TARGET DECK: MED::I::Signaling Pathways in Health and Disease::Physiology::02 - Potentials
Electro-Chemical Properties of Cells
Core Concept
Electro-chemical properties of cells carry important information. Neurons transmit electrical (and chemical) signals between cells (e.g., Neuron 1 → Neuron 2).
Resting Membrane Potential
Resting Membrane Potential Varies by Cell Type
| Cell Type | Resting Vm |
|---|---|
| Skeletal muscle cells | −95 mV |
| Smooth muscle cells | −50 mV |
| Neurons | −70 mV |
| Erythrocytes | −12 mV |
Anki cloze
The resting membrane potential of a neuron is {1:−70 mV}, while skeletal muscle cells have a resting Vm of {2:−95 mV}.
Chemical and Electrical Driving Forces
The Chemical Potential
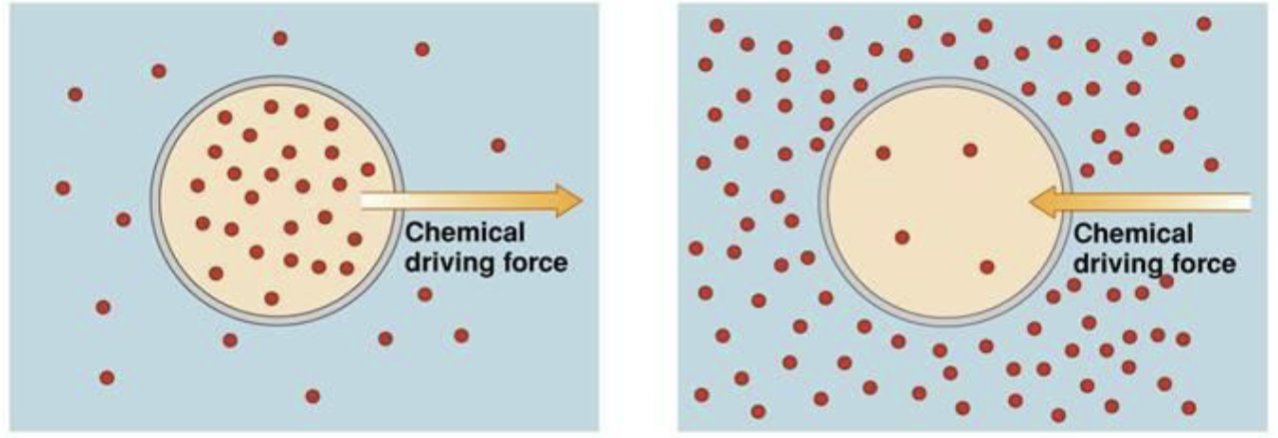
For a substance X with no electric charge and concentration , the Chemical Potential is:
For a solute in intracellular (int) and extracellular (ext) compartments:
The Electric Potential
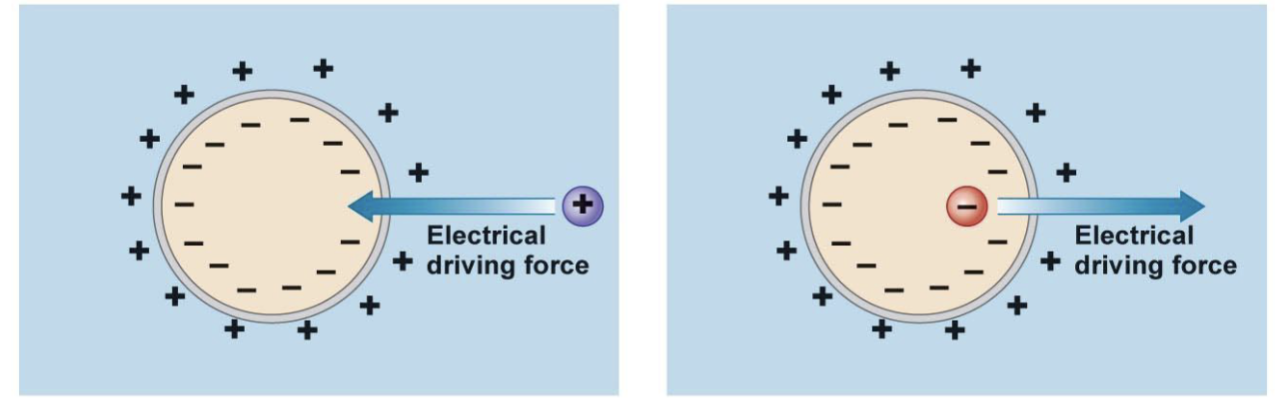
For a charged solute:
Constants
- = Gas constant = 8.314 J/(mol·K)
- = Temperature in K
- = valence of charged molecule (Na⁺ is 1, Ca²⁺ is 2)
- = Faraday constant = 96485.332 C·mol⁻¹
The Electrochemical Potential
At equilibrium, :
What is the condition for electrochemical equilibrium of an ion across a membrane?
, meaning
The Nernst Equation
If (potential difference between the inside and outside of the membrane generated by X), then:
Simplified Nernst Equation
Using instead of (since , i.e., ):
At 20°C:
At 37°C:
At 29.5°C:
Anki cloze
The Nernst equation at 37°C simplifies to {1:}.
Ion Concentrations and Equilibrium Potentials — Skeletal Muscle
| Ion (X) | (mM) | (mM) | (mV) |
|---|---|---|---|
| K⁺ | 4.5 | 155 | −95 |
| Na⁺ | 145 | 12 | +67 |
| Ca²⁺ | 1.0 | 10⁻⁴ | +123 |
| Cl⁻ | 116 | 4.2 | −19 |
| HCO₃⁻ | 24 | 12 | — |
Info
From the ion concentrations reported, the equilibrium potential () can be calculated using the Nernst Equation for each ion (, , ) involved in generating the resting membrane potential () of the skeletal muscle cell.
Warning
Compared to other excitable cells, skeletal muscle has unusually high Cl⁻ conductance, accounting for up to 85% of all membrane conductance at rest. (EC Aromataris, GY Rychkov, 2006)
Ion Concentrations and Equilibrium Potentials — Other Cell
| Ion (X) | (mM) | (mV) | |
|---|---|---|---|
| K⁺ | 120 | 0.038 | −88 |
| Na⁺ | 15 | 9.67 | +61 |
| Ca²⁺ | 10⁻⁴ | 10,000 | +123 |
| Cl⁻ | 15 | 9.6 | −47 |
| HCO₃⁻ | — | 1.6 | −13 |
Info
From the ion concentrations reported, the equilibrium potential () can be calculated using the Nernst Equation for each ion (, , ) involved in generating the resting membrane potential (). mV.
