TARGET DECK: MED::I::Morphology and Development::Histology::07 - Blood and Lymphoid System
Blood
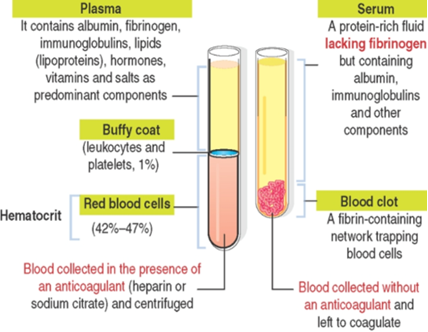
Blood, a specialized type of connective tissue, consists of plasma — the fluid component (the extracellular matrix) — and cells.
These components may be separated by centrifugation when blood is collected in the presence of anticoagulants.
Blood Composition by Centrifugation
- Sedimented RBCs: ~– of blood volume → this percentage of erythrocyte volume is the hematocrit
- Buffy coat layer (sitting on top of RBC layer): contains leukocytes and platelets
- Plasma (translucent supernatant fraction above packed RBCs)
Normal adult blood volume: 5 to 6 L
Plasma vs. Serum
Component Plasma Serum Definition Fluid component of blood Fibrinogen-free plasma Fibrinogen Present Absent Formation Blood + anticoagulant → centrifuge Blood without anticoagulant → clot → fluid portion Key proteins Albumin, fibrinogen, immunoglobulins, lipoproteins, hormones, vitamins, salts Albumin, immunoglobulins and other components
What is the hematocrit?
The percentage of erythrocyte volume relative to total blood volume; normally .
What is the difference between plasma and serum?
Plasma is the fluid component of blood containing fibrinogen; serum is essentially fibrinogen-free plasma, obtained when blood clots without anticoagulant.
Anki cloze
Blood is separated by centrifugation into: sedimented RBCs ({1:hematocrit} –), the {2:buffy coat} layer (leukocytes + platelets), and {3:plasma} (translucent supernatant).
Circulating Blood Cells
The total volume of blood of an average adult is about 5 L, circulating throughout the body within the confines of the circulatory system.
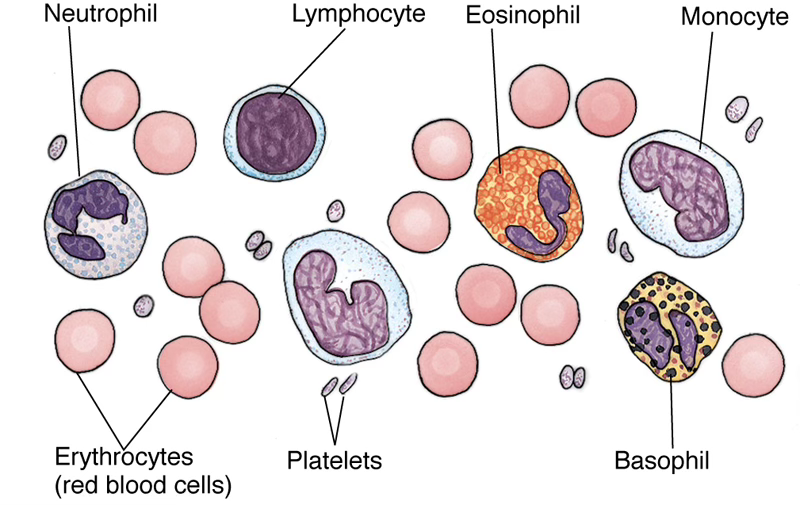
The formed elements are composed of cells and cell fragments, known as platelets.
Light microscopic examination of the formed elements is performed using either the Wright or Giemsa stains, and identification of blood cells is based on the colors produced by these stains.
The cells of blood are subdivided into two major components:
- Red blood cells (RBC)
- White blood cells (WBCs, leukocytes)
Red blood cells lose their nuclei and organelles during development; therefore mature, circulating RBCs are anucleated cells whose cytoplasm is filled with hemoglobin.
White blood cells are subdivided into:
- Agranulocytes — those without specific granules (lymphocytes and monocytes)
- Granulocytes — those housing granules (neutrophils, eosinophils, and basophils)
Platelets are round to oval cell fragments derived from megakaryocytes.
Anki cloze
Leukocytes are divided into {1:agranulocytes} (lymphocytes and monocytes, lacking specific granules) and {2:granulocytes} (neutrophils, eosinophils, basophils, containing specific granules).
Blood Cell Reference Values
| Cell Type | Count (per L or mm³) | Percentage |
|---|---|---|
| Erythrocytes | – | — |
| Leukocytes (total) | – | — |
| Neutrophils | – | |
| Eosinophils | – | |
| Basophils | ||
| Lymphocytes | ||
| Monocytes | ||
| Platelets | — | |
| Hematocrit | — | – |
Remember the percentages -viola
Anki cloze
Neutrophils: {1:60%–70%} | Eosinophils: {2:2%–4%} | Basophils: {3:0.5%} | Lymphocytes: {4:28%} | Monocytes: {5:5%} | Hematocrit: {6:42%–47%}
Anki basic
What stains are used to identify formed elements of blood by light microscopy? Wright stain and Giemsa stain.
Red Blood Cells (RBC; Erythrocytes)
RBCs, also called erythrocytes (Greek erythros, red; kytos, cell), are:
- Non-nucleated, biconcave-shaped cells
- Diameter: ~
- Lack organelles
- Consist only of a plasma membrane, its underlying cytoskeleton, hemoglobin and glycolytic enzymes
RBC Lifespan and Turnover
- – per mm³
- Circulate for 120 days
- Senescent RBCs are removed by phagocytosis or destroyed by hemolysis in the spleen
- Replaced by reticulocytes, which complete hemoglobin synthesis and maturation 1 to 2 days after entering circulation
- Reticulocytes account for to of circulating RBCs
- RBCs transport oxygen and carbon dioxide and are confined to the circulatory system
Hemoglobin
Red blood cells are packed with hemoglobin, a large tetrameric protein composed of four polypeptide chains, each of which is covalently bound to an iron-containing heme.
- Hemoglobin provides the unstained cell with its pale yellow color
- The globin moiety releases
- The iron binds to in regions of high oxygen concentration (e.g., the lung)
- In oxygen-poor regions (tissues), hemoglobin releases and binds
| Form | Ligand | Name |
|---|---|---|
| Hemoglobin + | Oxygen | Oxyhemoglobin |
| Hemoglobin + | Carbon dioxide | Carbaminohemoglobin |
What is the lifespan of a circulating RBC?
120 days. Senescent RBCs are removed by phagocytosis or hemolysis in the spleen.
Anki basic
What are reticulocytes and what percentage of circulating RBCs do they account for? Reticulocytes are immature RBCs that complete hemoglobin synthesis 1–2 days after entering circulation; they constitute – of circulating RBCs.
