TARGET DECK: MED::I::Morphology and Development::Histology::09 - Bone
Overview
What is Bone?
Bone is a rigid, inflexible connective tissue in which the extracellular matrix (ECM) has become impregnated with salts of calcium and phosphate by a process called mineralization. It is highly vascularized and metabolically very active. → first difference between bone and cartilage.
Functions of Bone
| Function | Details |
|---|---|
| Structural support & protection | Supports body; protects brain, spinal cord, and organs |
| Lever system | Multiplies muscle force to produce movement |
| Mineral reservoir | Stores ~99% of body’s calcium and phosphate ions |
| Hematopoiesis | Houses bone marrow in the central marrow cavity |
What process impregnates the bone ECM with calcium and phosphate salts?
Mineralization
Anki cloze
Bone stores approximately {1:99%} of the body’s calcium.
Gross Structure of Bone
Surface Coverings
- Periosteum — covers the external surface of bone (except when close to synovial articulations); consists of:
- Outer layer: dense fibrous connective tissue
- Inner (cellular) layer: contains osteoprogenitor (osteogenic) cells
- Endosteum — lines the central (marrow) cavity; a specialized thin connective tissue composed of a monolayer of osteoprogenitor cells and osteoblasts
Compared to the cartilage...
The only reservoir of stem cells in cartilage is… ???? TODO
What are the two layers of the periosteum?
- Outer layer: dense fibrous connective tissue
- Inner (cellular) layer: osteoprogenitor (osteogenic) cells
Composition of Bone Matrix
Fibers
- Primarily type I collagen
Ground Substance
- Rich in proteoglycans with chondroitin sulfate and keratan sulfate side chains.
- Those are the substances that will undergo mineralization.
- Glycoproteins present:
- Osteonectin
- Osteocalcin
- Osteopontin
- Bone sialoprotein
Anki cloze
The primary collagen type found in bone matrix is {1:type I collagen}.
Name four glycoproteins found in bone ground substance.
Osteonectin, osteocalcin, osteopontin, bone sialoprotein
Cells of Bone
There are four cell types in bone:
| Cell Type | Origin | Primary Function |
|---|---|---|
| Osteoprogenitor cells | Embryonic mesenchyme | Mitotic; differentiate into osteoblasts |
| Osteoblasts | Osteoprogenitor cells | Synthesize organic component of the bone matrix (osteoid) |
| Osteocytes | Trapped osteoblasts | Bone maintenance; mechanotransduction |
| Osteoclasts | Bone marrow (GM-CFU) | Bone resorption |
Mnemonic — Bone Cell Types
“Progenitors Build Cages, Clasts Demolish”
- Progenitor → osteoprogenitor
- Build → osteoblast (builds matrix)
- Cages → osteoCyte (trapped in lacunae)
- Demolish → osteoclast (resorbs bone)
Osteoprogenitor Cells
- Located in: inner cellular layer of periosteum, lining of haversian canals, endosteum
- Derived from embryonic mesenchyme
- Can undergo mitotic division and differentiate into osteoblasts
Osteoblasts
- Derived from osteoprogenitor cells
- Arranged in a sheet-like pattern of cuboidal to columnar cells on bone surfaces
- Synthesize organic matrix components: collagen, proteoglycans, glycoproteins
- When actively secreting matrix → basophilic cytoplasm (due to abundant RER)
The osteoblast secretes it’s own cage.
Anki cloze
When actively secreting matrix, osteoblasts exhibit a {1:basophilic} cytoplasm.
Osteocytes
- Osteoblasts that have become trapped within mineralized matrix
- Conform to the shape of their lacunae
- Nucleus is flattened; cytoplasm poor in organelles (scant RER, reduced Golgi)
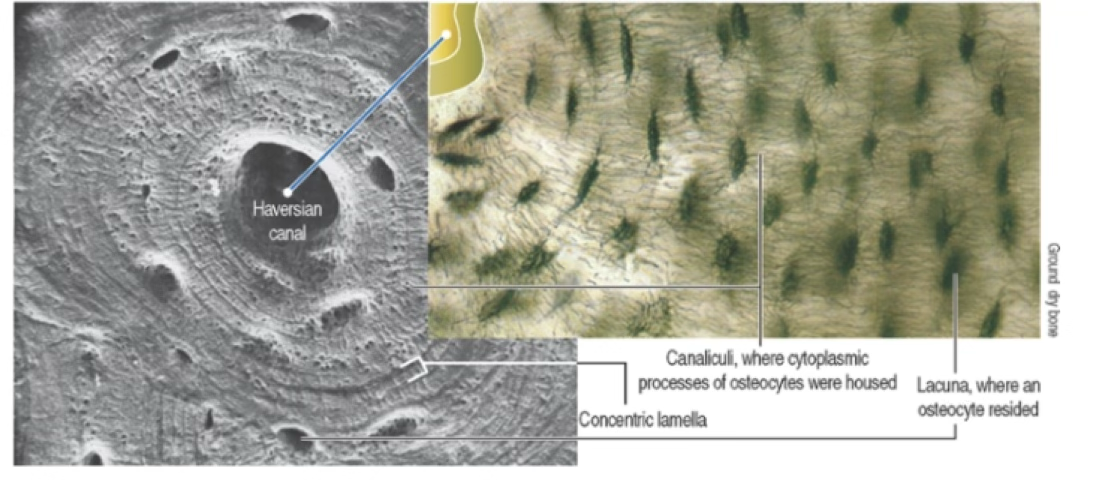
- Cell processes (projections) reside in canaliculi, which connect the osteocytes that live in different layers (lamellae)
- Although appearing inactive, they secrete substances necessary for bone maintenance
Mechanotransduction by Osteocytes
Osteocyte Mechanotransduction
Osteocytes respond to mechanical tension on bone by releasing:
- cAMP
- Osteocalcin
- Insulin-like growth factor (IGF)
These factors recruit preosteoblasts to assist in skeletal remodeling — both during growth and during long-term redistribution of forces.