Nernst Equilibrium Potentials — Neuron ( mV)
| Ionic Species | Intracellular Concentration | Extracellular Concentration | Equilibrium Potential |
|---|---|---|---|
| Sodium (Na⁺) | 15 mM | 145 mM | = +60.60 mV |
| Potassium (K⁺) | 150 mM | 4 mM | = −96.81 mV |
| Calcium (Ca²⁺) | 70 nM | 2 mM | = +137.04 mV |
| Hydrogen ion (H⁺) | 63 nM (pH 7.2) | 40 nM (pH 7.4) | = −12.13 mV |
| Magnesium (Mg²⁺) | 0.5 mM | 1 mM | = +9.26 mV |
| Chloride (Cl⁻) | 10 mM | 110 mM | = −64.05 mV |
| Bicarbonate (HCO₃⁻) | 15 mM | 24 mM | = −12.55 mV |
What is the equilibrium potential for Na⁺ in a typical neuron?
= +60.60 mV (intracellular 15 mM, extracellular 145 mM)
What is the equilibrium potential for K⁺ in a typical neuron?
= −96.81 mV (intracellular 150 mM, extracellular 4 mM)
Origin of the Resting Membrane Potential
What is the cause of the membrane resting potentials?
- The electrogenic effect of the Na⁺/K⁺ pump (3 Na⁺ out / per 2 K⁺ in)
- The Donnan effect
- The asymmetric distribution of ions across the membrane (selective permeability of the membrane – K⁺ leak channels)
Number 3 is the main reason.
Relative Contributions
- A blockade of the Na⁺/K⁺ pump will induce a 4–5 mV change in the membrane resting potential.
- The Donnan effect is negligible for excitable cells because Na⁺ and Cl⁻ permeability are very low.
- The asymmetric distribution of ions across the membrane is what better explains the origin of the membrane resting potential.
What happens to membrane resting potential if the Na⁺/K⁺ pump is blocked?
A blockade of the Na⁺/K⁺ pump will induce only a 4–5 mV change in the membrane resting potential.
Anki cloze
The {1:asymmetric distribution of ions} across the membrane (selective permeability – K⁺ leak channels) is what best explains the origin of the resting membrane potential.
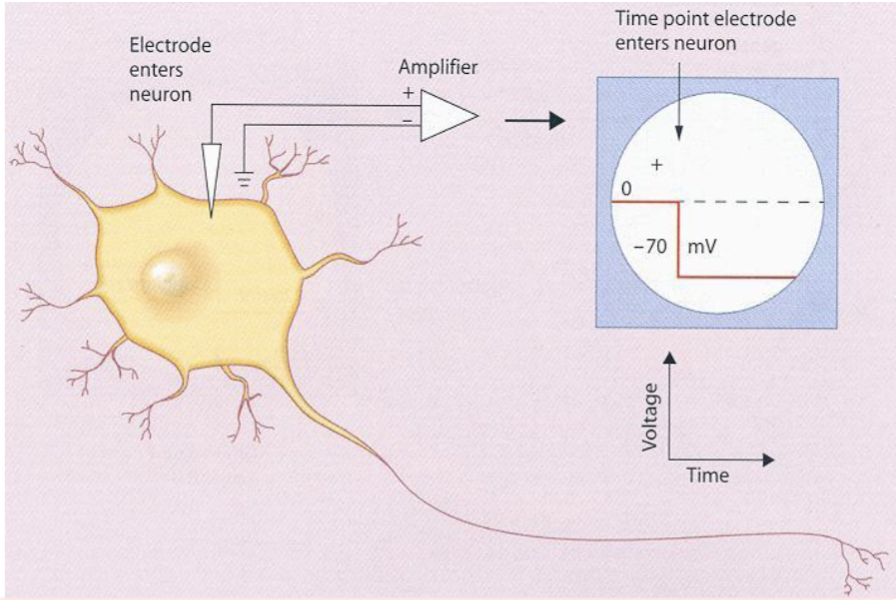
Recording Membrane Potential (Vm)
Technique
Glass micropipettes filled with concentrated salt solution serve as electrodes. Wires inserted into the back ends of the pipette and connected to a voltage amplifier and oscilloscope. This is a reliable technique started to develop in the late 1940s. Hodgkin, Huxley and Eccles won the Nobel Prize in 1963.
Intracellular recording micropipettes are filled with a solution with similar ionic composition to the intracellular environment. The diameter of the micropipette tip is small (< 1 μm) and can be inserted into the cell without damaging it.
Key Properties of Vm
- Neurons have a near the because the membrane is more permeable to K⁺ compared to Na⁺ and Cl⁻.
- The membrane of skeletal muscle cell has a higher permeability to Cl⁻ and will approximate .
- The actual is the consequence of a very small excess of charges in a very narrow region nearby the membrane.
- is directly proportional to the number of charges separated by the lipid bilayer.
- Reduction in charge separation = depolarization; increase = hyperpolarization.
Anki cloze
Neurons have a Vm near because the membrane is more permeable to {1:K⁺} compared to Na⁺ and Cl⁻.
Ion Channels and Vm
Two Major Classes of Ion Channels
- Passive channels: Always open; keep Vm steady in the absence of any signal ()
- Active (gated) channels: Can open or close in response to different triggers: ligand, mechanical stretching of the membrane, or changes in Vm (depolarization)
Electrotonic Potentials
Changes in Vm that do not lead to the opening of gated ion channels are passive responses termed electrotonic potentials. Hyperpolarizing potentials and small depolarizations are almost always electrotonic potentials.
Vr and Ion Distribution
is the result of the passive flux of individual ions through several passive ion channels. No single ion species is distributed equally across the 2 sides of a neuron membrane. Na⁺ and Cl⁻ are most abundant outside while K⁺ and anions are most abundant inside.
What are electrotonic potentials?
Changes in Vm that do not lead to the opening of gated ion channels are passive responses termed electrotonic potentials. Hyperpolarizing potentials and small depolarizations are almost always electrotonic potentials.
Determining Resting Potential: Three Hypothetical Situations
Figure 6-4 Kandel: Resting Potential and Open Ion Channels
A. Only K⁺ channels present:
K⁺ ions are in equilibrium and .B. K⁺ + a few Na⁺ channels:
Na⁺ ions diffuse into the cell; this influx begins to depolarize the membrane.C. Steady state (more K⁺ than Na⁺ channels):
The resting potential settles at a new level where the influx of Na⁺ is balanced by the efflux of K⁺. The aggregate conductance of K⁺ channels is much greater than that of Na⁺ channels because K⁺ channels are more numerous. A relatively small net driving force for K⁺ drives a current equal and opposite to the Na⁺ current driven by the much larger net driving force for Na⁺. This is a steady-state condition in which neither Na⁺ nor K⁺ is in equilibrium but the net flux of charge is null.
Info
In a resting nerve cell relatively few Na⁺ channels are open, so the membrane conductance of Na⁺ is quite low. Thus, despite the large chemical and electrical forces driving Na⁺ into the cell, the influx of Na⁺ is small. In contrast, many K⁺ channels are open in the membrane of a resting cell so that the membrane conductance of K⁺ is relatively large.
Because of the high relative conductance of K⁺ to Na⁺ in the cell at rest, the small net outward force acting on K⁺ is enough to produce a K⁺ efflux equal to the Na⁺ influx.
