TARGET DECK: MED::I::Signaling Pathways in Health and Disease::Metabolic Biochemistry::06 - Glycolisis
Topics Covered
- The reactions of glycolysis
- Reoxidation of NADH from glycolysis
- Formation of lactic acid
- Shuttles
- Utilization as reducing power (biosynthesis)
- Energy balance
- Entry of other sugars into glycolysis
1. Overview: Where Glycolysis Fits
Glucose sits at a metabolic crossroads:
- → Pentose phosphate pathway → Ribose-5-phosphate (biosynthesis, reducing power)
- → Glycolysis → Pyruvate (energy production)
- ← Glycogen / starch / sucrose (storage forms that feed in)
2. Stoichiometry of Glycolysis
In anaerobiosis (net reaction):
Full mechanistic breakdown:
In aerobiosis (there’s ):
In standard condition, negative .
But in physiological conditions it’s still irreversible but the is not as negative.
The purpose of the reaction is to produce .
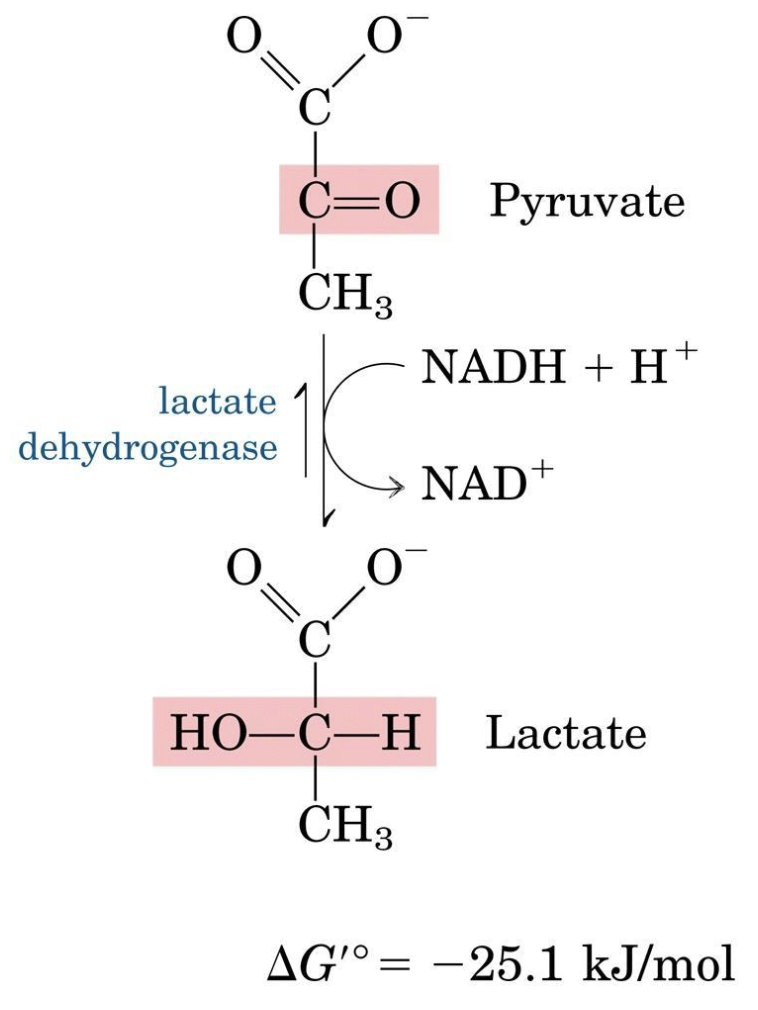
3. Pyruvate → Lactate (Lactate Dehydrogenase)
This reaction reoxidizes NADH, allowing glycolysis to continue under anaerobic conditions.
4. Net Reagents in Glycolysis
Historical note: The heat-labile fraction was called zymase, and the heat-stable cofactor fraction was called co-zymase (now known as NAD⁺).
Required cofactors: , ADP, NAD⁺, Mg²⁺
is reduced to .
This reduction process always produces a proton ().
→ Glycolysis produces an acidic environment.
