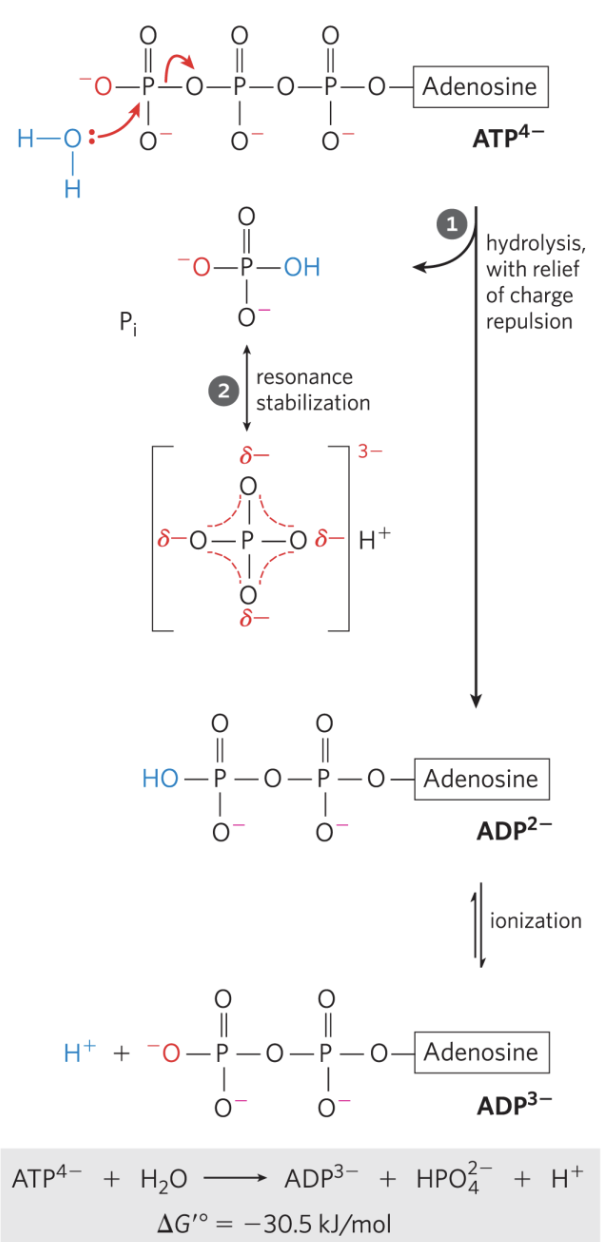
Chemical basis for ATP's high-energy release
The large of ATP hydrolysis arises from:
- Charge separation: the products and ADP carry separated negative charges
- Resonance stabilization of : inorganic phosphate forms a resonance hybrid, stabilizing the product
- Greater solvation of products ( and ADP) relative to ATP
A compound is considered high-energy if of hydrolysis .
Energy Charge
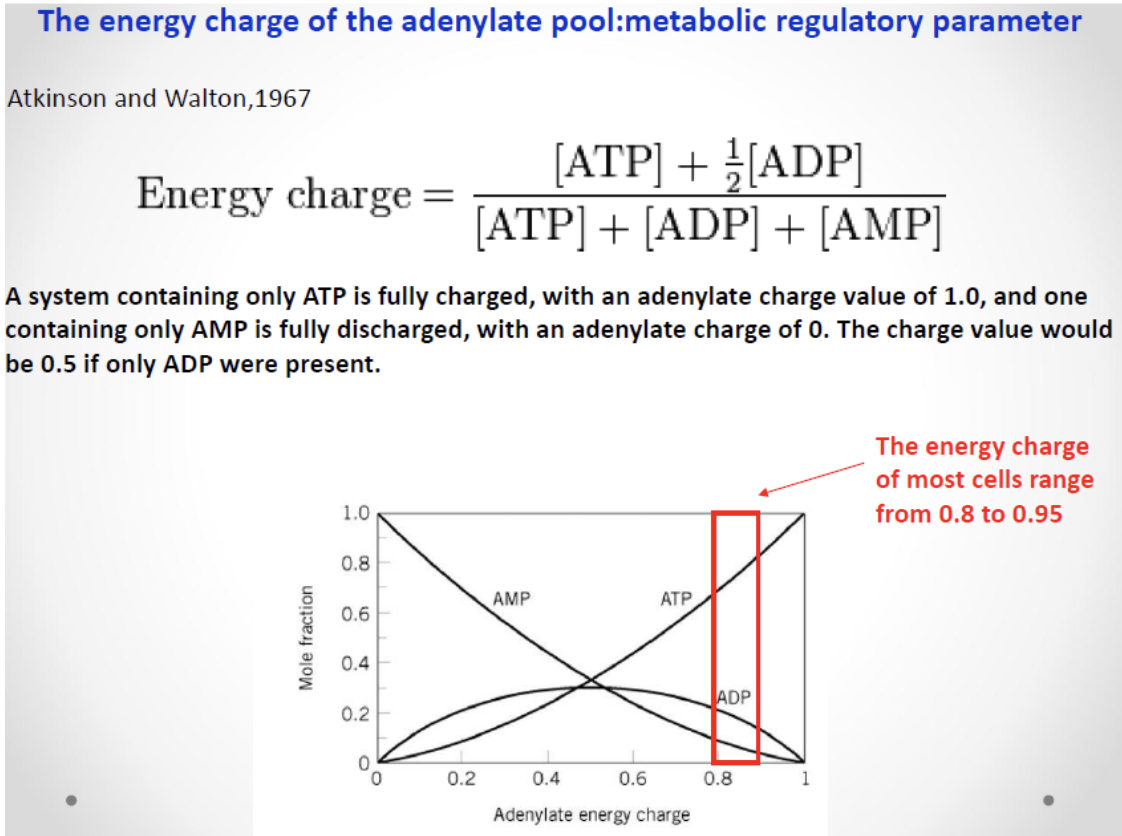
Sample calculations
If ATP = ADP = AMP = 10 mM, then EC = 0.5.
If ATP = 0, ADP = 10 mM, AMP = 0, then EC = 0.5.
If ATP = 10 mM, ADP = AMP = 0, then EC = 1.0.
Physiological energy charge
Most cells maintain an energy charge between 0.8 and 0.95, close to 1 (fully charged).
Metabolism and Cellular Structure