Anki cloze
Hemoglobin carrying oxygen is called {1:oxyhemoglobin}; hemoglobin carrying carbon dioxide is called {2:carbaminohemoglobin}.
Blood Groups
The extracellular surface of the RBC plasmalemma has specific inherited carbohydrate chains that act as antigens and determine the blood group of an individual.
- The most notable are the A and B antigens, determining the four primary blood groups: A, B, AB, and O
- The Rh group is named because it was first identified in rhesus monkeys
- Three Rh antigens (C, D, and E) are so common that erythrocytes of of Americans have one of these antigens → these individuals are Rh
Why is the Rh blood group named "Rh"?
Because it was first identified in rhesus monkeys.
Leukocytes
Leukocytes (– per mm³) are categorized as either granulocytes or agranulocytes.
- Granulocytes: contain primary and specific (secondary) cytoplasmic granules
- Agranulocytes: contain only primary granules
Granulocytes
Granulocytes are phagocytic cells with a multilobed nucleus, measuring 12 to 15 m in diameter. Three types distinguished by cytoplasmic granules:
- Neutrophils
- Eosinophils
- Basophils
Neutrophils
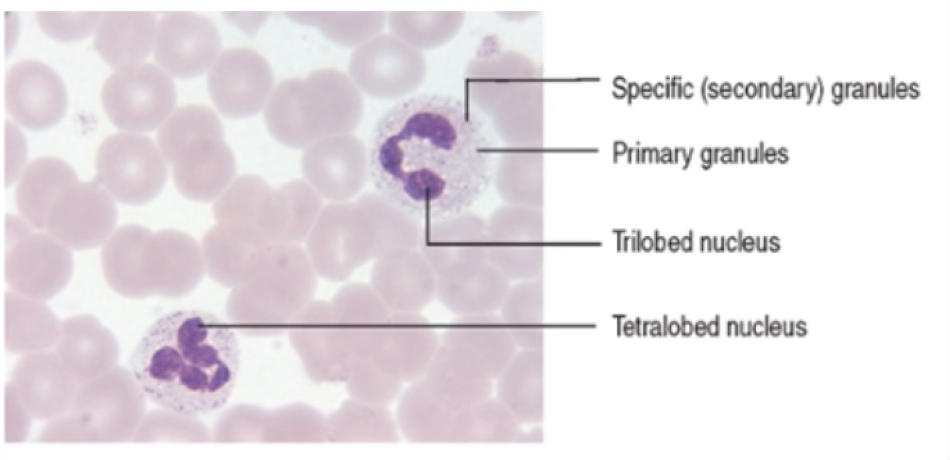
Neutrophil Key Facts
- Constitute – of circulating leukocytes
- Multilobed nucleus connected by slender chromatin threads
- Cytoplasm: secondary (specific) and primary granules
- Appear very pale pink in stained smears
- Life span: 6–7 hours in blood; up to 4 days in connective tissue
- Among the first cells to appear in acute bacterial infections
- Very small specific granules
Sex Chromatin in Neutrophils
In females, the nucleus presents a characteristic small appendage — the “drumstick” — which contains the condensed, inactive second X chromosome, but is not always evident in every cell.
Neutrophil Granule Contents
Granule Type Contents Primary granules Elastase, defensins, myeloperoxidase Secondary (specific) granules Lactoferrin, gelatinase, lysozyme, other proteases L-selectin and integrins in neutrophils bind to endothelial cell ligands ICAM-1 and ICAM-2 (intercellular-adhesion molecules 1 and 2). These ligands enable the antibacterial and anti-inflammatory function of neutrophils in the extravascular space.
After leaving the circulation, neutrophils act to eliminate bacteria or limit the extent of an inflammatory reaction in the connective tissue.
What is the "drumstick" in neutrophils?
A small nuclear appendage present in females, containing the condensed, inactive second X chromosome.
Anki cloze
Neutrophil primary granules contain {1:elastase, defensins, and myeloperoxidase}; secondary granules contain {2:lactoferrin, gelatinase, lysozyme, and other proteases}.
Anki cloze
Neutrophils constitute {1:–} of circulating leukocytes and have a life span of {2:6–7 hours} in the bloodstream and up to {3:4 days} in connective tissue.
Eosinophils
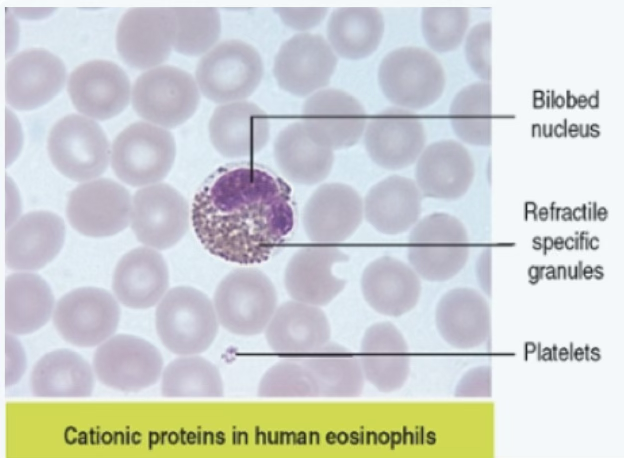
Eosinophil Key Facts
- Constitute – of circulating leukocytes (less than of total WBC population)
- Bilobed nucleus (sausage-shaped; two lobes connected by thin chromatin strand and nuclear envelope)
- Large, salmon pink colored specific granules (large, refractile, stain red in smears)
- Half-life: ~18 hours
- Leave circulation, recruited to connective tissue by IL-5
- First line of defense against parasites
- Phagocytose antigen-antibody complexes
- Participate in triggering bronchial asthma
Clinical Correlate — Eosinophils
Eosinophilic esophagitis, associated with eosinophilia, is clinically defined by dysphagia and abdominal pain. This condition is triggered by fungal and insect allergens.
What cytokine recruits eosinophils to connective tissue?
IL-5.
Anki cloze
Eosinophils have a {1:bilobed} nucleus and large {2:salmon pink} specific granules; they are the first line of defense against {3:parasites}.
Basophils
Basophil Key Facts
- Constitute less than of the total leukocyte population
- Round cells with an S-shaped nucleus, commonly masked by large dark blue to black specific granules
- Specific granules: large, stain dark blue or purple
- Life span: ~60 hours (contrast: mast cells survive for weeks and months)
- Express IgE receptors (FcεR1) on their plasmalemma
- Function very similar to mast cells
- Complete maturation in the bone marrow
Basophil vs. Mast Cell — Key Differences
Feature Basophil Mast Cell Maturation site Bone marrow (complete) Enter connective tissue/mucosae as immature cells lacking granules c-kit receptor Absent () Present () FcεR1 (IgE receptor) Present () Present () CD49b Present () Absent () Phenotype Lifespan ~60 hours Weeks to months
Basophils play a role in bronchial asthma and type 2 immunity in response to allergens (allergic skin reaction) and parasitic worms (helminths).