5. Energy Balance
The reaction can be split into two:
- Conversion of glucose to pyruvate (oxidation of glucose) → exergonic (the loss by the glucose is used to reduce ):
- ATP synthesis (from and ) - endergonic:
Overall:
Irreversibility
The remains negative.
→ Under both standard and cellular conditions, glycolysis is essentially irreversible overall.
What is the net energy balance of glycolysis?
Glycolysis yields 2 ATP and 2 NADH per glucose, with an overall negative ΔG.
6. Historical Origin & Fermentation
Glycolysis was discovered by Pasteur as alcoholic fermentation in Saccharomyces cerevisiae.
In yeast under anaerobiosis, pyruvate is first decarboxylated (to acetic aldehyde), then reduced:
| Molecule | Formula | SMILES |
|---|---|---|
| Pyruvate | 𝐶𝐻3𝐶𝑂𝐶𝑂𝑂− | $smiles=CC(=O)C(=O)[O-] |
| Acetaldehyde | 𝐶𝐻3𝐶𝐻𝑂 | $smiles=CC=O |
| Ethanol | 𝐶𝐻3𝐶𝐻2𝑂𝐻 | $smiles=CCO |
| The final molecule is pyruvate not ethanol. |
7. The Two Phases of Glycolysis
Preparatory phase (investment)
- Consumes 2 ATP
- Glucose (C₆) is phosphorylated and cleaved into two C₃ units
Payoff phase (recovery)
- Each C₃ unit yields: +1 NADH, +2 ATP
- Two C₃ units → +2 NADH, +4 ATP
Net Gain per Glucose
+2 ATP and +2 NADH
detto dalla lu...
we start form a 6C molecule
→ preparatory phase of glycolysis ( lose 2 ATP)
→ 6C molecule split into 2x 3C
→ payoff where both 3C molecules produce 1 NADH + 2 ATP⇒ net gain: 2 ATP and 2 NADH
8. The 10 Reactions of Glycolysis
Step 1 — Hexokinase / Glucokinase
The actual is even more negative because and :
Why phosphorylate?
Phosphorylation traps intermediates inside the cell (charged molecules cannot cross membranes) and enables the three irreversible control steps. Phosphorylation occurs on C-6 because C-1 bears a carbonyl group and cannot be phosphorylated.
Step 2 — Phosphohexose Isomerase
Moves the carbonyl from C-1 to C-2 — a prerequisite for the next phosphorylation and the aldolase cleavage.
Step 3 — Phosphofructokinase-1 (PFK-1)
The actual is much more negative. Now both C-1 and C-6 are phosphorylated, ensuring both products of cleavage are phosphorylated and interconvertible.
Key Control Point
PFK-1 is the primary regulated enzyme of glycolysis.
Why is PFK-1 considered the key control point of glycolysis?
It is the primary regulated enzyme and commits fructose-6-phosphate to the glycolytic pathway.
Step 4 — Aldolase
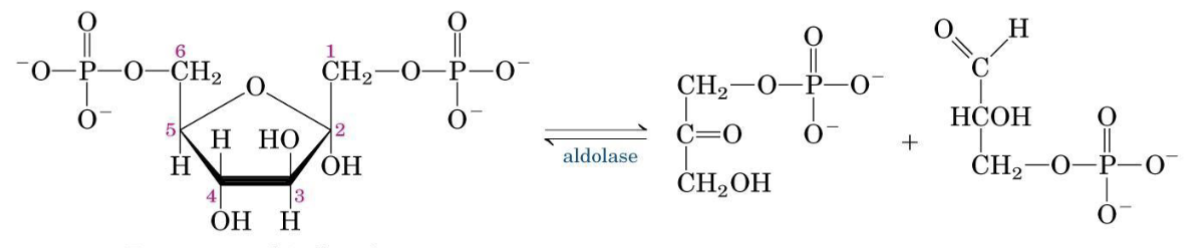
At physiological concentrations ([FBP] = 10 mM; [GAP] = [DHAP] = 1 mM):
Near-Equilibrium Reaction
This reaction is close to equilibrium and is reversible — it also operates in gluconeogenesis, depending on metabolite concentrations.
