Graphical Abstract
TARGET DECK: MED::I::Signaling Pathways in Health and Disease::Cell Signaling::03 - Classification of Hormones
We won’t describe all types.
In order to be a hormone, a molecule should be able to travel a long distance.
We won’t be making a difference here between the ones that travel long vs short distances (signals).
1. Chemical Signals
1.1 Classification by Type
| Type | Examples |
|---|---|
| Neurotransmitters | Acetylcholine, dopamine |
| Hormones | Insulin, cortisol |
| «Local hormones» (autacoids) | Cytokines |
| Growth factors | EGF, IGF |
1.2 Classification by Structure
- Amino acid derivatives
- Polypeptides
- Eicosanoids
- Steroids
Other important info
The hormone target, provenience and molecular mechanism of action.
2. Hormones – General Definition
Definition
Hormones are proteins and other small molecules produced at very low dosage by a gland tissue (endocrine tissue) and carried to target tissues through the bloodstream.
Physiological concentration: – M
3. Hydrophilic vs. Lipophilic Hormones
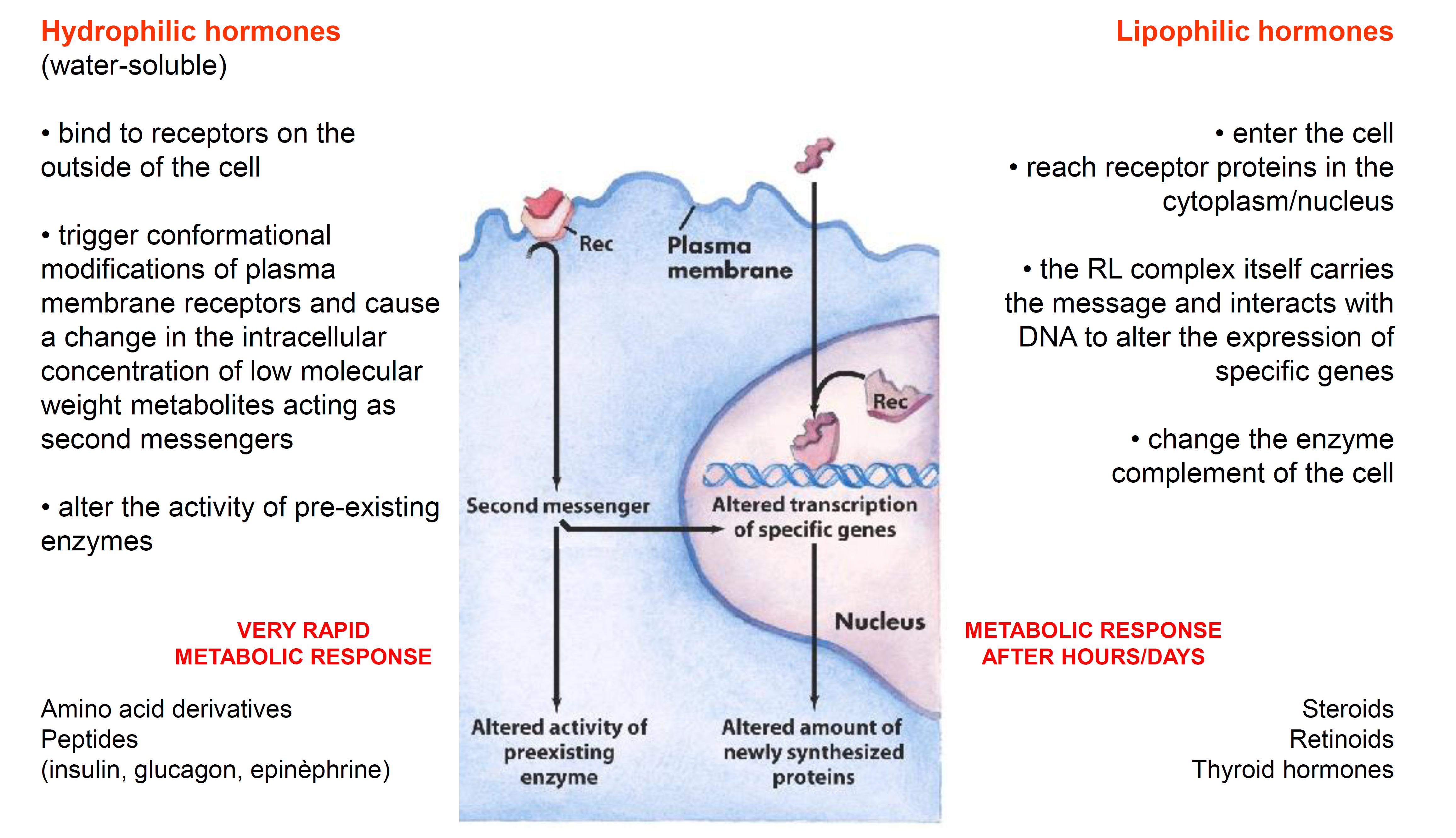
Hydrophilic Hormones
(Water-soluble)
-
Mechanism of Action:
- Bind to receptors located on the outside of the cell (plasma membrane).