The Na⁺-K⁺ Pump
Na⁺-K⁺ Pump Properties
- Dissipation of ionic gradients is prevented by the Na⁺-K⁺ pump.
- Requires energy from ATP hydrolysis.
- Movement of ions through the pump is 10,000 times slower than the flow across ion channels.
- Electrogenic: extrudes 3 Na⁺ and brings in 2 K⁺ → net flux of ions outward.
- During periods of intense neuronal activity, increased influx of Na⁺ leads to increased Na⁺-K⁺ pump activity → prolonged outward current → prolonged hyperpolarizing after-potential that can last for several minutes.
- Inhibited by ouabain or digitalis plant alkaloids.
- The Na⁺-K⁺ pump is a P-type ATPase, like the Ca²⁺ pump.
Why is the Na⁺-K⁺ pump considered electrogenic?
Because it extrudes 3 Na⁺ and brings in only 2 K⁺, resulting in a net outward flux of positive charge.
Anki cloze
The Na⁺-K⁺ pump is inhibited by {1:ouabain} or {2:digitalis} plant alkaloids.
The Goldman-Hodgkin-Katz (GHK) Equation
Electrodiffusion Theory
The contemporary action of multiple ions on the membrane resting potential is a special case of the theory of electrodiffusion. To facilitate its application, some simplifications are made:
- The membrane is a homogeneous piece of constant thickness.
- The potential difference across the membrane changes linearly with distance (constant electric field — principle of the constant field).
- The movement of an ion across the membrane is independent from the movement of other ions (principle of independence).
- The permeability coefficient of the membrane to an ion X is constant along the entire membrane.
The Goldman-Hodgkin-Katz (GHK) equation allows calculation of the of a cell (neuron). depends on of each ion that can cross the membrane and on (cm/s) of the membrane for that ion. It lies at a level intermediate to , , and :
Info
This equation applies only when is not changing. When permeability to one ion is exceptionally high, the Goldman equation reduces to the Nernst equation for that ion.
Squid Giant Axon (Hodgkin and Katz)
At rest:
At the peak of the action potential:
What is the ratio PK:PNa:PCl in the squid giant axon at rest vs. at peak action potential?
At rest: 1.0 : 0.04 : 0.45. At peak of action potential: 1.0 : 20 : 0.45.
Equivalent Ci6ewù
ì+ù
ù
+rcuit Model of the Membrane
Membrane as a Capacitor
Info
The membrane is a lipid insulator with electrical capacitance (1 μF per cm² for a neuron). The number of ions that must move across the cell membrane to create Vm is a tiny fraction of the total number of ions in the cell.
Calculation for a Glial Cell
Assume a glial cell with membrane permeable to K⁺:
- mV ( V); cell diameter = 20 μm
Cell surface area cm²
F/cm² cm² F
Charge separation:
Because 1 mol of K⁺ contains 96,480 C:
With cell volume and intracellular mmol/L:
Intracellular K⁺ moles
Therefore:
Ohm’s Law Applied to Ion Channels
The current through a K⁺ channel:
This is a modified version of Ohm’s equation. is called the electrochemical driving force.
Using conductance:
is the electromotive force that pushes ion X through the membrane with conductance .
- If : is negative → inward current
- If : current flows outward
Anki cloze
The electrochemical driving force for an ion X is expressed as {1:}.
Depolarization and Hyperpolarization Conventions
Conventions
- When becomes less negative (more positive) → DEPOLARIZATION
- When becomes more negative (less positive) → HYPERPOLARIZATION
- An influx of positive ions or an outflux of negative ions → depolarization
- An outflux of positive ions or an influx of negative ions → hyperpolarization
- Conventionally, inward currents → depolarization; outward currents → hyperpolarization
⚠️ CARE with Cl⁻: conventions for Cl⁻ current direction are opposite to those for cations.
Cl⁻ Current
When (e.g., −35 mV), Cl⁻ leaves the cell, trying to bring Vm to .
When , Cl⁻ enters the cell.
Conventionally, currents below are inward (Cl⁻ moves in the opposite direction; negative ions leaving the cell = positive ions entering the cell). Currents above are outward: Cl⁻ enters the cell (negative ions coming in = positive ions going out).Note: K⁺ enters the cell for inward current ( mV) while Na⁺ leaves the cell for outward current ( mV).
Conventionally, an inward current (IC) is indicated by downward deflection, while an outward current (OC) is indicated by an upward deflection.
IC depolarizes the cell; OC hyperpolarizes the cell.
Membrane Equivalent Circuit Components
Two Elements
- Capacitor: The membrane acts as a dielectric between two flat surfaces (the 2 faces of the membrane) with charges. ; measured in Farads = Coulombs/Volt.
- Resistance / Electromotive Forces: Within ion channels. Ohm’s law: ; commonly the inverse is used: (conductance); ; measured in Siemens.
Specific Capacitance
The capacitance of a capacitor increases with the area of the plates and decreases with the separation between the plates:
where is the dielectric constant, is the membrane thickness. As the thickness of the membrane is only 2.5 nm, the of the membrane is very high, equivalent to about 1 μF/cm².
Membrane Current Components
The membrane current () has two components:
- Resistive ()
- Capacitive ()
Time Constant (τ)
Info
The membrane equivalent circuit has both a resistive and a capacitive component; has an exponential dynamic.
at time , , depends on the maximum steady-state value () and from the time constant :
is the time interval needed for to reach 63% of (37% less than ).
Anki cloze
The time constant τ = {1:} and represents the time needed for Vm to reach {2:63%} of its maximum value.
Passive Electrical Properties and Signal Propagation
Length Constant (λ)
Info
The conduction of the action potential faces two different kinds of resistance:
- : the internal (axial) resistance along the axon
- : the resistance across the membrane
If a constant current is injected for , the membrane capacitance is fully charged and membrane potential reaches a steady value. The variation of with distance depends on the relative values of and :
The length constant is:
where:
- = membrane resistance (per unit length)
- = axial (internal) resistance (per unit length)
The geometric form:
where:
- = membrane resistivity (Ω·cm²)
- = intracellular resistivity (Ω·cm)
- = axon radius
Info
is the distance between the point of origin of the potential () and the point where becomes of the original potential.
Anki cloze
The length constant λ is defined as the distance at which the voltage decays to {1:37% (1/e)} of its original value.
Length Constants in Mammals
- Unmyelinated axons: 0.5 mm
- Myelinated axons: 1–3 mm
Calculation: Myelinated Axon (λ = 3.0 mm)
Using , what distance X produces a depolarization of 15 mV (threshold) given mV?
Considering that the average internodal distance between two nodes of Ranvier is about 2 mm, was large enough to depolarize almost 3 nodes of Ranvier from the origin of the action potential. This works as a safety measure to ensure the propagation of the action potential.
Passive Spread and Axon Diameter
Role of Axial Resistance ( )
From Ohm’s law ():
- → current flow along the axon
- current → slower depolarization of adjacent segments
- High internal resistance slows conduction
Role of Membrane Capacitance ( )
The membrane behaves as a capacitor. More capacitance → more charge required to change . Higher slows voltage changes.