Step 5 — Triose Phosphate Isomerase
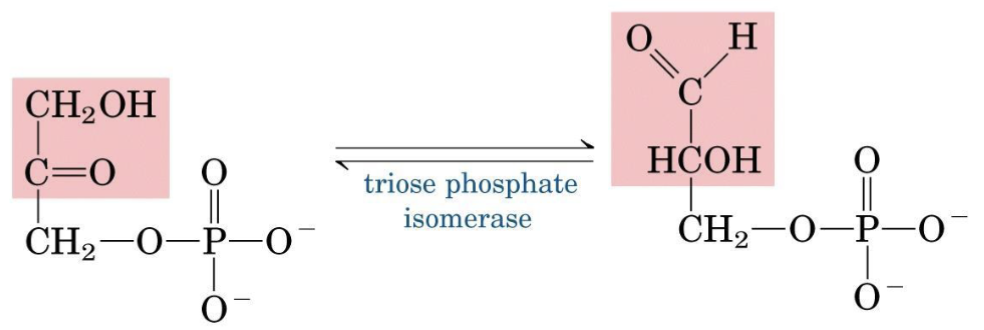
Converts the “dead-end” DHAP into GAP, funneling both products of step 4 into a single productive pathway.
Step 6 — Glyceraldehyde-3-Phosphate Dehydrogenase (GAPDH)
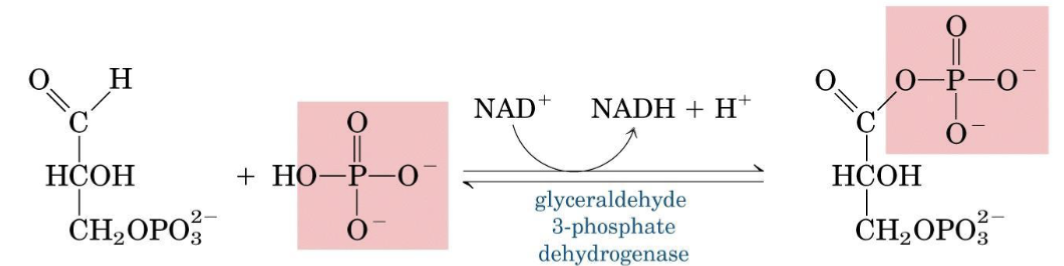
The strongly exergonic oxidation of an aldehyde to a carboxylate is coupled to the endergonic incorporation of phosphate → net reaction is small and reversible.
Mechanism:
- Active-site Cys (pKₐ ≈ 5.5 when NAD⁺ is bound) attacks GAP → thiohemiacetal
- NAD⁺ oxidizes the intermediate → thioester + NADH
- NADH leaves; fresh NAD⁺ enters
- Phosphorolysis by Pᵢ releases 1,3-BPG
Inhibitor
Iodoacetate irreversibly alkylates the active-site Cys residue → enzyme inactivation.
What does GAPDH produce, and what inhibits it?
GAPDH converts GAP to 1,3-BPG while producing NADH; it is inhibited by iodoacetate.
Step 7 — Phosphoglycerate Kinase
First ATP-generating step (substrate-level phosphorylation). Reaction is near equilibrium in vivo → also functions in gluconeogenesis.
Substrate-level phosphorylation mechanism:
Step 8 — Phosphoglycerate Mutase
Moves the phosphoryl group from C-3 to C-2, setting up dehydration in the next step.
Step 9 — Enolase
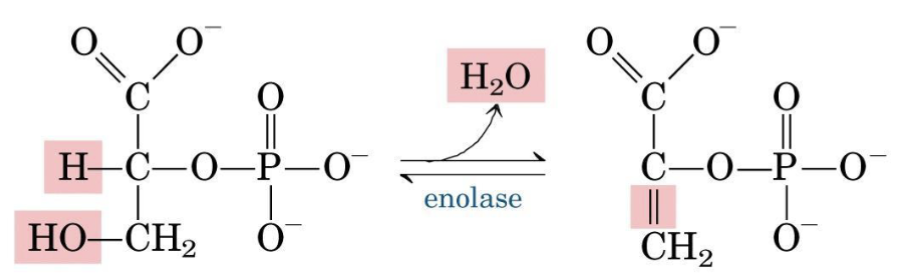
Dehydration creates a high-energy enol phosphate, activating the phosphoryl group for transfer to ADP.