- Trigger conformational modifications of plasma membrane receptors.
- Cause a change in the intracellular concentration of low molecular weight metabolites acting as second messengers.
- Alter the activity of pre-existing enzymes.
-
Speed: Result in a VERY RAPID METABOLIC RESPONSE.
-
Examples:
- Amino acid derivatives
- Peptides (e.g., insulin, glucagon, epinephrine)
Lipophilic Hormones
(Lipid-soluble)
- Mechanism of Action:
- Enter the cell directly through the plasma membrane.
- Reach receptor proteins (Rec) in the cytoplasm or nucleus.
- The RL (Receptor-Ligand) complex itself carries the message and interacts with DNA to alter the expression of specific genes.
- Change the enzyme complement of the cell by altering the amount of newly synthesized proteins.
- Speed: Result in a METABOLIC RESPONSE AFTER HOURS/DAYS, since it goes through the mechanism of expressions of genes → maturation of those mRnas, then polypeptides…
- Examples:
- Steroids
- Retinoids
- Thyroid hormones
| Feature | Hydrophilic Hormones | Lipophilic Hormones |
|---|---|---|
| Receptor Location | Plasma membrane (Surface) | Cytoplasm or Nucleus (Internal) |
| Primary Action | Activates second messengers | Alters gene transcription |
| Effect on Enzymes | Modifies existing enzymes | Creates new enzymes/proteins |
| Response Time | Very Rapid | Slow (Hours to Days) |
What is the mechanism of action of hydrophilic hormones?
They bind to receptors on the outside of the cell, trigger conformational changes in plasma membrane receptors, increase intracellular second messengers, and alter the activity of pre-existing enzymes → very rapid metabolic response.
What is the mechanism of action of lipophilic hormones?
They enter the cell, bind to cytoplasmic/nuclear receptors; the receptor–ligand complex interacts with DNA to alter gene expression, changing the enzyme complement of the cell → metabolic response after hours/days.
Anki cloze
Hydrophilic hormones act via {1:second messengers} and produce a {2:very rapid} metabolic response, while lipophilic hormones act via {3:nuclear receptor–ligand complexes} and produce a response after {4:hours/days}.