How do basophils and mast cells differ in terms of c-kit and CD49b expression?
Basophils are c-kit FcεR1 CD49b; mast cells are c-kit FcεR1 CD49b.
Anki cloze
Basophil-specific granules stain {1:dark blue or purple} and the nucleus is often {2:obscured} by them; basophils have a lifespan of about {3:60 hours}.
Agranulocytes
Lymphocytes
Lymphocyte Key Facts
- Constitute – of the total circulating leukocyte population
- Round cells, somewhat larger than RBCs
- Slightly indented, dense, round nucleus occupying most of the cell
- Cytoplasm: basophilic, often appearing as a thin rim around the nucleus; a few primary granules may be present
- May live for a few days or several years
Two size classes:
- Small lymphocytes (; –)
- Large lymphocytes (; –)
Two functional categories:
| Type | Origin & Maturation | Function |
|---|---|---|
| B lymphocytes (B cells) | Produced and mature in bone marrow | Antigen-stimulated B cells differentiate into antibody-secreting plasma cells |
| T lymphocytes (T cells) | Produced in bone marrow; complete maturation in the thymus | Activated T cells participate in cell-mediated immunity |
Where do B cells mature vs. T cells?
B cells: produced and mature in bone marrow.
T cells: produced in bone marrow, complete maturation in the thymus.
Anki cloze
Lymphocytes constitute {1:–} of total circulating leukocytes; their cytoplasm is {2:basophilic} and often appears as a thin {3:rim} around the nucleus.
Monocytes
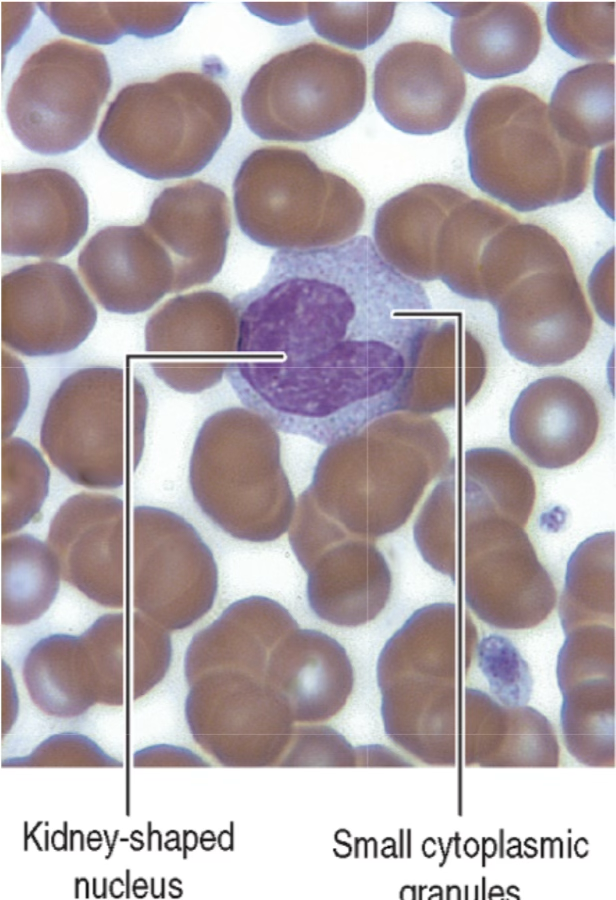
Monocyte Key Facts
- Largest leukocytes: –
- Constitute – of the leukocyte population
- Large, kidney-shaped nucleus whose lobe-like extensions seem to overlap one another
- Chromatin network: coarse but not overly dense
- Cytoplasm: bluish gray, with numerous azurophilic granules (lysosomes) and occasional vacuole-like spaces; no specific granules
- Travel briefly (12–100 hours) in bloodstream, then enter peripheral tissues
Monocyte Differentiation
- In connective tissue: differentiate into macrophages → involved in bacterial phagocytosis, antigen presentation, and clean-up of dead cell debris
- In bone: differentiate into osteoclasts under the control of osteoblasts
Macrophages are avid phagocytes and as members of the mononuclear phagocyte system, they:
- Phagocytose and destroy dead cells, antigens and foreign particulate matter (e.g., bacteria)
- Have a major role in the immune response
What is the largest circulating leukocyte, and what shape is its nucleus
Monocyte (–); kidney-shaped nucleus.
Anki cloze
Monocytes differentiate into {1:macrophages} in connective tissue and into {2:osteoclasts} in bone.
Platelets
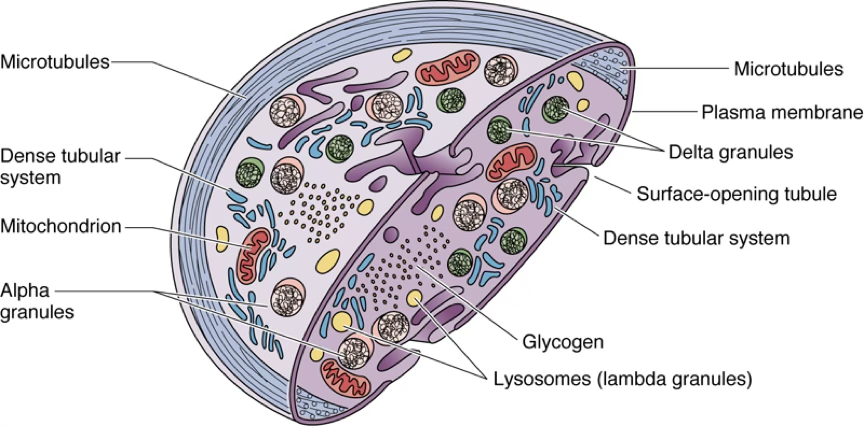
Platelets are small (–) cytoplasmic discoid fragments derived from the megakaryocyte under the control of thrombopoietin, a – glycoprotein produced in the kidneys and liver.
- Megakaryocytes develop cytoplasmic projections → proplatelets → fragment into platelets
- Differentiation process: 7–10 days
- Platelets bind and degrade thrombopoietin → a mechanism that regulates platelet production
Platelet Ultrastructure — Granule Types
Granule Type Also Called Alpha granules — Delta granules — Lysosomes Lambda granules Other structures: microtubules, plasma membrane, dense tubular system, surface-opening tubule, mitochondria, glycogen.
The periphery is occupied by actin filaments that encircle the platelet and maintain the discoid morphology.
Platelet Activation If the endothelial lining of a blood vessel is disrupted and platelets come in contact with subendothelial collagen, they become activated:
- Release contents of their granules
- Platelet adhesion — adhere to the damaged region of the vessel wall
- Platelet aggregation — adhere to each other
Interactions of tissue factors, plasma-borne factors, and platelet-derived factors form a blood clot.