Step 10 — Pyruvate Kinase
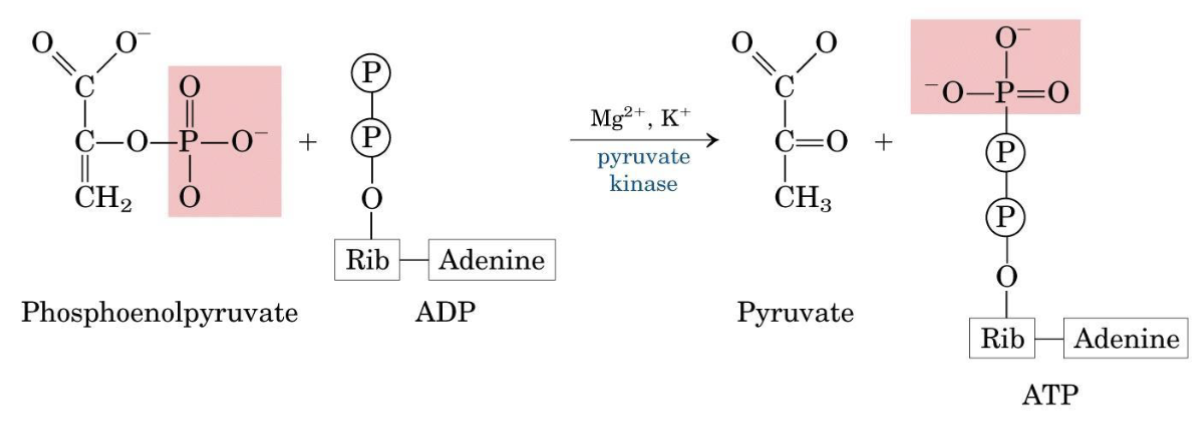
Second ATP-generating step. Although [ATP] > [ADP] makes , it remains sufficiently negative to make the reaction irreversible. PEP → pyruvate involves tautomerization of the enol form to the more stable keto form.
Irreversible Step
This is the third irreversible, regulated step of glycolysis.
What are the three irreversible, regulated steps of glycolysis?
Hexokinase, PFK-1, and pyruvate kinase.
9. Free-Energy Changes Summary (Erythrocytes)
| Step | Reaction | (kJ/mol) | in vivo (kJ/mol) |
|---|---|---|---|
| 1 | Glucose → G6P | −16.7 | −33.4 |
| 2 | G6P → F6P | +1.7 | −2.5 |
| 3 | F6P → F1,6BP | −14.2 | −22.0 |
| 4 | F1,6BP → DHAP + GAP | +23.8 | −1.2 |
| 5 | DHAP → GAP | +7.4 | +2.0 |
| 6 | GAP → 1,3-BPG | +6.3 | ≈0 |
| 7 | 1,3-BPG → 3-PG | −18.8 | −1.2 |
| 8 | 3-PG → 2-PG | +4.4 | +0.8 |
| 9 | 2-PG → PEP | +7.6 | +3.0 |
| 10 | PEP → Pyruvate | −31.4 | −16.7 |
Steps bypassed in gluconeogenesis are steps 1, 3, and 10 (the irreversible ones).
10. Fate of Pyruvate
| Condition | Pathway | Products |
|---|---|---|
| Aerobic (animals, plants, microbes) | TCA cycle + oxidative phosphorylation | 6 CO₂ + 6 H₂O |
| Anaerobic (muscle, RBCs, some microbes) | Lactate fermentation | Lactate |
| Anaerobic (yeast) | Alcoholic fermentation | Ethanol + CO₂ |
Pyruvate can also donate carbon skeletons for:
- Alanine synthesis
- Fatty acid synthesis (via acetyl-CoA)
What are the main fates of pyruvate?