4. Seven Classes of Hormones
The 7 Hormone Classes
- Peptide
- Catecholamine
- Eicosanoid
- Steroid
- Retinoid
- Thyroid
- Nitric oxide
Mnemonic – " Please Come Eat Some Really Tasty Nachos"
Peptide · Catecholamine · Eicosanoid · Steroid · Retinoid · Thyroid · Nitric oxide
5. Peptide Hormones
Peptide Hormones – Structure and Synthesis
- Range from 3 to more than 200 amino acid residues
- Synthesized as precursor proteins (pro-hormones)
- Packed into secretory vesicles
- Proteolytically cleaved to form active peptides
- Carboxyl- and amino-terminal residues are modified to remove charges
- Released via high density secretory granule exocytosis
Examples of Peptide Hormones
- Pancreatic hormones: insulin, glucagon, somatostatin
- Parathyroid hormone (calcitonin)
- Hypothalamus and pituitary hormones
5.1 Hypothalamic Peptide Hormones
| Hormone | Function |
|---|---|
| Anti-Diuretic Hormone (ADH) | Regulates water levels in the body; affects blood pressure and volume |
| Corticotropin-Releasing Hormone (CRH) | Drives the body’s response to physical and emotional stress; stimulates anxiety; suppresses appetite |
| Gonadotropin-Releasing Hormone (GnRH) | Stimulates release of hormones acting on testes and ovaries to initiate and maintain reproductive function; levels increase in puberty to trigger sexual maturation |
| Growth Hormone-Releasing Hormone (GHRH) | Controls normal physical development in children, metabolism in adults; increased by sleep, stress, exercise, and low blood glucose |
| Oxytocin | Controls aspects of human behavior (sexual arousal, recognition, trust, anxiety, mother-infant bonding) and key aspects of the reproductive system (childbirth and lactation in women, ejaculation and conversion of testosterone into dihydrotestosterone in men) |
| Somatostatin | In the central nervous system, inhibits other hormones, most notably growth and thyroid-stimulating hormones |
| Thyrotropin-Releasing Hormone (TRH) | Stimulates production of thyroid hormone, which plays an important role in metabolism, heart and digestive functions, muscle control, brain development, and preservation of bones |
5.2 Hypothalamus–Pituitary Axis
Anatomy and Connectivity
- The hypothalamus is located on the undersurface of the brain, attached by a stalk to the pituitary gland.
- One set of nerve cells in the hypothalamus produces stimulating and inhibiting hormones that reach the anterior lobe (adenohypophysis) via a network of blood vessels running down through the pituitary stalk. → [HRHs that stimulate secretion of adenohypophyseal hormones]
- The other set sends the hormones they produce down through the pituitary stalk to the posterior lobe (neurohypophysis), where these hormones are stored/released as needed into the bloodstream. → [ADH; oxytocin]
Which hypothalamic hormones are released from the neurohypophysis?
ADH (antidiuretic hormone) and oxytocin are produced by hypothalamic neurons and stored/released from the posterior pituitary (neurohypophysis).
Anki cloze
The anterior pituitary (adenohypophysis) receives hypothalamic releasing hormones via {1:a portal blood vessel network in the pituitary stalk}, while ADH and oxytocin are stored and released from the {2:posterior pituitary (neurohypophysis)}.
6. Catecholamine Hormones
Catecholamines – Definition and Properties
- Aromatic amines named after catechol (1,2-dihydroxybenzene)
- In the nervous system, they function as neurotransmitters
- Also secreted by the adrenal gland (exocytosis) → act as hormones via metabotropic receptors
- Examples: epinephrine (adrenaline), norepinephrine (noradrenaline)
6.1 Biosynthesis of Catecholamines