What is thrombopoietin and where is it produced?
Thrombopoietin is a – glycoprotein that controls platelet production; it is produced in the kidneys and liver.
Anki cloze
Platelets are derived from {1:megakaryocytes}; the differentiation process takes {2:7–10 days}; platelet size is {3:–}.
Bone Marrow and Hematopoiesis
Bone Marrow Overview
Bone marrow is a gelatinous, vascular connective tissue located in the marrow cavity, richly endowed with cells responsible for hemopoiesis.
Red vs. Yellow Marrow
Type Description Red marrow (newborn) Active hematopoiesis; great number of erythrocytes being produced Yellow marrow (adults, diaphyses of long bones by age 20) Accumulation of large quantities of fat; absence of hematopoiesis in the shafts Red marrow (adults, cancellous bone cavities) Still present in cancellous bone; hematopoiesis occurs there
Hematopoiesis
Hematopoiesis (Greek haima, blood; poiein, to make) is the process of blood formation.
Timeline of Fetal Hematopoiesis
- First trimester — initial wave in yolk sac (islands of hematopoiesis); develops from hemangioblasts, the common progenitors of hematopoietic and endothelial cells
- After second trimester — continues in the liver, then the spleen (second definitive wave)
- 7th month of intrauterine life — bone marrow becomes the primary site; remains so during adulthood
Adult Hematopoiesis Capacity An approximate volume of 1.7 L of marrow contains hematopoietic cells, producing:
- ~ RBCs per hour
- ~ leukocytes per hour
Anki cloze
Hematopoiesis begins in the {1:yolk sac} during the first trimester, continues in the {2:liver then spleen}, and the bone marrow becomes the primary site at the {3:7th month} of intrauterine life.
Bone Marrow Niches
The bone marrow consists of two microenvironmental domains, called niches:
- The vascular niche
- The endosteal niche
Niches provide physical support, soluble factors and cell-mediated interactions to regulate cell self-renewal, differentiation and quiescence of hematopoietic stem cells (HSCs).
Under normal conditions, niches enable balanced (homeostatic) cell self-renewal and differentiation of HSCs. Under pathologic conditions (myelodysplasia, aging, bone marrow malignancies), niches can alter or restrain normal hematopoiesis.
Vascular Niche
- Consists of blood vessels surrounded by a distinct population of non-hematopoietic stromal cells: mesenchymal stem cells, adipose cells, endothelial cells, reticular stromal cells, and macrophages
- Provides a microenvironment for short-term proliferation and differentiation of HSCs
Endosteal Niche
- Located at the endosteum–bone marrow interface
- Consists of preosteoblasts (osteoprogenitor cells), osteoblasts and osteoclasts interacting with HSCs
- Most abundant extracellular component: Type I collagen
- Regarded as a site for long-term storage of quiescent HSCs
- Osteoblasts produce multiple hematopoietic cytokines: G-CSF, M-CSF, GM-CSF, IL-1, IL-6, IL-7
What is the difference between the vascular and endosteal niches?
The vascular niche supports short-term proliferation and differentiation of HSCs; the endosteal niche (at the endosteum–bone marrow interface) is a site for long-term storage of quiescent HSCs.
Anki cloze
Osteoblasts in the endosteal niche produce hematopoietic cytokines including {1:G-CSF, M-CSF, GM-CSF, IL-1, IL-6, and IL-7}.
Hematopoietic Cell Populations
The bone marrow consists of three major populations:
- HSCs — capable of self-renewal
- Committed precursor cells — responsible for the generation of distinct cell lineages
- Maturing cells — resulting from the differentiation of the committed precursor cell population
HSCs can self-renew and produce two committed precursor cells:
- Myeloid stem cell
- Lymphoid stem cell
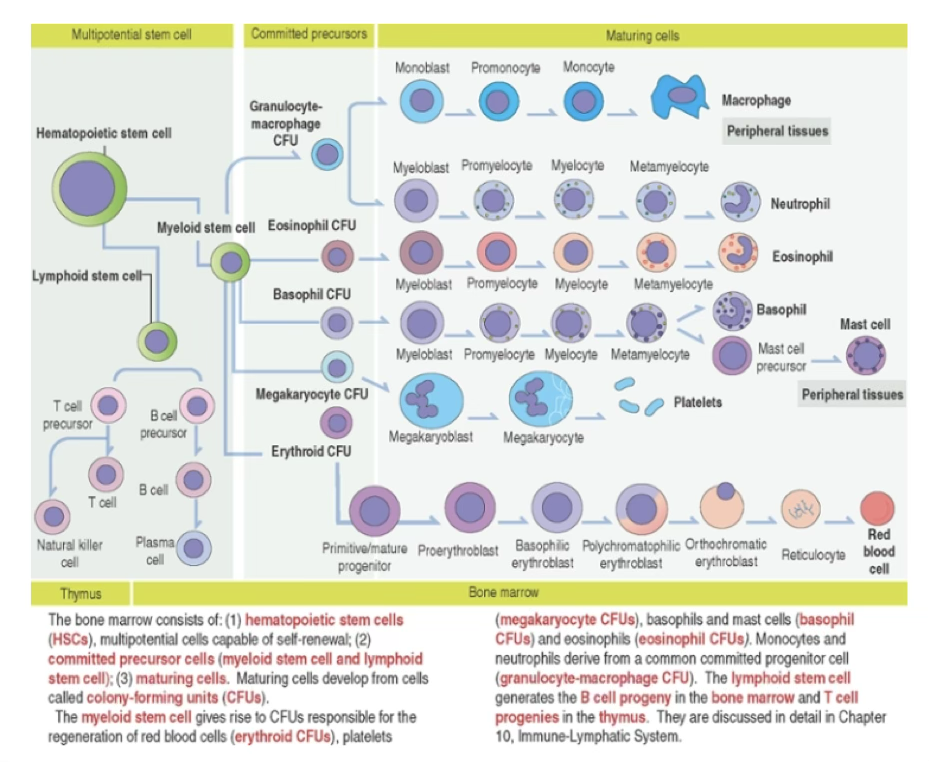
Myeloid vs. Lymphoid Lineages
Stem Cell Colony-Forming Units (CFUs) Progeny Myeloid stem cell (CFU-GEMM): granulocyte, erythrocyte, monocyte, megakaryocyte Erythroid CFUs, megakaryocyte CFUs, basophil CFUs, eosinophil CFUs; monocytes and neutrophils from granulocyte-macrophage CFU Erythrocytes, granulocytes, monocytes, platelets Lymphoid stem cell (CFU-Ly) B cell progeny in bone marrow; T cell progenies in thymus T cells and B cells
HSC Properties
- Self-renewal preserves the pool of stem cells; critical for feeding common myeloid progenitor and common lymphoid progenitor into the differentiation/maturation pathway
- HSCs represent
of total hematopoietic cells (– stem cells)- In bone marrow transplantation, only of normal HSCs are needed to repopulate the entire bone marrow
- HSCs cannot be identified by morphology; recognized by specific cell surface markers: c-kit receptor and Thy-1
Granulocyte Precursor Cells
The myeloblast and promyelocyte intermediaries in the formation of eosinophils, neutrophils, and basophils are indistinguishable for the three cell types.