Metabolism is inseparable from cell structure and compartmentation. Membranes enable concentration gradients, separation from the environment, and selective permeability.
Proteins function as molecular machines through specific interactions determined by their three-dimensional structure, which depends on amino acid sequence.
Role of Enzymes in Metabolic Regulation
Metabolic regulation occurs at the enzyme level through:
- Compartmentalization
- Thermodynamic and kinetic constraints
- Allosteric regulation
- Covalent regulation (including reversible phosphorylation)
- Genetic regulation
Metabolic regulation: mechanisms maintaining molecular homeostasis.
Metabolic control: mechanisms changing pathway output over time.
Six Classes of Enzymes
| Class | Function | Example |
|---|---|---|
| Oxidoreductases | Catalyse redox reactions (electron transfer) | Dehydrogenases |
| Transferases | Transfer functional groups (e.g. methyl, phosphate) | Kinases |
| Hydrolases | Catalyse hydrolysis (use water to break bonds) | Proteases |
| Lyases | Break or form double bonds without ATP or water | Aldolase |
| Isomerases | Rearrange atoms within a molecule | Phosphoglucose isomerase |
| Ligases | Join two molecules using ATP | Synthetases |
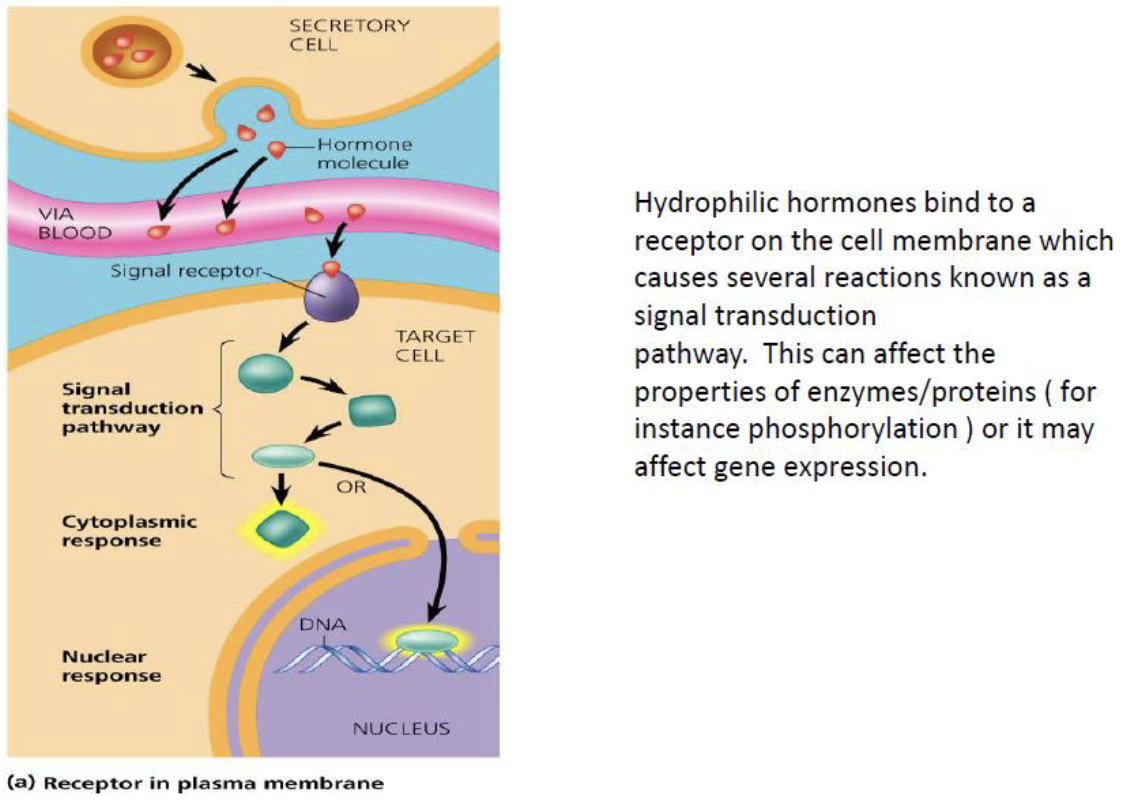
Hormone Signaling and Enzyme Regulation
Hormonal regulation of metabolism
- Hydrophilic hormones bind to receptors on the cell membrane → activate signal transduction pathways → modify enzyme activity (e.g. via phosphorylation) or alter gene expression.
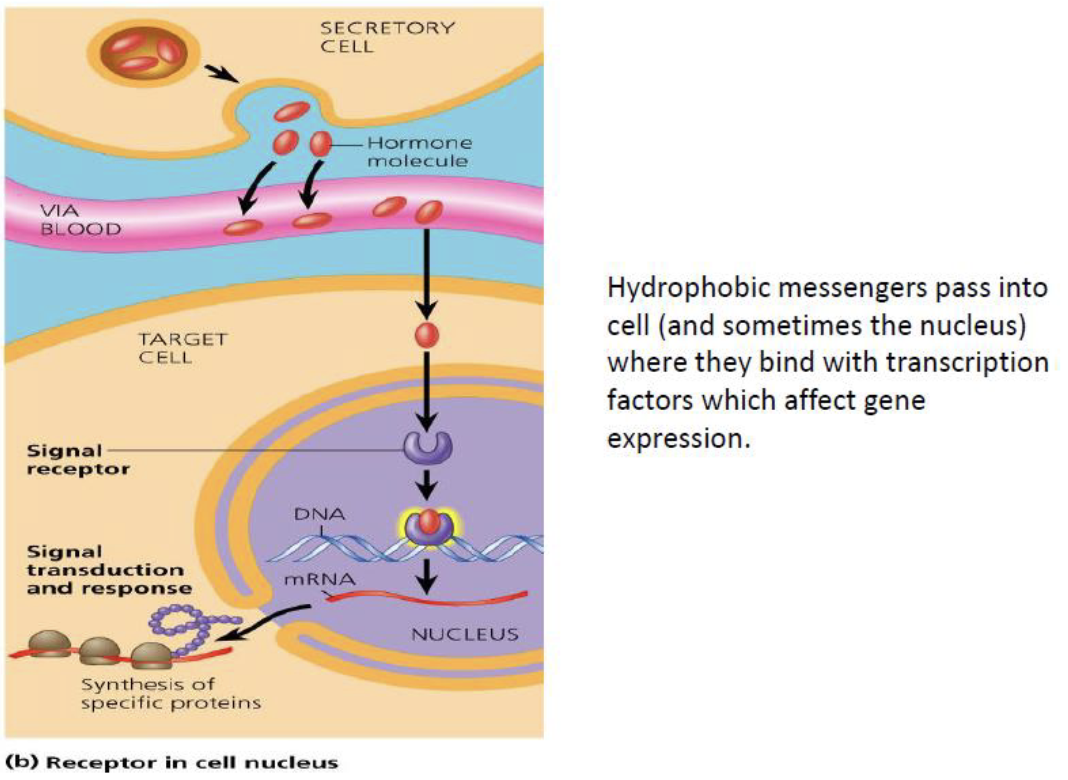
- Hydrophobic messengers diffuse into the cell (and sometimes the nucleus) → bind to intracellular receptors/transcription factors → regulate gene expression directly.