Effect of Axon Radius (r)
- → larger axon → much lower internal resistance
- → larger surface → higher capacitance
- Larger radius → → → voltage spreads farther
Important
Larger axons conduct faster because they have lower axial resistance and a larger length constant.
The speed of passive voltage spread depends on: lower and lower → faster spread.
Anki cloze
Larger axons conduct faster because they have {1:lower axial resistance} and a larger {2:length constant (λ)}.
Myelination and Conduction Velocity
Myelination: Second Strategy to Increase Conduction Speed
Myelin acts as an electrical insulator:
- decreases (thicker insulation → less charge needed)
- increases (less current leakage)
This reduces the time required to change membrane voltage.
Consequences:
- → faster charging of the membrane
- → current remains inside the axon and travels farther
- Overall: faster passive spread of depolarization
Myelination is more efficient because it decreases and increases .
How does myelination increase conduction velocity?
Myelin decreases membrane capacitance (Cm) and increases membrane resistance (rm), resulting in faster passive spread of depolarization along the axon.
Axon Classification by Diameter, Speed, and Function
| Group | Diameter (μm) | Speed (m/sec) | Sensory Receptors |
|---|---|---|---|
| Aβ | 13–20 | 80–120 | Proprioceptors of skeletal muscle |
| Aδ | 6–12 | 35–75 | Mechanoreceptors of skin |
| — | 1–5 | 5–30 | Pain, temperature |
| IV / C | 0.2–1.5 | 0.5–2 | Temperature, pain, itch |
Axon Caliber Facts
- Axon caliber (diameter) in mammalian PNS ranges from 0.1 μm to 20 μm.
- Unmyelinated axons: < 2 μm; myelinated axons: > 1–2 μm.
- In the human CNS, almost all axons with diameters greater than 0.2 μm are myelinated.
- A large myelinated axon may have up to 250 to 300 turns of myelin.
- The ratio between axon diameter and total nerve fiber diameter: 0.6–0.7.
- Between two adjacent myelin segments: approximately 1 μm long gaps called nodes of Ranvier.
- The nodal membrane is rich in voltage-gated Na⁺ channels and can generate an intense depolarizing inward Na⁺ current.
- This organization is metabolically favorable because current only flows at the nodes and Na⁺-K⁺ pumps spend less energy to restore chemical gradient.
Myelin Composition
| Component | CNS (%) | PNS (%) |
|---|---|---|
| Protein | 30.0 | 29.5 |
| Lipid | 70.0 | 70.5 |
Myelin Proteins
Lipids: cholesterol, phospholipid, and galactolipid. Composition of myelin in the CNS is similar to the PNS.
Proteins — CNS:
- Proteolipid protein (PLP)
- Myelin basic protein (MBP)
Proteins — PNS:
- Major peripheral nerve protein (P0)
- A smaller concentration of PLP
Saltatory Conduction
Info
The action potential is transmitted in a non-decremental way because it is regenerated:
- In every spot in an unmyelinated axon
- Only at the nodes of Ranvier in a myelinated axon (saltatory conduction — slowing down at the Ranvier node)
Role of Juxtaparanodal K⁺ Channels
Both central and peripheral myelinated axons have fairly high densities of K⁺ channels under the myelin sheath near the two ends of each internodal segment. Their normal function is to suppress any action potential that may be generated by axon membrane under the myelin sheath. In demyelinating diseases, these channels become exposed and thus inhibit the ability of the bare axon to conduct action potentials.
What is saltatory conduction?
Saltatory conduction is the propagation of an action potential along a myelinated axon, where the AP jumps from node of Ranvier to node of Ranvier rather than being regenerated continuously. It is non-decremental.
Action Potentials
Definition and Function
Action potentials are transient and regenerative electrical signals whose amplitude does not tend to decrease along the axon length. In other types of cells, their main function is to activate intracellular processes. In muscle cells, an action potential is the first step in the chain of events leading to contraction.
Historical Note
In 1938, Cole and Curtis were the first to describe that the action potential was associated with an increase in membrane conductance. 10 years later, Hodgkin and Huxley drew the hypothesis that the action potential initiates as a consequence of a depolarization that induces a change in membrane conductance — it becomes permeable to Na⁺ rather than K⁺.
Voltage Clamp Technique
Info
Hodgkin and Huxley were able to describe the ion currents associated with action potentials by using the voltage clamp technique on the squid giant axon.
To study ion currents underlying AP generation, we only need to analyze the resistive currents, not the capacitive currents. Capacitive currents () only flow when is changing; during the voltage clamp experiment, only flows at the beginning and at the very end of the step change in .
= leakage current
Characteristics of the Action Potential
Threshold and All-or-None Behavior
- Characteristics of the action potential are its threshold and all-or-none behavior.
- A small subthreshold depolarization increases but also increases and .
- Because of the high voltage sensitivity and rapid kinetics, the increase in exceeds and .
- The point when is regenerative (threshold) is when form from outward becomes inward.
Anki cloze
The threshold of an action potential is reached when {1:} transitions from net outward to net inward current.
Phases of the Action Potential
| Phase | Description |
|---|---|
| Phase 0 (Depolarization) | Opening of voltage-gated Na⁺ channels |
| Phase 1 (Partial repolarization) | Closure of Na⁺ channels, initial K⁺ activation |
| Phase 2 (Plateau) | Balance of inward Ca²⁺ and outward K⁺ (cardiac) |
| Phase 3 (Repolarization) | Closure of Na⁺ channels; opening of voltage-gated K⁺ channels |
| Phase 4 (Hyperpolarization / Resting) | Voltage-gated K⁺ channels remain open after potential reaches threshold level; slow return to RMP |
The Hodgkin Cycle
Important
Membrane depolarization → Opening of Na⁺ voltage-gated channels → More depolarization → More channels open → … (positive feedback)
This Hodgkin cycle explains the explosive increase of Vm during the depolarization phase, once threshold is reached.
Inactivation and Refractory Periods
Once opened, Na⁺ voltage-gated channels go through a longer cycle: channel open → inactive → closed. This explains the duration of the absolute refractory period.
- Absolute refractory period: Na⁺ channels in inactivated state (~0.5–1.0 ms)
- Relative refractory period: Channels recovering from inactivation; a larger-than-normal stimulus can trigger an AP
What is the Na⁺ channel cycle that explains the absolute refractory period?
Na⁺ voltage-gated channels cycle through: open → inactive (refractory, ~0.5–1.0 ms) → closed. The inactive state prevents re-opening, creating the absolute refractory period.
Conduction of the Action Potential
Info
The local depolarization spreads passively to adjacent areas. Na⁺ channels open locally in response to a stimulus, generating an action potential. Some depolarizing current passively flows down the axon. Local depolarization causes neighboring Na⁺ channels to open and generates a new action potential.
Axon Hillock
The action potential is typically generated at the axon hillock. This region has the lowest threshold because of the number of voltage-gated Na⁺ channels. It also has other voltage-gated channels that are very sensitive to relatively small deviations from (including M-type and A-type K⁺ channels or low voltage-activated Ca²⁺ channels).