Aerobic oxidation to acetyl-CoA and the TCA cycle, anaerobic lactate fermentation, alcoholic fermentation in yeast, or conversion to alanine and fatty acids.
11. Intracellular Organization
In vivo, glycolytic enzymes form multi-enzyme complexes rather than floating freely. This channeling improves efficiency by passing intermediates directly between active sites. When cells are broken and diluted, complexes dissociate and activity may be reduced.
A notable example: GAPDH and phosphoglycerate kinase physically associate, channeling 1,3-BPG between them.
12. Entry of Other Sugars
| Sugar | Entry Point | Notes |
|---|---|---|
| Galactose | Glucose-1-P → G6P (via UDP-galactose) | Via Leloir pathway |
| Fructose (liver) | Fructose-1-P → DHAP + glyceraldehyde | Via fructokinase (unregulated!) |
| Fructose (muscle) | F6P | Via hexokinase |
| Mannose | Mannose-6-P → F6P | Via phosphomannose isomerase |
| Trehalose | Glucose | Via trehalase |
Fructose in the Liver
Liver fructokinase is unregulated (unlike PFK-1). Excessive fructose → ↑ acetyl-CoA → ↑ fatty acid synthesis + ↑ glycerol-3-P → fatty liver, obesity, metabolic syndrome.