What surface markers identify hematopoietic stem cells (HSCs)?
c-kit receptor and Thy-1.
Anki cloze
HSCs represent approximately {1:} of total hematopoietic cells; in bone marrow transplantation only {2:} of normal HSCs are needed to repopulate the entire marrow.
Lymphoid Organs and the Immune System
Primary vs. Secondary Lymphoid Organs
Classification of Lymphoid Organs
Type Function Organs Primary lymphoid organs Produce cell components of the immune system Bone marrow, Thymus Secondary lymphoid organs Sites where immune responses occur Lymph nodes, Spleen, Tonsils, BALT (bronchial-associated lymphoid tissue), GALT (gut-associated lymphoid tissue, including Peyer’s patches)
Key Cell Components of the Immune System
The two key cell components of the immune system are lymphocytes and accessory cells.
Lymphocytes include two major cell groups:
- B cells — responding to cell-free and cell-bound antigens
- T cells — subdivided into:
- Helper T cells
- Cytolytic or cytotoxic T cells
- T cells respond to cell-bound antigens presented by specific molecules
Types of Immunity
Immunity is the reaction of cells and tissues to foreign (non-self) substances or pathogens, including bacterial, viral and parasite antigens.
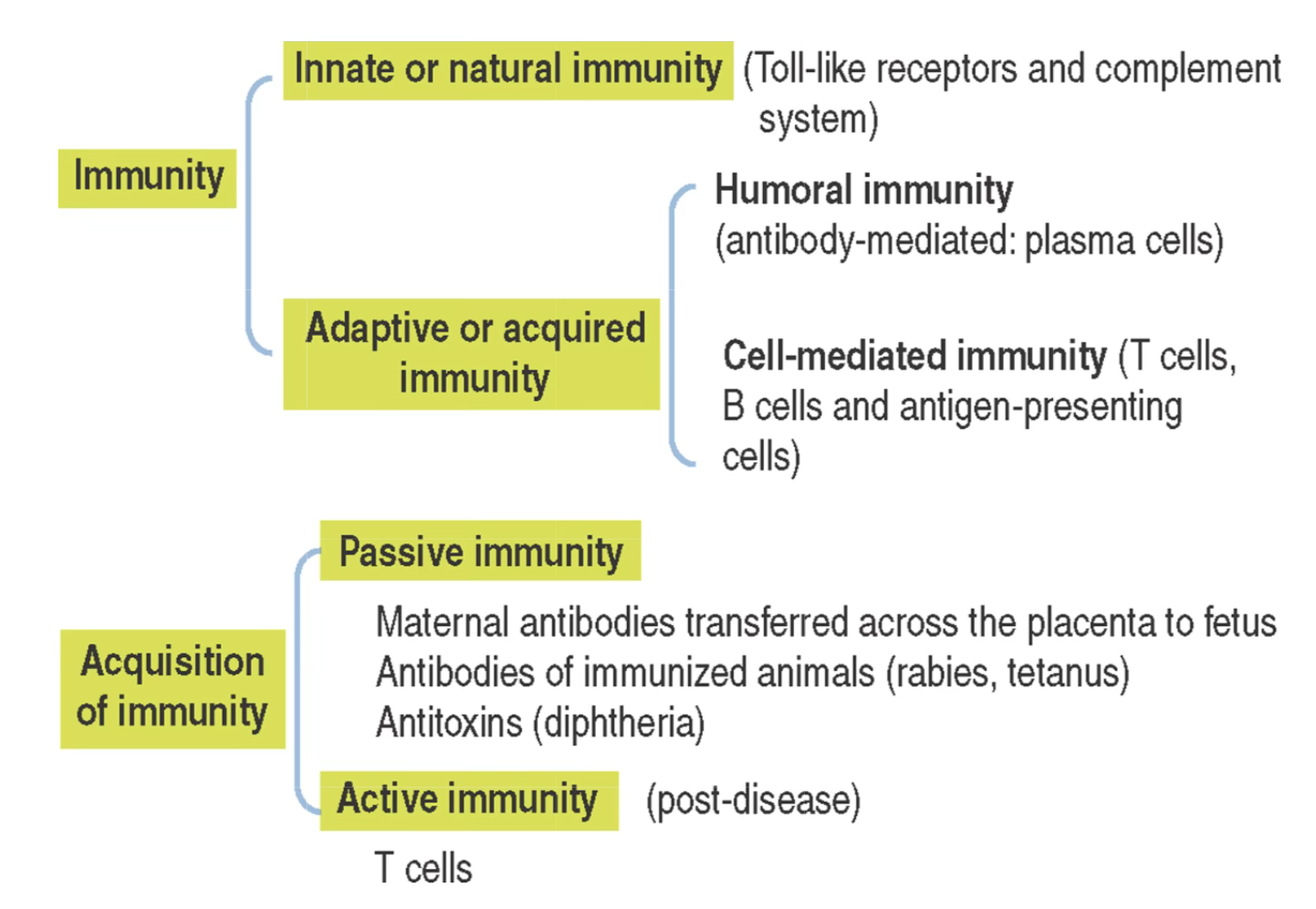
Two Types of Immunity
Type Characteristics Innate (natural) immunity Does not require previous exposure; elicits rapid responses by macrophages and dendritic cells; mediated by Toll-like receptors (TLRs) Adaptive (acquired) immunity Develops upon exposure to pathogen; aims to eliminate pathogen and generate immunologic memory; requires clonal selection via gene rearrangement
Innate Immunity — Toll-Like Receptors
- Toll-like receptors (TLRs) initiate innate immunity against components of invading pathogens (nucleic acids, proteins, lipids and polysaccharides)
- Different TLRs recognize distinct types of conserved microbial structures → provides specificity to the innate response
- Innate immunity of the newborn is the simplest mechanism of protection
Adaptive Immunity
To achieve adaptive immunity, it is necessary to select lymphocytes (clonal selection) from a vast repertoire of cells bearing antigen-specific receptors generated by a mechanism known as gene rearrangement.
Summary — Branches of Immunity
- Innate/natural immunity: Toll-like receptors and complement system
- Humoral immunity: antibody-mediated; plasma cells
- Cell-mediated immunity: T cells, B cells and antigen-presenting cells
- Maternal antibodies transferred across the placenta to fetus
- Antibodies of immunized animals (rabies, tetanus)
- Antitoxins (diphtheria)
What is the role of Toll-like receptors (TLRs) in innate immunity?