Ion Channels — Types and Properties
Extracellular and Intracellular Ion Concentrations
| Ion | Squid Neuron Intracellular (mM) | Squid Neuron Extracellular (mM) | Mammalian Neuron Intracellular (mM) | Mammalian Neuron Extracellular (mM) |
|---|---|---|---|---|
| K⁺ | 400 | 20 | 140 | 5 |
| Na⁺ | 50 | 440 | 5–15 | 145 |
| Cl⁻ | 40–150 | 560 | 4–30 | 110 |
| Ca²⁺ | 0.0001 | 10 | 0.0001 | 1–2 |
Classes of Voltage-Gated Ion Channels
IUPHAR Classification of Voltage-Gated Ion Channels
- Calcium-Activated Potassium Channels
- CatSper and Two-Pore Channels
- Cyclic Nucleotide-Regulated Channels
- Inwardly Rectifying Potassium Channels
- Transient Receptor Potential Channels
- Two-P Potassium Channels
- Voltage-Gated Calcium Channels
- Voltage-Gated Potassium Channels
- Voltage-Gated Sodium Channels
Voltage-Gated K⁺ Channel Subtypes
Info
Most neurons contain voltage-gated Ca²⁺ channels that open in response to depolarization and help depolarizing the cell (there is a strong electrochemical gradient for Ca²⁺; mV).
Some neurons have voltage-gated Cl⁻ channels that contribute to membrane repolarization.
Many neurons have HCN (hyperpolarization-activated cation) channels permeable to both K⁺ and Na⁺, with a reversal potential around –40 to –30 mV. They give rise to an inward depolarizing current when the membrane repolarizes or becomes hyperpolarized.
Types of Voltage-Activated K⁺ Channels
- Delayed rectifier: The slowly activating K⁺ channel described by Hodgkin and Huxley.
- Calcium-activated K⁺ channel: Activated by increase in intracellular Ca²⁺ when nearby voltage-gated Ca²⁺ channels open. One subclass is voltage-dependent; Ca²⁺ binding shifts voltage-gating to allow opening at more negative potentials.
- A-type K⁺ channel: Activated rapidly by depolarization; also inactivates rapidly if depolarization is prolonged.
- M-type K⁺ channel: Requires only small depolarizations from resting potential; however, activates very slowly (tens of milliseconds). Distinctive feature: closed by acetylcholine.
There are at least five major types of voltage-gated Ca²⁺ channels and eight types of voltage-gated Na⁺ channels expressed in the nervous system.
What is distinctive about the M-type K⁺ channel?
The M-type K⁺ channel requires only small depolarizations to open, activates very slowly (tens of milliseconds), and is distinctively closed by the neurotransmitter acetylcholine.
Modulation of Voltage-Gated Channels
Info
In a typical neuron the opening and closing of certain voltage-gated ion channels can be modulated by various cytoplasmic factors.
Calcium concentration is one important cytoplasmic factor. Intracellular free Ca²⁺ is very low (~10⁻⁷ M); when voltage-activated Ca²⁺ channels open, its concentration may change many fold → calcium-activated voltage-sensitive K⁺ channels may open.
Changes in intracellular Ca²⁺ can also influence neurotransmitter release and gene expression.
The activity of voltage-gated channels can also be regulated by neurotransmitters through the recruitment of second-messenger pathways. These pathways typically affect the kinetics or voltage sensitivity of channel gating.
Ion Selectivity of Channels
Hydration and Selectivity Filter
Because of their electric charge, ions interact with polar molecules of the solvent (solvation; with water: hydration). Ions lose their hydration while entering a channel. Cation channels use the C=O bond to filter for cations (especially the interaction between O²⁻ and the ion).
- Na⁺ does not enter K⁺ channels: Na⁺ has a smaller radius and its interaction with water is stronger; its small diameter precludes interaction with O²⁻ in the selectivity filter.
- Na⁺ channels are larger than K⁺ channels and allow the passage of one Na⁺ still hydrated with two molecules of water — this is energetically favorable.
Two Major Classes of Voltage-Gated Channels
1. Cation Channels
Info
The main function of cation channels is generation and propagation of cell bioelectricity.
A) Na⁺ voltage-gated channels: Mostly involved in membrane depolarization.
B) Ca²⁺ voltage-gated channels: Mostly involved in mediating specific cell responses.
C) K⁺ voltage-gated channels: Mostly involved in the fine tuning of membrane potentials.
Voltage-Gated Na⁺ Channels (Nav)
Structure
Formed from a single polypeptide (α-subunit) consisting of 4 domains (I–IV), each with 6 transmembrane segments. Of each domain:
- The 4th segment (S4) contains positively charged lysine and arginines → primarily responsible for voltage sensing
- S5 and S6 segments form the loop of the channel pore region
Voltage-gated sodium channels associate single transmembrane proteins called β-subunits (with an immunoglobulin-like extracellular domain) that regulate Nav channels.
The four domains pair their voltage-sensitive segments (S4) and segments that build the wall of the pore (S5, S6).
TTX-Sensitive vs. TTX-Resistant
There are 2 distinct types of voltage-gated sodium channels:
- Tetrodotoxin-sensitive (TTX-S Na⁺ channel): TTX binds with nanomolar affinity
- Tetrodotoxin-resistant (TTX-R Na⁺ channel): TTX binds with low micromolar affinity; located primarily in cardiac tissue
Anki cloze
Voltage-gated Na⁺ channels are formed from a single α-subunit with {1:4 domains}, each containing {2:6} transmembrane segments. The {3:S4} segment is responsible for voltage sensing.
Nav Channel Nomenclature and Distribution
| Channel | Gene | Chromosomal Location | TTX Sensitivity | Major Tissue | Effect of Mutation |
|---|---|---|---|---|---|
| Nav1.1 | SCN1A | 2q24 | Sensitive | CNS, PNS | Epilepsy |
| Nav1.2 | SCN2A | 2q23-24 | Sensitive | CNS, PNS | Epilepsy |
| Nav1.3 | SCN3A | 2q24 | Sensitive | CNS, PNS | None reported |
| Nav1.4 | SCN4A | 17q23-25 | Sensitive | Skeletal muscle | Myotonia, periodic paralysis |
| Nav1.5 | SCN5A | 3p21 | Resistant | Heart | Long QT, Brugada syndrome, progressive familial heart block |
| Nav1.6 | SCN8A | 12q13 | Sensitive | CNS, PNS | Cerebellar atrophy |
| Nav1.7 | SCN9A | 2q24 | Sensitive | PNS | Increased and decreased pain sensitivity |
| Nav1.8 | SCN10A | 3p21-24 | Resistant | PNS (PAs) | None reported |
| Nav1.9 | SCN11A | 3p21-24 | Resistant | PNS | None reported |
| Nax | SCN6/7A | 2q21-23 | Non-functional | Glia | — |
What cardiac channelopathies are associated with Nav1.5 mutations?
Nav1.5 (SCN5A) mutations are associated with Long QT syndrome, Brugada syndrome, and progressive familial heart block.