13. Regulation of Glycolysis
Three irreversible, allosterically regulated steps:
In muscle:
| Enzyme | Inhibitors | Activators |
|---|---|---|
| Hexokinase | Glucose-6-P | — |
| PFK-1 | ATP, citrate, fatty acids | AMP, ADP, Fructose-2,6-bisP |
| Pyruvate kinase | ATP, acetyl-CoA, fatty acids | Fructose-1,6-bisP |
In liver: Regulation is tightly coupled and symmetrical with gluconeogenesis — the same “irreversible” steps have dedicated bypass enzymes running in the opposite direction.
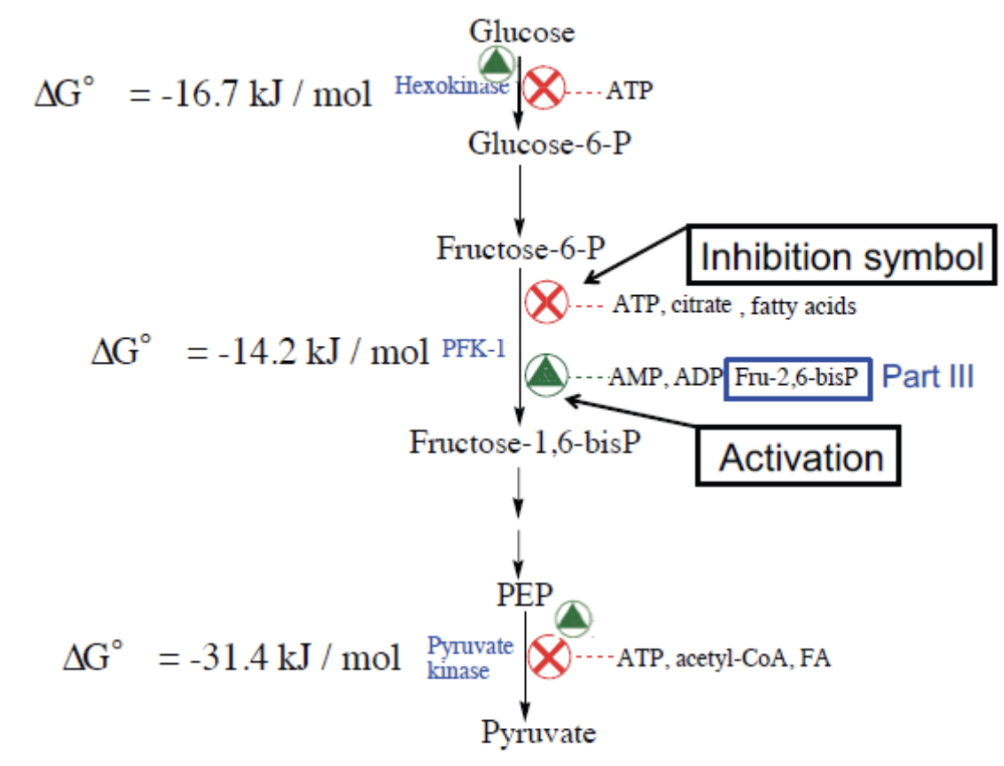
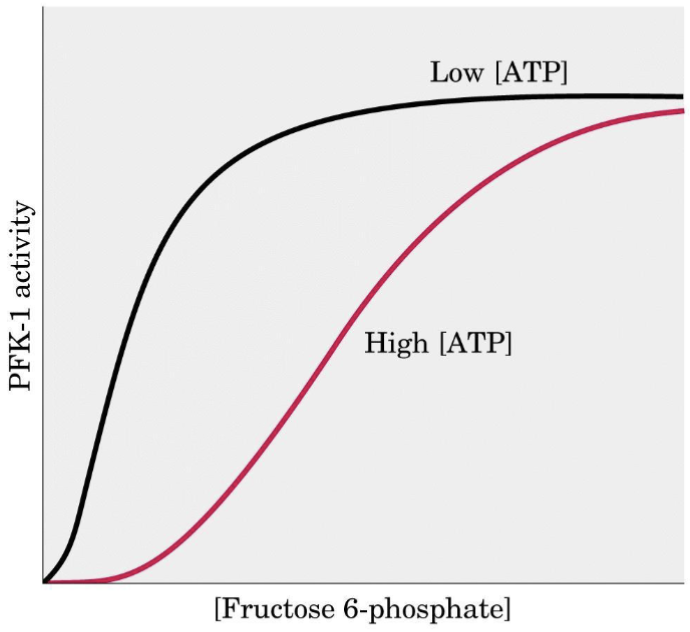
Why does PFK-1 slow with high [ATP]?
ATP binds to an allosteric regulatory site (distinct from the active site). This causes a conformational change that dramatically reduces catalytic rate → glycolysis slows when energy charge is high.
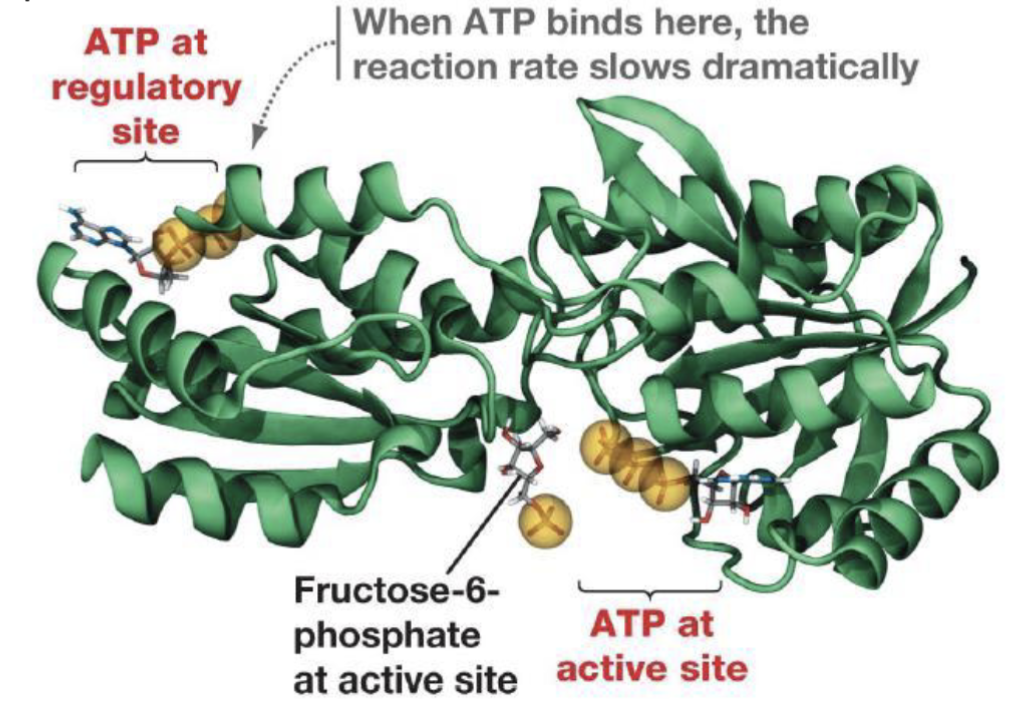
Logic of Regulation
The cell uses energy status ([ATP]/[AMP] ratio) as the signal: high ATP = stop breaking down glucose; low ATP = accelerate glycolysis.