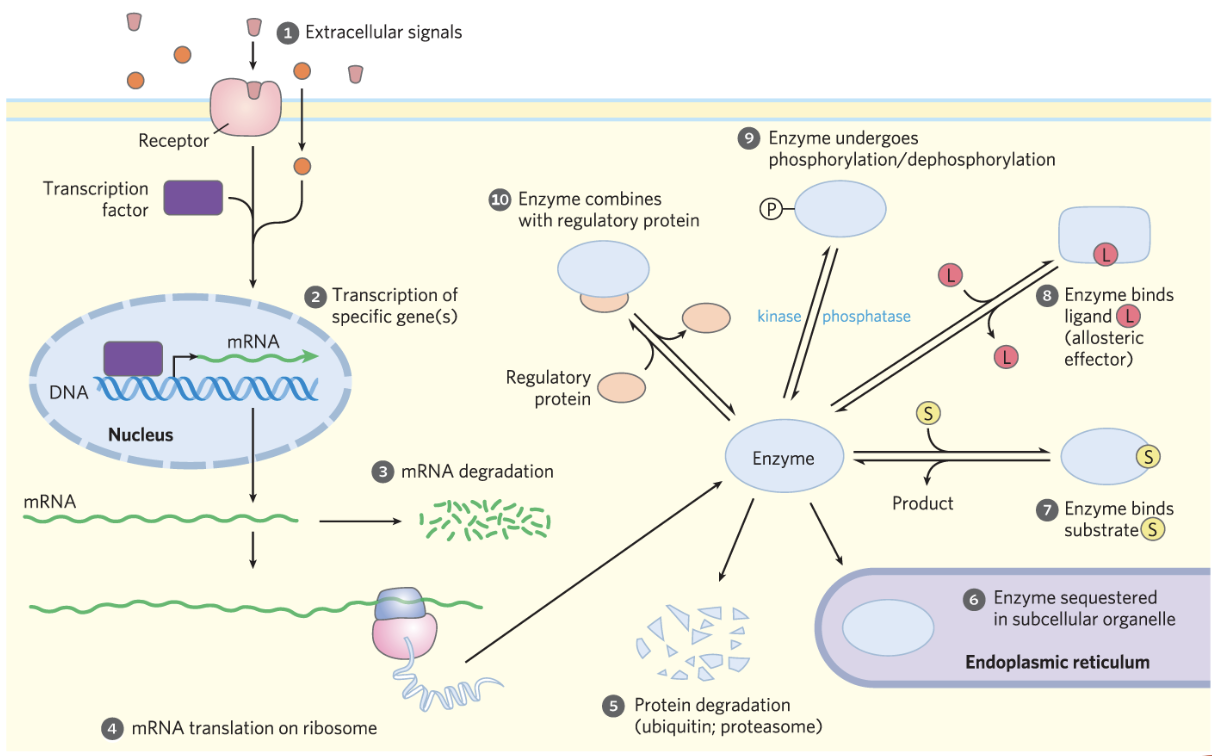
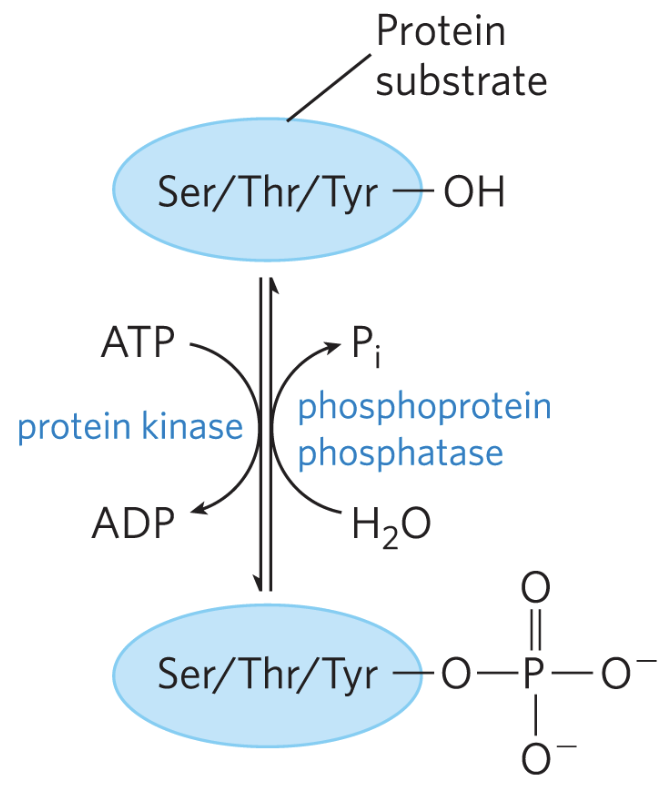
An irreversible reaction often corresponds to regulation at a rate-limiting enzyme step. A strongly negative helps but is not strictly required.
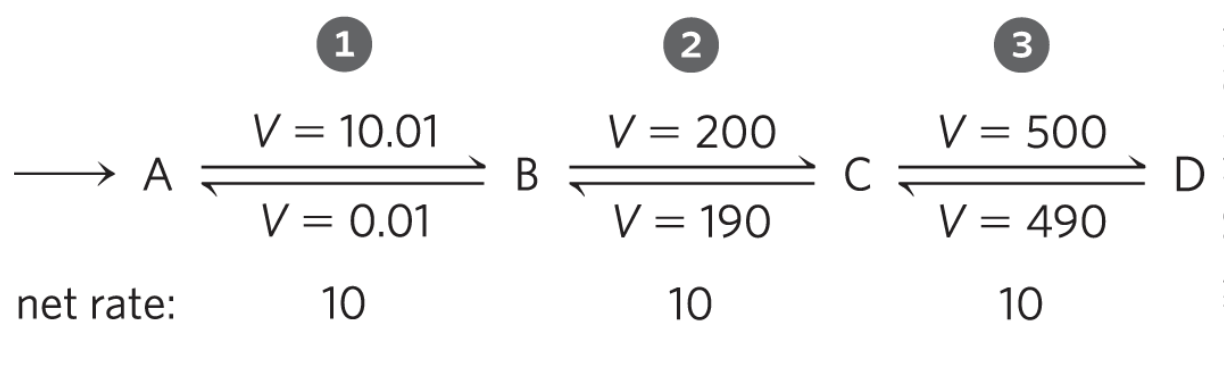
Roles of enzymes
Flashcards
Anki cloze
The three broad stages of catabolism are {1:polymer breakdown}, {1:redox coenzyme generation}, and {1:ATP production}.
Anki cloze
At metabolic {1:steady state}, intermediate concentrations are approximately {2:constant} because input and output fluxes are {2:balanced}.
Anki cloze
{1:Thermodynamics} determines reaction {2:direction}, while {1:kinetics} determines reaction {2:rate}.
Anki cloze
For , free energy is: {1:}.
Anki cloze
At equilibrium: {1:} and therefore {1:}.
Anki cloze
In metabolism, a reaction with positive can proceed by {1:coupling} to a reaction with sufficiently negative so that total is {2:negative}.
Anki cloze
Oxidation is loss of {1:electrons}; reduction is gain of {1:electrons}.
Anki cloze
Typical dehydrogenation half-reaction: {1:}.
Anki cloze
The relationship between free energy and redox potential is {1:}.
Anki cloze
In substrate-level phosphorylation, ATP is produced by {1:direct phosphate transfer} from a high-energy intermediate to {2:ADP}.
Anki cloze
In orthophosphoric ATP cleavage, products are typically {1:ADP and a phosphorylated substrate}.