Tetrodotoxin (TTX)
Tetrodotoxin (TTX)
- Found in the family of puffer fish (Tetraodontidae, >120 species) in skin, liver, intestine, and gonads.
- In humans, TTX is deadly: blocks action potentials in nerves by binding to the voltage-gated fast Na⁺ channels in nerve cell membranes, preventing affected nerve cells from firing.
- The binding site is located at the pore opening of the voltage-gated Na⁺ channel.
- In Japan, puffer fish meat is a delicacy called fugu, prepared only by licensed chefs trained for almost 3 years.
- TTX is apparently produced by Vibrio bacteria within the digestive system.
- Captain James Cook and his German naturalists (J.R. Forster Jr. and J.A.G. Forster) were the first to record TTX poisoning during the second Cook’s voyage (1774).
TTX Clinical Grading (Fukuda and Tani, 1941)
| Degree | Symptoms |
|---|---|
| First | Oral numbness and paraesthesia, sometimes accompanied by gastrointestinal symptoms (nausea) |
| Second | Numbness of face and other areas, advanced paraesthesia, motor paralysis of extremities, incoordination, slurred speech, but still normal reflexes |
| Third | Gross muscular incoordination, aphonia, dysphagia, dyspnoea, cyanosis, drop in blood pressure, fixed/dilated pupils, precordial pain, but victims are still conscious |
| Fourth | Severe respiratory failure and hypoxia, severe hypotension, bradycardia, cardiac arrhythmia, heart continues to pulsate for a short period |
Info
Local anesthetics such as lidocaine and procaine prevent the generation of action potentials by inhibiting voltage-gated Na⁺ channels of sensory neurons.
How does TTX block action potentials?
TTX binds to the pore opening of voltage-gated fast Na⁺ channels, blocking ion flow and preventing nerve cell firing.
Batrachotoxin (BTX)
Batrachotoxin (BTX)
- Produced by the golden poison-dart frog (Phyllobates terribilis), endemic to the Pacific coast of Colombia.
- Released from skin glands when stressed or scared; extremely potent cardiotoxic and the most potent alkaloid neurotoxin known.
- In the PNS, BTX binds selectively and almost irreversibly to the alpha subunit of voltage-gated Na⁺ channels, which become persistently active (open) at the resting membrane potential.
- The permanent Na⁺ influx permanently depolarizes the cell membrane, blocking any AP.
- BTX also interferes with AP conduction in the heart, causing arrhythmias, extrasystoles, and ventricular fibrillation.
- Frogs do not produce BTX themselves; they obtain it from their diet, possibly from Choresine beetles which contain BTX in high concentrations. These beetles likely obtain the BTX backbone from phytosterols in the plants they eat.
- Frogs tolerate BTX because they have evolved voltage-gated Na⁺ channels that are unaffected by BTX.
What is the mechanism of batrachotoxin (BTX)?
BTX binds selectively and almost irreversibly to the alpha subunit of voltage-gated Na⁺ channels, causing them to become persistently open at resting membrane potential, permanently depolarizing the cell and blocking action potential generation.
Voltage-Gated Ca²⁺ Channels (Cav)
Structure
Closely related to Nav channels; formed by a single polypeptide (α-subunit) consisting of 4 domains, each with 6 transmembrane segments. Positively charged arginines or lysines in S4 are responsible for voltage sensing; S5 and S6 form the pore loop.
Associated with auxiliary subunits: an α2-δ complex, an intracellular β-subunit, and occasionally a γ-subunit with 4 transmembrane segments.
Physiological Function and Pharmacology of Ca²⁺ Channels
| Channel | Current | Localization | Specific Antagonists | Cellular Functions |
|---|---|---|---|---|
| Cav1.1 | L | Skeletal muscle; transverse tubules | Dihydropyridines; phenylalkylamines; benzothiazepines | Excitation-contraction coupling |
| Cav1.2 | L | Cardiac myocytes; smooth muscle; endocrine cells; neuronal cell bodies; proximal dendrites | Dihydropyridines; phenylalkylamines; benzothiazepines | Excitation-contraction coupling; hormone release; regulation of transcription; synaptic integration |
| Cav1.3 | L | Endocrine cells; neuronal cell bodies and dendrites; cardiac atrial myocytes and pacemaker cells; cochlear hair cells | Dihydropyridines; phenylalkylamines; benzothiazepines | Hormone release; regulation of transcription; synaptic regulation; cardiac pacemaking; hearing; neurotransmitter release from sensory cells |
| Cav1.4 | L | Retinal rod and bipolar cells; spinal cord; adrenal gland; mast cells | Phenylalkylamines; benzothiazepines | Neurotransmitter release from photoreceptors |
| Cav2.1 | P/Q | Nerve terminals and dendrites; neuroendocrine cells | ω-Agatoxin IVA | Neurotransmitter release; dendritic Ca²⁺ transients; hormone release |
| Cav2.2 | N | Nerve terminals and dendrites; neuroendocrine cells | ω-Conotoxin-GVIA | Neurotransmitter release; dendritic Ca²⁺ transients; hormone release |
| Cav2.3 | R | Neuronal cell bodies and dendrites | SNX-482 | Repetitive firing; calcium transients |
| Cav3.1 | T | Neuronal cell bodies and dendrites; cardiac and smooth muscle myocytes | None | Pacemaking; repetitive firing |
| Cav3.2 | T | Neuronal cell bodies and dendrites; cardiac and smooth muscle myocytes | None | Pacemaking; repetitive firing |
L = Long lasting; P = Purkinje; N = Neither T nor L; R = Resistant to blockers; T = Transient
What type of Ca²⁺ channel is responsible for excitation-contraction coupling in cardiac muscle, and what drug class blocks it?
Cav1.2 (L-type); blocked by dihydropyridines, phenylalkylamines, and benzothiazepines.
Voltage-Gated K⁺ Channels (Kv)
Structure
Built from 4 monomers, each with 6 transmembrane sections, with functions similar to Nav and Cav channels. The four monomers also pair their functionally analogous sections.