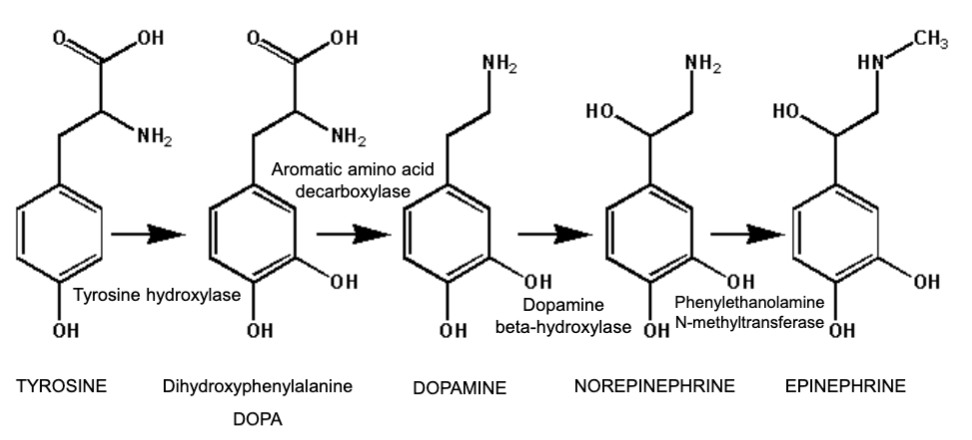
Catecholamine Synthesis Pathway
Starting from Tyrosine:
SMILES structures:
Tyrosine:
N[C@@H](Cc1ccc(O)cc1)C(=O)ODOPA (Dihydroxyphenylalanine):
N[C@@H](Cc1ccc(O)c(O)c1)C(=O)ODopamine:
NCCc1ccc(O)c(O)c1Norepinephrine:
NCC(O)c1ccc(O)c(O)c1Epinephrine:
CNC C(O)c1ccc(O)c(O)c1Mnemonic – Catecholamine Synthesis
“The Dark Night Makes Everybody Nervous”
Tyrosine → DOPA → Dopamine → Norepinephrine → Epinephrine (Enzymes: Tyrosine hydroxylase → AADC → Dopamine β-hydroxylase → PNMT)
What are the four enzymes in catecholamine biosynthesis from tyrosine?
- Tyrosine hydroxylase (Tyrosine → DOPA)
- Aromatic amino acid decarboxylase (DOPA → Dopamine)
- Dopamine β-hydroxylase (Dopamine → Norepinephrine)
- Phenylethanolamine N-methyltransferase (Norepinephrine → Epinephrine)
Anki cloze
Catecholamines are aromatic amines named after {1:catechol (1,2-dihydroxybenzene)} and include {2:epinephrine}, {3:norepinephrine}, and dopamine. They are secreted by the {4:adrenal gland} via exocytosis.
7. Eicosanoid Hormones
Eicosanoids – Definition and Properties
- Derived from arachidonate (20:4)
- Not stored
- Paracrine hormones (secreted into the interstitial fluid and acting nearby)
- Widely produced/distributed in mammalian tissues
- Examples: prostaglandins, thromboxanes, leukotrienes
7.1 Arachidonic Acid Release
Rate-Limiting Step Since the concentration of free arachidonic acid in the cells is very low, the biosynthesis of the eicosanoids depends mainly on the availability of phospholipase A2 (hydrolases) capable of releasing arachidonate from the middle carbon of glycerol of the phospholipids.
Arachidonic acid (SMILES):
CCCCCC=CCC=CCC=CCC=CCCCCC(=O)OPhospholipase cleavage sites on membrane phospholipids:
| Enzyme | Cleavage Site |
|---|---|
| Phospholipase A1 | sn-1 fatty acid ester bond |
| Phospholipase A2 | sn-2 fatty acid ester bond (releases arachidonate) |
| Phospholipase C | Head group–glycerol bond (glycerol side) |
| Phospholipase D | Head group–glycerol bond (head group side) |
7.2 Biogenesis of Eicosanoids
MEMBRANE PHOSPHOLIPIDS
↓ Phospholipase A2
POLYUNSATURATED FATTY ACIDS (C-20)
├─ Cyclooxygenase → PROSTAGLANDINS + THROMBOXANES
└─ Lipoxygenase → LEUKOTRIENES
7.3 Nomenclature and Abbreviations
| Class | Abbreviation |
|---|---|
| Prostaglandins | PG |
| Thromboxanes | TX |
| Leukotrienes | LT |
Nomenclature Breakdown: PGF2α
- PG = prostaglandin
- F = type of ring substitution in C9 and C11 (e.g., –OH; =O)
- 2 = degree of unsaturation (number of double bonds)
- α = cis isomerism of substituents in C9 and C11 (β = trans)
7.4 Key Eicosanoids and Their Functions
PGF2α (Prostaglandin F2α / Dinoprost)
- Potent vasoconstrictor produced by the uterus when stimulated by oxytocin
- Stimulates contraction of uterine and bronchial smooth muscle
- Used pharmaceutically (as Dinoprost) to induce labour and to terminate pregnancy, including missed or partial abortion
TXA2 (Thromboxane A2)
- Potent vasoconstrictor produced by endothelial cells, macrophages, and platelets upon platelet activation
- Main role: amplification of platelet activation and recruitment of additional platelets to the site of injury
Leukotrienes (LTs)
- Important inflammatory lipid mediators derived from arachidonic acid
- Oxidized by 5-lipoxygenase (5-LO) on the nuclear envelope
- Important role in airway inflammation:
- Mucus hypersecretion
- Increased vascular permeability
- Mucosal edema
- Potent bronchoconstriction
- Named due to their source from leukocytes and the presence of three conjugated double bonds in their structure
Why does eicosanoid biosynthesis depend on phospholipase A2?