Anki cloze
In pyrophosphoric ATP cleavage, ATP forms {1:AMP + PP_i}; hydrolysis of {2:} to {2:} helps pull reactions forward.
Anki cloze
Energy charge formula: {1:}.
Anki cloze
A pathway control point is often an {1:irreversible} step catalyzed by a {2:rate-limiting enzyme}.
Anki cloze
Main levels of metabolic regulation include {1:allosteric}, {1:covalent}, and {1:genetic} control.
Anki cloze
Equilibrium cannot sustain energy production because at equilibrium {1:}, meaning {1:no net free energy} is released.
Anki cloze
A compound is considered high-energy if its standard free energy of hydrolysis is {1:}.
Anki cloze
ATP provides energy primarily through {1:phosphoryl group transfer} to a substrate, not by {1:direct hydrolysis}.
Anki cloze
The three chemical reasons for ATP’s large of hydrolysis are {1:charge separation}, {1:resonance stabilization of }, and {1:greater solvation of products}.
Anki cloze
Most cells maintain an energy charge between {1:0.8} and {1:0.95}.
Anki cloze
The six enzyme classes are {1:oxidoreductases}, {1:transferases}, {1:hydrolases}, {1:lyases}, {1:isomerases}, and {1:ligases}.
Anki cloze
{1:Hydrophilic} hormones act at the {2:cell membrane receptor}, while {1:hydrophobic} hormones diffuse into the cell and act on {2:intracellular receptors or transcription factors}.
Anki cloze
Alzheimer’s disease is linked to impaired {1:cerebral glucose metabolism}; bipolar disorder is linked to {1:mitochondrial dysfunction}; colorectal cancer is linked to {1:hyperinsulinemia}.
TARGET DECK: MED::I::Signaling Pathways in Health and Disease::Metabolic Biochemistry::01 - Introduction to metabolism
TLDR - 01 - Introduction to Metabolism
What is Metabolism?
Metabolism is the complete set of enzyme-catalyzed reactions that produce energy and biosynthetic building blocks. It is compartmentalized across membranes and tightly regulated at both enzyme and gene levels.
Catabolism vs. Anabolism
| Catabolism | Anabolism | |
|---|---|---|
| Direction | Oxidative | Reductive |
| Energy | Releases ATP | Requires ATP |
| Example | Palmitate → 8 acetate (14 oxidations, +28 ATP) | 8 acetate → palmitate (14 reductions, −7 ATP) |
Pathway Types
- Linear (e.g., glycolysis)
- Branched — converging (e.g., → pyruvate) or diverging (e.g., from isopentenyl pyrophosphate)
- Cyclic (e.g., TCA cycle, ornithine cycle)
Thermodynamics & Kinetics
- Thermodynamics sets reaction direction:
- Kinetics sets reaction rate: controlled by enzymes
- At steady state: intermediate concentrations are constant; input flux = output flux
- At equilibrium: , so
Coupled Reactions
Unfavorable reactions () proceed when coupled to favorable ones so that . ATP hydrolysis () is the most common coupling agent.
Redox Reactions
- Oxidation = loss of electrons; often dehydrogenations ()
- ; electrons flow from low to high reduction potential
ATP Metabolism
| Mechanism | Products | Example |
|---|---|---|
| Orthophosphoric cleavage | X-P + ADP | Glucose → G6P |
| Pyrophosphoric cleavage | X-AMP + PP | Amino acid activation |
| Substrate-level phosphorylation | ATP | Glycolysis steps |
| Oxidative phosphorylation | ATP | Mitochondrial ETC |
Energy charge = ; ranges from 0 (fully depleted) to 1 (fully charged).
Metabolic Regulation
Control occurs at irreversible, rate-limiting steps via:
- Allosteric regulation — small molecule effectors
- Covalent modification — e.g., reversible phosphorylation
- Genetic regulation — enzyme expression levels
- Compartmentalization — separation by membranes
Regulation = maintaining homeostasis; Control = changing pathway output over time.
Enzyme Classes
| Class | Action |
|---|---|
| Oxidoreductases | Redox / electron transfer |
| Transferases | Group transfer (methyl, phosphate…) |
| Hydrolases | Hydrolysis |
| Lyases | Break/form double bonds (no ATP/water) |
| Isomerases | Isomer rearrangement |
| Ligases | Join molecules (requires ATP) |