Why must intermediates be phosphorylated?
- Retention: Phosphorylated intermediates cannot cross the plasma membrane → kept in the cell
- Control: Phosphorylation enables the three irreversible, allosteric control points
Without the initial phosphorylation, the pathway would be:
Glucose → Fructose → (Glyceraldehyde + Dioxyacetone) → 1-Phosphoglycerate → Glycerate → Pyruvate
— and all three irreversible control steps would be lost.
Why must glycolytic intermediates be phosphorylated?
Phosphorylation traps metabolites inside the cell and enables the three irreversible control points.
TLDR - 06 - Glycolysis
Detailed TLDR — Glycolysis
What it is: Glycolysis (Embden–Meyerhof–Parnas pathway) is the universal 10-step conversion of one glucose (C₆) into two pyruvate (C₃) molecules. It occurs in the cytosol, requires no oxygen, and is the first stage of carbohydrate catabolism.
Two phases:
- Preparatory phase (steps 1–5): Glucose is double-phosphorylated and cleaved into two triose-phosphates (GAP). Costs 2 ATP.
- Payoff phase (steps 6–10): Each GAP is oxidized to pyruvate with substrate-level phosphorylation. Yields 4 ATP + 2 NADH total (×2 because two C₃ units).
Net yield per glucose: , , . Overall → essentially irreversible.
Why phosphorylate intermediates? Phosphorylation (1) traps metabolites inside the cell and (2) enables the three irreversible control steps. Without it, all regulation is lost.
Three irreversible, regulated steps:
- Hexokinase (step 1) — inhibited by its own product G6P (product inhibition)
- PFK-1 (step 3) — the master regulator; inhibited by ATP, citrate, fatty acids; activated by AMP, ADP, F-2,6-bisP. PFK-1 has a separate allosteric ATP-binding site that causes a conformational shift and dramatic slowdown when energy charge is high.
- Pyruvate kinase (step 10) — inhibited by ATP, acetyl-CoA, fatty acids; activated by F-1,6-bisP (feedforward)
Fate of pyruvate:
- Aerobic: → Acetyl-CoA → TCA cycle → oxidative phosphorylation (full oxidation)
- Anaerobic (muscle/RBCs): → Lactate (via LDH; reoxidizes NADH to keep glycolysis running)
- Anaerobic (yeast): → Acetaldehyde + CO₂ → Ethanol
Key enzymes to know mechanistically:
- GAPDH (step 6): forms a thioester intermediate at active-site Cys (pKₐ ≈ 5.5); phosphorolysis releases 1,3-BPG. Inhibited by iodoacetate.
- Aldolase (step 4): near-equilibrium under physiological concentrations () → reversible, operates in both glycolysis and gluconeogenesis.
- Enolase (step 9): dehydration creates the high-energy PEP, priming the large release at pyruvate kinase.
In liver: Regulation is symmetrically linked with gluconeogenesis. The same irreversible steps have separate bypass enzymes so both pathways can be independently controlled.
Fructose caveat: Liver fructokinase bypasses PFK-1 regulation → uncontrolled flux → excess acetyl-CoA + glycerol-3-P → fat synthesis → fatty liver and metabolic syndrome.
Other sugars: Galactose (via UDP-galactose → G1P → G6P), mannose (→ F6P), fructose (→ F1P or F6P), and trehalose all feed into the pathway at various points.