Because the concentration of free arachidonic acid in cells is very low; phospholipase A2 releases arachidonate from the middle carbon (sn-2 position) of glycerol in membrane phospholipids, making it available for eicosanoid synthesis.
Anki cloze
Eicosanoids are derived from {1:arachidonate (20:4)} and act as {2:paracrine} hormones. Cyclooxygenase produces {3:prostaglandins and thromboxanes}, while lipoxygenase produces {4:leukotrienes}.
What is the pharmaceutical name of PGF2α and what is it used for?
Dinoprost. It is used to induce labour and to terminate pregnancy, including missed or partial abortion.
8. Steroid Hormones
Steroids – Structure and Properties
- Structures derived from cyclopentanoperhydrophenanthrene (steranes) — 4 rings, C-17+
- Hexagonal cycles adopt the chair configuration
- Not storable
- Since liposoluble, they require protein carriers to circulate in plasma:
- Albumin (low affinity)
- α-globulin (provides protection against inactivating metabolism while travelling to the target organ)
8.1 Steroid Biosynthesis Pathway
8.2 Classes and Functions
| Steroid | Function |
|---|---|
| Cortisol (glucocorticoid) | Affects protein and carbohydrate metabolism; suppresses immune response, inflammation, and allergic responses |
| Aldosterone (mineralocorticoid) | Regulates reabsorption of , , in the kidney |
| Testosterone / Estradiol (sex hormones) | Influence secondary sexual characteristics; regulate the female reproductive cycle |
Anki cloze
Steroid hormones are derived from {1:cyclopentanoperhydrophenanthrene (steranes)} and require {2:albumin or α-globulin} as plasma carriers because they are {3:liposoluble}.
Why do steroid hormones require plasma carrier proteins?
Because they are liposoluble (not water-soluble), they need protein carriers (albumin – low affinity; α-globulin) to circulate in plasma and to protect against inactivating metabolism during transport to the target organ.
9. Retinoid Hormones
Retinoids – Definition
- Structure derived from Vitamin A
- Many tissues convert retinol into retinoic acid
- Act via nuclear receptors
9.1 Retinoid Metabolism and Transport
Intestine:
- Ingested β-carotene is cleaved by β-carotene dioxygenase to yield retinal (all-trans)
- Retinal is reduced to retinol by retinaldehyde reductase (NADPH-requiring enzyme)
- Retinol (Vitamin A alcohol) is esterified to palmitic acid and delivered to the blood via chylomicrons
Liver:
- Uptake of chylomicron remnants → delivery of retinol for storage as a lipid ester (also in adipose tissue)
- Transport of retinol from the liver occurs by binding of hydrolyzed retinol to aporetinol binding protein (RBP)
- The retinol–RBP complex is transported to the cell surface within the Golgi and secreted
- Plasma transport of retinoic acid is accomplished by binding to albumin
Extrahepatic tissues:
- Retinol is bound to cellular retinol binding protein (CRBP)
- Retinol is converted to all-trans-retinoic acid
- Following binding, the receptor–vitamin complex interacts with specific sequences in several genes involved in growth and differentiation and affects expression of these genes
Retinol and retinoic acid are considered hormones in this capacity.