TLRs initiate innate immunity by recognizing distinct conserved microbial structures (nucleic acids, proteins, lipids, polysaccharides), providing specificity to the innate response.
Anki cloze
Adaptive immunity requires {1:clonal selection} of lymphocytes bearing antigen-specific receptors generated by {2:gene rearrangement}.
Major Lymphoid Organs
The major lymphoid organs are:
- The lymph nodes
- The thymus
- The spleen
Lymph Nodes
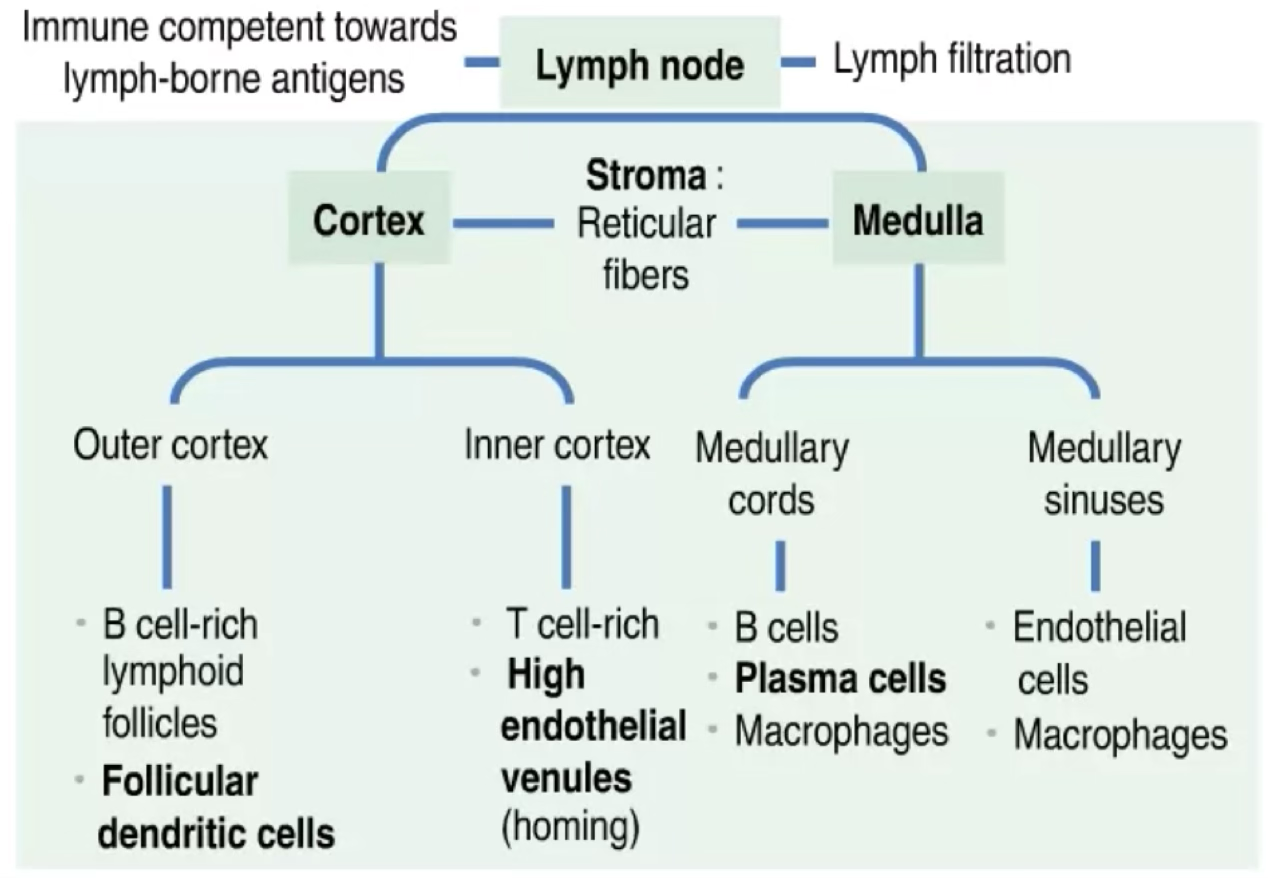
Function: filter the lymph, maintain and differentiate B cells, and house T cells; detect and react to lymph-borne antigens.
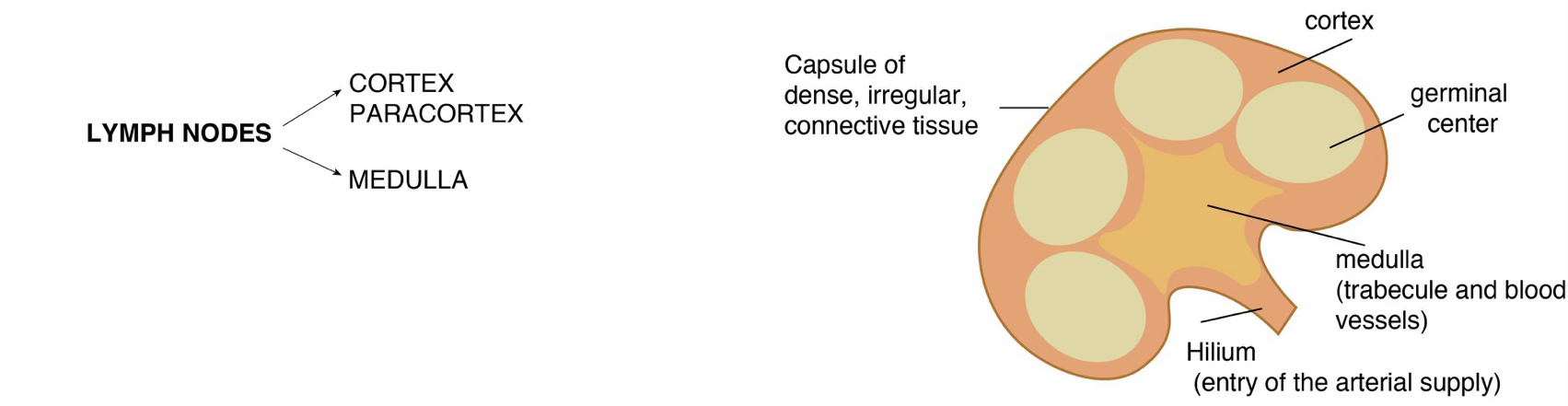
Lymph Node Structure
- Surrounded by a capsule (dense irregular connective tissue surrounded by adipose tissue)
- Parenchyma divided into a cortex and a medulla
- Capsule pierced at convex surface by afferent lymphatic vessels (have valves to prevent reflux)
- Stroma/parenchyma: interconnected fibroblastic reticular cells and fibers forming an open network → enables B-cell and T-cell compartmentalization and survival, transport of antigens and signaling molecules
Cortex
The cortex has two zones:
| Zone | Contents |
|---|---|
| Outer cortex | Lymphoid follicles, rich in B lymphocytes; follicular dendritic cells (FDCs), migrating dendritic cells, macrophages, supporting reticular cells |
| Deep/inner cortex (paracortex) | CD4 helper T cells; venules lined by high endothelial cells (site of homing) |
Lymphoid Follicle Structure
A lymphoid follicle consists of:
- A mantle (facing the cortex)
- A germinal center containing mainly proliferating B cells or lymphoblasts, resident follicular dendritic cells (FDCs), migrating dendritic cells, macrophages, and supporting reticular cells (which produce reticular fibers = type III collagen)
The deep/inner cortex is a zone where mainly CD4 helper T cells interact with B cells to induce their proliferation and differentiation when exposed to a specific antigen.