Kv Channel Families (full table)
| IUPHAR | HGNC | Other |
|---|---|---|
| Kv1.1 | KCNA1 | Shaker-related family |
| Kv1.2 | KCNA2 | |
| Kv1.3 | KCNA3 | |
| Kv1.4 | KCNA4 | |
| Kv1.5 | KCNA5 | |
| Kv1.6 | KCNA6 | |
| Kv1.7 | KCNA7 | |
| Kv1.8 | KCNA10 | |
| Kv2.1 | KCNB1 | Shab-related family |
| Kv2.2 | KCNB2 | |
| Kv3.1 | KCNC1 | Shaw-related family |
| Kv3.2 | KCNC2 | |
| Kv3.3 | KCNC3 | |
| Kv3.4 | KCNC4 | |
| Kv4.1 | KCND1 | Shal-related family |
| Kv4.2 | KCND2 | |
| Kv4.3 | KCND3 | |
| Kv5.1 | KCNF1 | Modifier |
| Kv6.1 | KCNG1 | Modifiers |
| Kv6.2 | KCNG2 | |
| Kv6.3 | KCNG3 | |
| Kv6.4 | KCNG4 | |
| Kv7.1 | KCNQ1 | KVLQT1 |
| Kv7.2 | KCNQ2 | KQOT2 |
| Kv7.3 | KCNQ3 | |
| Kv7.4 | KCNQ4 | |
| Kv7.5 | KCNQ5 | |
| Kv8.1 | KCNV1 | Modifiers |
| Kv8.2 | KCNV2 | |
| Kv9.1 | KCNS1 | Modifiers |
| Kv9.2 | KCNS2 | |
| Kv9.3 | KCNS3 | |
| Kv10.1 | KCNH1 | eag1 |
| Kv10.2 | KCNH5 | eag2 |
| Kv11.1 | KCNH2 | erg1 |
| Kv11.2 | KCNH6 | erg2 |
| Kv11.3 | KCNH7 | erg3 |
| Kv12.1 | KCNH8 | elk1, elk3 |
| Kv12.2 | KCNH3 | elk2 |
| Kv12.3 | KCNH4 | elk |
2. Anion Channels
CLC (Cl⁻ Voltage-Gated Channels)
The main voltage-gated anion channel. CLC-1 is particularly abundant in the membrane of myocytes.
Structure: Built by dimers of monomers, each made from 10–12 transmembrane helices. CLC voltage sensitivity is not given by a specific segment, but by the movement of cations and anions associated with its complex structure. They are not selective for Cl⁻ and are involved in setting and restoring the resting membrane potential of skeletal muscles.
Other Important Anion Channels
- The cystic fibrosis transmembrane conductance regulator (CFTR) is another important anion channel, but it is not voltage-sensitive.
- GABA-A and Glycine receptors are also Cl⁻ channels.
What is the function of CLC-1 channels in skeletal muscle?
CLC-1 channels are not selective for Cl⁻ and are involved in setting and restoring the resting membrane potential of skeletal muscles.
Channel Oligomeric Structure
Info
Each subunit in a voltage-gated channel consists of six helical segments. Channels are usually oligomeric complexes composed of multiple subunits:
Channel Type Oligomeric Assembly Voltage-gated Na⁺, Ca²⁺, and K⁺ channels; Ca-release channel Tetramer Nicotinic ACh receptor channel Pentamer Half gap-junction channel (connexon, made up of six connexins) Hexamer
Gap Junctions
Info
Connexons are made up of six connexin proteins. Two connexons from adjacent cells pair to form a gap junction, creating a hydrophilic channel with a 2–4 nm intercellular space.
Connexin structure includes:
- Transmembrane domain (M1–M4)
- Extracellular loops (E1 and E2)
- Cytoplasmic loop (CL)
- C-terminus (COOH)
Cardiac Action Potentials
Conduction System of the Heart
Info
The cardiac conduction system includes: SA node → Interatrial tract (Bachmann’s bundle) → AV node → Bundle of His → Left bundle branch (anterosuperior + posteroinferior) + Right bundle branch → Purkinje fibers → Ventricular muscle.
The initiation time, shape, and duration of AP are distinctive for different parts of the heart, reflecting their different functions.
Cardiac AP Phases — Ventricular Myocyte
| Phase | Name | Description |
|---|---|---|
| Phase 0 | Depolarization | Rapid Na⁺ influx (INa) |
| Phase 1 | Partial repolarization | Closure of Na⁺ channels; initial K⁺ activation |
| Phase 2 | Plateau | Balance of inward Ca²⁺ (ICa) and outward K⁺ (IK) |
| Phase 3 | Repolarization | K⁺ channels open; IK drives repolarization |
| Phase 4 | Resting potential | — |
ERP = effective refractory period; RRP = relative refractory period
Four Major Membrane Currents of Cardiac AP
- INa (Na⁺ current): Responsible for the rapid depolarizing phase in atrial and ventricular muscle and Purkinje fibers.
- ICa (Ca²⁺ current): Responsible for the rapid depolarizing phase in SA node and AV node; in all cardiomyocytes responsible for the plateau phase which triggers contraction.
- IK (K⁺ current): Responsible for the repolarizing phase in all cardiomyocytes.
- If (pacemaker/“funny” current): Responsible for the rate of pacemaker activity in SA nodal cells, AV nodal cells, and Purkinje fibers.
Anki cloze
In cardiac AP, the plateau (phase 2) results from the balance between inward {1:Ca²⁺ current (ICa)} and outward {2:K⁺ current (IK)}.
Ca²⁺-Induced Ca²⁺ Release (CICR) in Cardiac Muscle
Important
Cardiac contraction has an absolute requirement for Ca²⁺ influx through L-type Ca²⁺ channels during AP. This induces an increase in cellular Ca²⁺ concentration that is not, however, sufficient to initiate contraction.
Thus, Ca²⁺ influx by L-type Ca²⁺ channels is greatly amplified by Ca²⁺-induced Ca²⁺ release (CICR) from the SR through Ca²⁺-release channels. Since these channels remain open for a longer period than L-type Ca²⁺ channels, the contribution of CICR is far greater than that of L-type channels.
It appears that each L-type Ca²⁺ channel controls only one SR Ca²⁺-release channel due to their physical proximity.
The membrane of cardiomyocytes can extrude Ca²⁺ by means of the Na⁺/Ca²⁺ exchanger (NCX) and the Ca²⁺ pump.
What is Ca²⁺-induced Ca²⁺ release (CICR)?
CICR is the amplification of Ca²⁺ influx through L-type Ca²⁺ channels by triggering Ca²⁺ release from the SR through Ca²⁺-release channels. Each L-type Ca²⁺ channel controls one SR Ca²⁺-release channel via physical proximity.
SA Node Action Potential and Pacemaker Current
SA Node AP Phases
Phase Description Phase 0 Depolarization due to opening of L-type Ca²⁺ channels at threshold Phase 2 Repolarization Phase 3 Repolarization Phase 4 Spontaneous depolarization (pacemaker)
Pacemaker / "Funny" Current (If)
- The most important current of pacemaker cells.
- Received its name for its fundamental role in initiating depolarization of SA cells, AV cells, and Purkinje fibers, and for the unusual property of being activated during hyperpolarization (end of phase 3).
- If is an inward current produced by HCN (hyperpolarization-activated, cyclic nucleotide-gated) channels responding to both voltage and chemical messenger (cAMP).
- HCN channels conduct both K⁺ and Na⁺; reversal potential of If is around –20 mV (between mV and mV in the heart).
- If is concomitant with an inward Ca²⁺ current from the phasic opening of transient Ca²⁺ voltage-dependent channels (iCaT).
- HCN channels do not conduct at positive potentials but are slowly activated by hyperpolarization characterizing the end of phase 3.
What is the "funny current" (If) and what channels carry it?
If is an inward depolarizing pacemaker current carried by HCN (hyperpolarization-activated, cyclic nucleotide-gated) channels, which conduct both K⁺ and Na⁺. It is activated by hyperpolarization at the end of phase 3.