9.2 Retinoid Signaling in Embryonic Cells
(Source: https://doi.org/10.1038/nrg2340)
Retinoid Signaling Pathway (Embryonic)
- Retinoid sources: circulating maternal retinol (placental species); carotenoid stores in yolk (oviparous species)
- Retinol is taken up by embryonic retinol binding protein 4 (RBP4)
- Transferred intracellularly by the receptor STRA6
- Transformed into retinaldehyde, mainly by retinol dehydrogenase 10 (RDH10)
- Cleavage of β-carotene by β-carotene oxygenase (BCO) enzyme(s) directly generates retinaldehyde
- Retinaldehyde dehydrogenases (RALDH1 to RALDH3) then generate retinoic acid (RA)
- RA acts within the nucleus as a ligand for nuclear receptors [heterodimers of RA receptors (RARs) and retinoid X receptors (RXRs)] to regulate transcriptional activity of target genes
- All-trans-RA is the major in vivo RAR ligand
- CYP26-mediated metabolism degrades RA (hydroxylation → conjugation → elimination)
What nuclear receptor heterodimer mediates retinoic acid (RA) signaling?
Heterodimers of RA receptors (RARs) and retinoid X receptors (RXRs), which bind retinoic acid and regulate transcriptional activity of target genes. All-trans-RA is the major in vivo RAR ligand.
Anki cloze
In the intestine, ingested β-carotene is cleaved by {1:β-carotene dioxygenase} to yield {2:retinal (all-trans)}, which is then reduced to {3:retinol} by {4:retinaldehyde reductase} (requiring NADPH).
10. Thyroid Hormones
Thyroid Hormones – Synthesis and Structure
- Act via nuclear receptors
- Thyroglobulin: a protein precursor with numerous enzymatically condensated residues of iodinated-Tyr, accumulates in thyroid follicles (colloid)
- Release of both thyroid hormones as a result of proteolytic processes (stimulated by TSH)
| Hormone | Name | Iodo-substitution |
|---|---|---|
| T3 | 3,5,3′-Triiodothyronine | R = H |
| T4 | Thyroxine | R = I |
How are thyroid hormones synthesized and released?
Thyroglobulin (a precursor protein with iodinated tyrosine residues) accumulates in thyroid follicles (colloid). Both T3 and T4 are released by proteolytic processes stimulated by TSH.
Anki cloze
Thyroid hormones T3 and T4 are derived from {1:iodinated tyrosine residues} in {2:thyroglobulin}, stored in {3:thyroid follicles (colloid)}, and released by {4:proteolytic processes stimulated by TSH}.
11. Nitric Oxide (NO)
Nitric Oxide – Properties
- Free radical (gas), poor stability
- Acts near its synthesis/release site
- Freely diffusible through membranes
- Also known as ‘endothelium-derived relaxing factor’ (EDRF)
11.1 Biosynthesis of Nitric Oxide
Enzyme: Nitric oxide synthase (NOS) — related to cytochrome P450 reductase
SMILES:
Arginine:
N[C@@H](CCCNC(=N)N)C(=O)OHydroxyarginine:
N[C@@H](CCCNC(=N)NO)C(=O)OCitrulline:
N[C@@H](CCCNC(=O)N)C(=O)O11.2 NOS Isoforms
| Isoform | Gene | Location | Notes |
|---|---|---|---|
| nNOS (NOS-1) | NOS1 | Neurons | Constitutive expression |
| iNOS (NOS-2) | NOS2 | Macrophages | Inducible – expression awaits an appropriate stimulus (e.g., ingestion of a parasite); levels of nNOS and eNOS are relatively steady |
| eNOS (NOS-3) | NOS3 | Endothelial cells lining the lumen of blood vessels | Constitutive expression |
iNOS vs. Constitutive NOS Whereas the levels of nNOS and eNOS are relatively steady, expression of iNOS genes awaits an appropriate stimulus (e.g., ingestion of a parasite).