Medulla
The medulla is surrounded by the cortex, except at the hilum (a concave surface where efferent lymphatic vessels and a single vein leave, and an artery enters).
| Component | Contents |
|---|---|
| Medullary sinusoids | Spaces lined by endothelial cells surrounded by reticular cells and macrophages |
| Medullary cords | B cells, macrophages and plasma cells |
Plasma Cell Positioning
Activated B cells migrate from the cortex, enter the medullary cords, and become plasma cells. This is a strategic location because plasma cells can secrete immunoglobulins directly into the lumen of the medullary sinuses without leaving the lymph node.
What type of T cells are found in the deep cortex (paracortex) of a lymph node?
CD4 helper T cells.
Anki cloze
The outer cortex of lymph nodes is rich in {1:B lymphocytes} organized in lymphoid follicles; the deep/inner cortex houses mainly {2:CD4 helper T cells} and high endothelial venules.
Anki cloze
Activated B cells migrate to medullary cords and become {1:plasma cells}, which can secrete {2:immunoglobulins} directly into medullary sinuses.
Thymus
The thymus consists of two lobes subdivided into several incomplete lobules, each consisting of an independent cortex and a shared medulla.
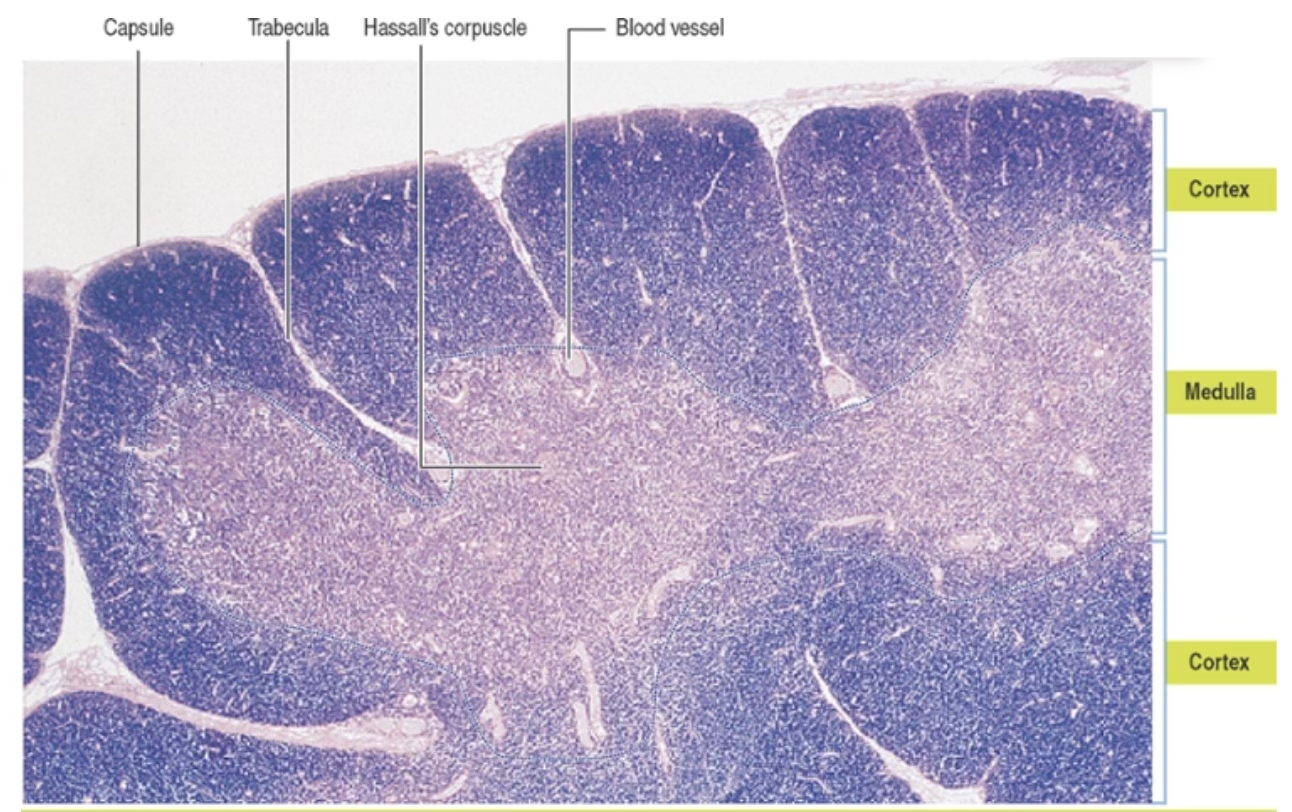
Thymic Medulla
- Contains mature thymocytes migrating from the cortex
- Maturation of thymocytes is completed in the medulla; functional thymocytes enter postcapillary venules at the corticomedullary junction to exit
- Harbors medullary thymic epithelial cells (mTECs); a subset forms Hassall’s corpuscles
- Hassall’s corpuscles: aggregates of terminally differentiated cornified mTECs; present only in the medulla of the thymus
- Blood-thymus barrier is not present in the medulla
Thymus — Unique Stromal Feature
Unlike the lymph node and spleen (which contain reticular cells and reticular fibers with no stromal epithelial cells), the stroma of the thymus consists of thymic epithelial cells (TECs) organized in a dispersed network to allow for intimate contact with developing thymocytes.
Specialized cTECs (cortical) and mTECs (medullary) provide the required checkpoints for thymocyte selection.
Thymus Development
- The mesenchyme gives rise to the capsule, trabeculae and vessels of the thymus
- The thymic epithelial rudiment attracts bone marrow–derived thymocyte precursors, dendritic cells and macrophages
- During fetal life, the thymus contains lymphocytes derived from the liver
- T-cell progenitors formed in the bone marrow enter the thymus as immature thymocytes and mature to become predominantly CD4 or CD8 cells, which are carried into lymph nodes, spleen and other lymphatic tissues
- The thymus is fully developed before birth
- T cell production is significant before puberty
- After puberty, the thymus begins to involute and T cell production decreases
What are Hassall's corpuscles and where are they found?