HCN Channels
Info
HCN channels have 4 similar or identical α-subunits, each having 6 transmembrane segments (like other voltage-dependent K⁺ channels).
The channel is modulated by cAMP through a direct action on the channel itself, not by phosphorylation (unlike other ionic channels). cAMP binding induces a conformational change that increases the probability that the channel is open during hyperpolarization. This allosteric link during the opening status stabilizes the opening conformation.
Anki cloze
HCN channels are modulated by {1:cAMP} through a direct allosteric action on the channel, {2:not by phosphorylation}, increasing the probability of opening during hyperpolarization.
Smooth Muscle Action Potentials
Unitary (Visceral) Smooth Muscle
Info
Unitary smooth muscle is the predominant smooth muscle type within the walls of visceral organs (gastrointestinal tract, uterus, many blood vessels). Also referred to as visceral smooth muscle.
Action potentials in unitary smooth muscle have a slower upstroke and longer duration (up to ~100 ms) than in skeletal muscle (~2 ms).
The AP in a smooth muscle cell can be:
- i) a simple spike
- ii) a spike followed by a plateau
- iii) a series of spikes on top of slow waves of Vm
Info
The depolarizing phase of AP reflects the opening of voltage-gated Ca²⁺ channels. The inward Ca²⁺ current further depolarizes the membrane causing more Ca²⁺ channels to open. AP in smooth muscle rises with a lower rate because Ca²⁺ channels open more slowly than Na⁺ channels.
Pacemaker Potentials and Slow Waves
Info
Many smooth muscle cells are capable of initiating spontaneous electrical activity, called pacemaker potential. Currents result from spontaneous increases in inward, or depolarizing currents (e.g., voltage-gated Ca²⁺ currents) or spontaneous decreases in outward, or hyperpolarizing currents (e.g., voltage-gated K⁺ currents). These cause depolarization until Vm reaches threshold, triggering an action potential.
In other smooth muscle cells, spontaneous electrical activity results in regular, repetitive oscillations in Vm at a frequency of several oscillations per minute — referred to as slow waves.
Multiunit Smooth Muscle
Info
Multiunit smooth muscle is capable of finer motor control (electrical isolation of cells). Found in the iris and ciliary body of the eye, the piloerector muscles of the skin, and some blood vessels.
Action potentials usually do not occur in multiunit smooth muscle. Excitatory neurotransmitters cause a local depolarization, the junctional potential, which is similar to the end-plate potential in skeletal muscle. Junctional potentials spread electrotonically throughout the muscle fiber, altering Vm and triggering the entry of Ca²⁺ through voltage-gated L Ca²⁺ channels.
Thus, whereas action potential generation is essential for initiating contraction of skeletal and cardiac muscle, many smooth muscle cells contract despite being unable to generate an action potential.
For example, changes in Vm may modulate (by an unknown mechanism) the activity of the enzyme phospholipase C, which cleaves membrane phosphoinositides to release the intracellular second messengers diacylglycerol (DAG) and IP3 (pharmaco-mechanical coupling).
What is pharmaco-mechanical coupling in smooth muscle?
Changes in Vm modulate the activity of phospholipase C, which cleaves membrane phosphoinositides to release the second messengers diacylglycerol (DAG) and IP3, allowing smooth muscle contraction without action potential generation.
TLDR
Key Concepts: Membrane and Action Potentials
- Resting membrane potential varies by cell type: neurons −70 mV, skeletal muscle −95 mV, smooth muscle −50 mV, erythrocytes −12 mV.
- The Chemical Potential for uncharged solute X: . The Electric Potential: . At equilibrium, .
- The Nernst equation calculates equilibrium potential for a single ion: . Simplified at 37°C: .
- Resting Vm is primarily due to the asymmetric ion distribution (K⁺ leak channels), with minor contributions from the Na⁺/K⁺ pump (~4–5 mV) and negligible Donnan effect.
- The GHK equation accounts for multiple permeant ions; at rest ; at AP peak .
- Equivalent circuit model: membrane = capacitor (1 μF/cm²) + resistors (ion channels); ; ; only ~0.002% of intracellular K⁺ must move to establish Vm.
- Electrotonic potentials are passive, decremental; graded. Action potentials are active, all-or-none, non-decremental.
- Na⁺/K⁺ pump extrudes 3 Na⁺/2 K⁺; electrogenic; P-type ATPase; inhibited by ouabain/digitalis; intense activity → prolonged hyperpolarizing after-potential.
- Action potential phases: depolarization (Na⁺ channel opening via Hodgkin cycle), repolarization (Na⁺ inactivation + K⁺ channel opening), hyperpolarization (K⁺ channels remain open), return to rest.
- Absolute refractory period: Na⁺ channels in inactivated state (open→inactive→closed cycle).
- Length constant : potential decays to 37% at distance λ. Larger axons have lower → larger λ → faster conduction.
- Myelination ↓ and ↑ → faster conduction; saltatory conduction at nodes of Ranvier. Unmyelinated λ = 0.5 mm; myelinated = 1–3 mm.
- Nav channels: 4 domains × 6 TM segments; S4 = voltage sensor; S5/S6 = pore. TTX-S (nanomolar binding) vs. TTX-R (micromolar; cardiac). Nav1.4 → skeletal muscle myotonia; Nav1.5 → cardiac arrhythmias.
- TTX blocks pore opening of Nav, causing paralysis. BTX locks Nav persistently open, causing permanent depolarization.
- Cav channels: same structural homology as Nav; L-type (Cav1.x) for E-C coupling; N/P/Q-type (Cav2.x) for neurotransmitter release; T-type (Cav3.x) for pacemaking.
- Kv channels: tetrameric; delayed rectifier (Hodgkin-Huxley), A-type (fast inactivation), M-type (closed by ACh), Ca²⁺-activated (modulated by [Ca²⁺]i).
- CLC channels: dimer structure; not selective for Cl⁻; critical for skeletal muscle resting Vm; CFTR and GABA-A/glycine receptors also Cl⁻ channels.
- Cardiac AP: INa = depolarization (atria/ventricle/Purkinje); ICa = depolarization (SA/AV node) + plateau (all); IK = repolarization; If = pacemaker (HCN channels, activated by hyperpolarization and cAMP).
- CICR: Each L-type Ca²⁺ channel triggers one SR Ca²⁺ release channel; amplifies Ca²⁺ signal for contraction. NCX and Ca²⁺ pump restore Ca²⁺.
- HCN channels: 4 α-subunits × 6 TM; modulated directly by cAMP (not phosphorylation); reversal potential ~−20 mV; activated by hyperpolarization.
- Unitary smooth muscle: Ca²⁺-dependent AP (slower than skeletal); can have spikes, plateaus, or slow waves; pacemaker potentials from spontaneous changes in Ca²⁺ or K⁺ currents.
- Multiunit smooth muscle: no AP; junctional potentials spread electrotonically; Ca²⁺ entry via L-type channels; pharmaco-mechanical coupling via phospholipase C → DAG + IP3.