What are the three NOS isoforms, where are they found, and which is inducible?
- nNOS (NOS-1): neurons (constitutive)
- iNOS (NOS-2): macrophages (inducible – requires a stimulus, e.g., pathogen ingestion)
- eNOS (NOS-3): endothelial cells (constitutive)
Anki cloze
Nitric oxide is synthesized from {1:arginine} by {2:nitric oxide synthase (NOS)}, also consuming {3:NADPH} and {4:O₂}. The reaction produces {5:citrulline} as a co-product.
Anki cloze
The three NOS isoforms are {1:nNOS (NOS-1)} in neurons, {2:iNOS (NOS-2)} in macrophages (inducible), and {3:eNOS (NOS-3)} in endothelial cells.
TLDR
Lecture A.03 – Complete Summary
Chemical Signals:
- Classified by type (neurotransmitters, hormones, local hormones/autacoids, growth factors) and by structure (amino acid derivatives, polypeptides, eicosanoids, steroids)
Hormones – General:
- Produced at very low concentrations (– M) by endocrine tissue; transported via bloodstream
- Hydrophilic → membrane receptors → second messengers → rapid enzyme activity changes
- Lipophilic → nuclear/cytoplasmic receptors → RL complex interacts with DNA → slow gene expression changes (hours/days)
7 Hormone Classes (P-C-E-S-R-T-N):
Peptide – 3 to 200+ aa; synthesized as pro-hormones; proteolytically cleaved; released by exocytosis; examples include all hypothalamic-pituitary hormones (ADH, oxytocin, CRH, GnRH, GHRH, somatostatin, TRH)
- Hypothalamus → adenohypophysis via portal blood; → neurohypophysis (ADH, oxytocin) via axons
Catecholamine – derived from tyrosine; named after catechol; neurotransmitters AND adrenal hormones (exocytosis); metabotropic receptors; pathway: Tyrosine → DOPA → Dopamine → Norepinephrine → Epinephrine (enzymes: tyrosine hydroxylase, AADC, dopamine β-hydroxylase, PNMT)
Eicosanoid – derived from arachidonate (20:4); not stored; paracrine; biosynthesis requires phospholipase A2 (releases arachidonate from sn-2 of membrane phospholipids); cyclooxygenase → prostaglandins + thromboxanes; lipoxygenase → leukotrienes
- PGF2α (Dinoprost): vasoconstrictor, induces labour/abortion
- TXA2: vasoconstrictor, platelet activation amplifier
- LTs: airway inflammation, bronchoconstriction (from leukocytes, via 5-LO)
Steroid – derived from cyclopentanoperhydrophenanthrene; cholesterol precursor; not storable; lipophilic → carrier proteins (albumin, α-globulin); glucocorticoids (cortisol), mineralocorticoids (aldosterone), sex hormones (testosterone, estradiol)
Retinoid – Vitamin A derivatives; retinol → retinoic acid; nuclear receptors (RAR/RXR heterodimers); all-trans-RA is major RAR ligand; metabolism: β-carotene → retinal → retinol (intestine); retinol → retinoic acid (extrahepatic tissues); CYP26 degrades RA; transported by RBP (retinol) and albumin (retinoic acid)
Thyroid (T3/T4) – iodinated tyrosine derivatives; thyroglobulin precursor in follicular colloid; released by TSH-stimulated proteolysis; T3 = triiodothyronine (R=H); T4 = thyroxine (R=I); nuclear receptors
Nitric Oxide – free radical gas; EDRF; freely membrane-diffusible; short-lived; synthesized from arginine + NADPH + O₂ via NOS → citrulline + NO•; 3 isoforms: nNOS (neurons, constitutive), iNOS (macrophages, inducible), eNOS (endothelium, constitutive)