Hassall’s corpuscles are aggregates of terminally differentiated cornified medullary thymic epithelial cells (mTECs); they are found only in the medulla of the thymus.
Anki cloze
The thymus stroma consists of {1:thymic epithelial cells (TECs)}, unlike the lymph node and spleen which have {2:reticular cells and reticular fibers}.
Spleen
The spleen is the largest secondary lymphoid organ of the body. It lacks a cortex and a medulla.
The spleen has two major components:

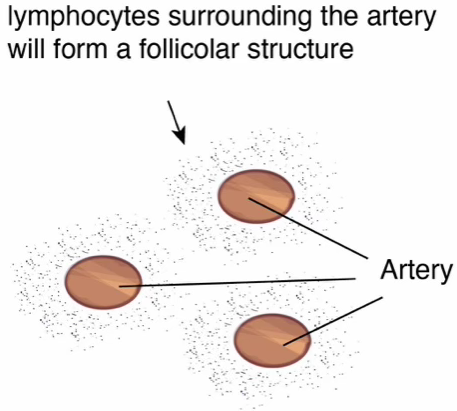
| Component | Function |
|---|---|
| White pulp | Immune component; similar cell composition to lymph node except antigens enter from blood (not lymph); has no afferent lymphatic vessels |
| Red pulp | Filter: removes aged and damaged RBCs and microorganisms from blood; storage site for RBCs |
| ![[Pasted image 20260413173002.png | 500]] |
White Pulp — Four Components
- The central artery/arteriole
- The periarteriolar lymphoid sheath (PALS)
- A splenic follicle formed by B cells (in the perifollicular zone), reticular cells and antigen-presenting cells
- A germinal center
Red Pulp
- Surrounds the white pulp
- Receives a significant blood supply from the blood
- White and red pulp interact at the marginal channel (a permeable gate between the two compartments)
- Bacteria can be recognized by macrophages of the red pulp and removed directly or after being coated with complement proteins (produced in the liver) and immunoglobulins (produced in the white pulp)
Spleen vs. Lymph Node — Key Differences
- The central artery/arteriole is a distinctive feature of the spleen (not present in lymph nodes)
- The spleen has no afferent lymphatic vessels; antigens enter from the blood
- Lymph nodes receive antigens through afferent lymphatic vessels
What are the two major compartments of the spleen and their functions?
White pulp: immune responses (similar to lymph node, but antigens arrive from blood). Red pulp: filters aged/damaged RBCs and microorganisms; stores RBCs.
Anki cloze
The spleen’s white pulp consists of the central artery, {1:periarteriolar lymphoid sheath (PALS)}, a {2:splenic follicle} with B cells, and a {3:germinal center}.
Anki cloze
Unlike lymph nodes, the spleen has {1:no afferent lymphatic vessels} and antigens enter via the {2:blood}; a distinctive feature of the spleen is the {3:central artery/arteriole}.
TLDR
Blood and Lymphoid System — Key Points
- Blood is a specialized connective tissue; plasma () + formed elements (); hematocrit = –; serum = fibrinogen-free plasma
- RBCs: biconcave, anucleate, , -day lifespan; filled with hemoglobin (tetrameric, iron-heme); oxyhemoglobin () vs. carbaminohemoglobin (); replaced by reticulocytes (–); A/B/O and Rh blood groups from surface carbohydrate antigens
- Leukocytes: granulocytes (primary + specific granules) vs. agranulocytes (primary granules only)
- Neutrophils: – WBCs; multilobed nucleus; pale pink; first responders in bacterial infection; primary granules (elastase, defensins, myeloperoxidase) + secondary granules (lactoferrin, lysozyme, gelatinase); lifespan 6–7 h in blood, ≤4 days in tissue; “drumstick” in females = inactive X chromosome
- Eosinophils: – WBCs; bilobed nucleus; salmon-pink granules; anti-parasite defense + antigen-antibody complex phagocytosis; recruited by IL-5; bronchial asthma and eosinophilic esophagitis
- Basophils: WBCs; S-shaped nucleus masked by dark blue/purple granules; IgE receptors (FcεR1); role in asthma, type 2 immunity; phenotype: c-kit FcεR1 CD49b (vs. mast cells: c-kit FcεR1 CD49b); shorter-lived than mast cells
- Lymphocytes: – WBCs; B cells (mature in bone marrow → plasma cells) and T cells (mature in thymus → cell-mediated immunity); thin basophilic rim cytoplasm
- Monocytes: largest leukocyte (), kidney-shaped nucleus, bluish-gray azurophilic cytoplasm; differentiate into macrophages (connective tissue) or osteoclasts (bone)
- Platelets: discoid megakaryocyte fragments; thrombopoietin (kidney + liver) regulates production; activation by subendothelial collagen → adhesion → aggregation → blood clot; granule types: alpha, delta, lambda (lysosomes)
- Bone marrow: red (active hematopoiesis) vs. yellow (fat, inactive); vascular niche (short-term HSC proliferation) and endosteal niche (long-term quiescent HSC storage, Type I collagen, osteoblasts secrete G-CSF/M-CSF/GM-CSF/IL-1/IL-6/IL-7)
- Hematopoiesis: yolk sac (1st trimester) → liver/spleen (after 2nd trimester) → bone marrow (7th month, permanent); adult: cells in 1.7 L marrow; RBCs/hour
- HSCs: of hematopoietic cells; c-kit Thy-1; self-renewal critical; only needed to repopulate marrow in transplant; give rise to CFU-GEMM (myeloid) and CFU-Ly (lymphoid)
- Immunity: innate (TLRs, rapid, no prior exposure needed) vs. adaptive (clonal selection, gene rearrangement, immunologic memory); humoral (B cells/plasma cells/antibodies) vs. cell-mediated (T cells)
- Primary lymphoid organs: bone marrow (B cells), thymus (T cells)
- Secondary lymphoid organs: lymph nodes, spleen, tonsils, BALT, GALT (Peyer’s patches)
- Lymph node: filters lymph; outer cortex (B cells, follicles with FDCs, germinal centers, type III collagen); paracortex (CD4 helper T cells, HEVs); medulla (sinusoids + cords with plasma cells secreting Ig directly)
- Thymus: two lobes, incomplete lobules; stroma = TECs (unique — not reticular cells); cortex (immature thymocytes) + medulla (mature thymocytes, Hassall’s corpuscles from mTECs, no blood-thymus barrier); involutes after puberty; cTECs + mTECs provide thymocyte selection checkpoints
- Spleen: largest secondary lymphoid organ; no cortex/medulla; white pulp (PALS, splenic follicle, germinal center, central artery — key distinguishing feature; antigens from blood, no afferent lymphatic vessels) + red pulp (removes aged RBCs; macrophages destroy bacteria using complement + immunoglobulins); marginal channel separates compartments