What three factors do osteocytes release in response to mechanical tension?
cAMP, osteocalcin, and insulin-like growth factor (IGF)
Osteoclasts
Overview
- Large, motile, multinucleated cells (~ in diameter)
- Contain up to 50 nuclei
- Acidophilic cytoplasm
- Responsible for bone resorption; undergo apoptosis after completion
Origin
They do not derive from the same progenitor as the other bone-related cells.
- Previously thought to derive from fusion of blood-derived monocytes
- Current evidence: originate from a bone marrow precursor shared with monocytes → the granulocyte-macrophage progenitor cell (GM-CFU)
- Precursor cells stimulated by:
- Macrophage colony-stimulating factor (M-CSF)
- OPGL (osteoprotegerin ligand)
- In the presence of bone, precursors fuse to form the multinucleated osteoclast
What is the bone marrow precursor of osteoclasts?
The granulocyte-macrophage progenitor cell (GM-CFU), shared with monocytes
Receptors on Osteoclasts
- Osteoclast-stimulating factor
- Colony-stimulating factor-1
- OPGL
- Osteoprotegerin
- Calcitonin (inhibits resorption)
Howship’s Lacunae
Osteoclasts occupy shallow depressions called Howship's lacunae — these identify regions of active bone resorption.
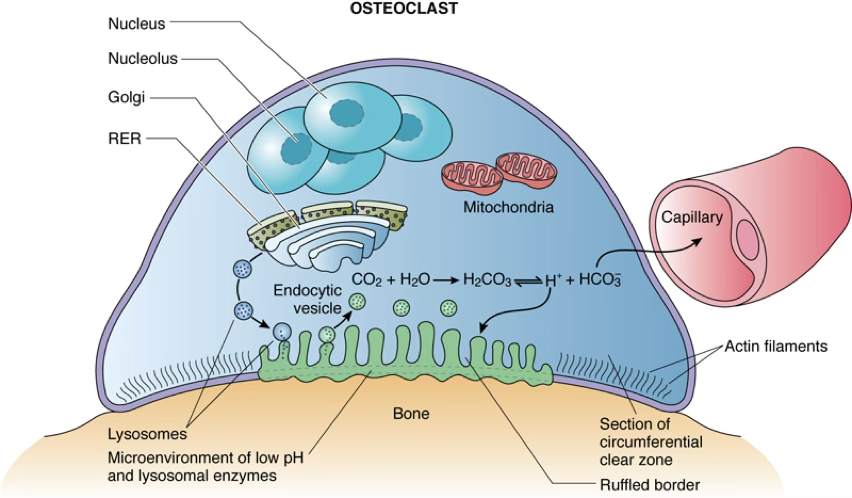
Morphological Zones of an Active Osteoclast
| Zone | Description |
|---|---|
| Ruffled Border | Highly folded plasma membrane facing bone; formed by fusion of lysosomal vesicles; increases surface area for secretion of and lysosomal proteases |
| Clear Zone (Sealing Zone) | (Periphery to the Ruffled Border) Dense area rich in actin filaments and integrins; physically anchors osteoclast to bone; seals off the resorption pit |
| Vesicular Zone | (Identified inside the cell) Packed with lysosomes, vesicles, mitochondria; transports enzymes to ruffled border and resorbed materials out of cell |
| Basal Zone (Basolateral Membrane) | Opposite side of cell; involved in exocytosis of debris; contains high concentrations of transporters and ion channels |
Mnemonic — Osteoclast Zones (outside to inside, facing bone)
“Ruffled Cats Vomit Badly” Ruffled border → Clear zone → Vesicular zone → Basal zone
What is the function of the ruffled border in osteoclasts?
It is a highly folded plasma membrane (formed by lysosomal vesicle fusion) that secretes HCl and lysosomal proteases to resorb bone, increasing surface area for this process.
Anki cloze
The {1:clear (sealing) zone} of an osteoclast is rich in actin filaments and integrins and anchors the cell to bone.
Macroscopic Structure of Mature Bone
Two Gross Forms
| Type | Appearance | Location |
|---|---|---|
| Compact (dense) bone | Solid mass | Diaphysis (cortex of long bones) |
| Spongy (trabecular/cancellous) bone | Network of bony spicules/trabeculae with marrow-filled spaces | Epiphyses; inner core |
Long Bone Anatomy
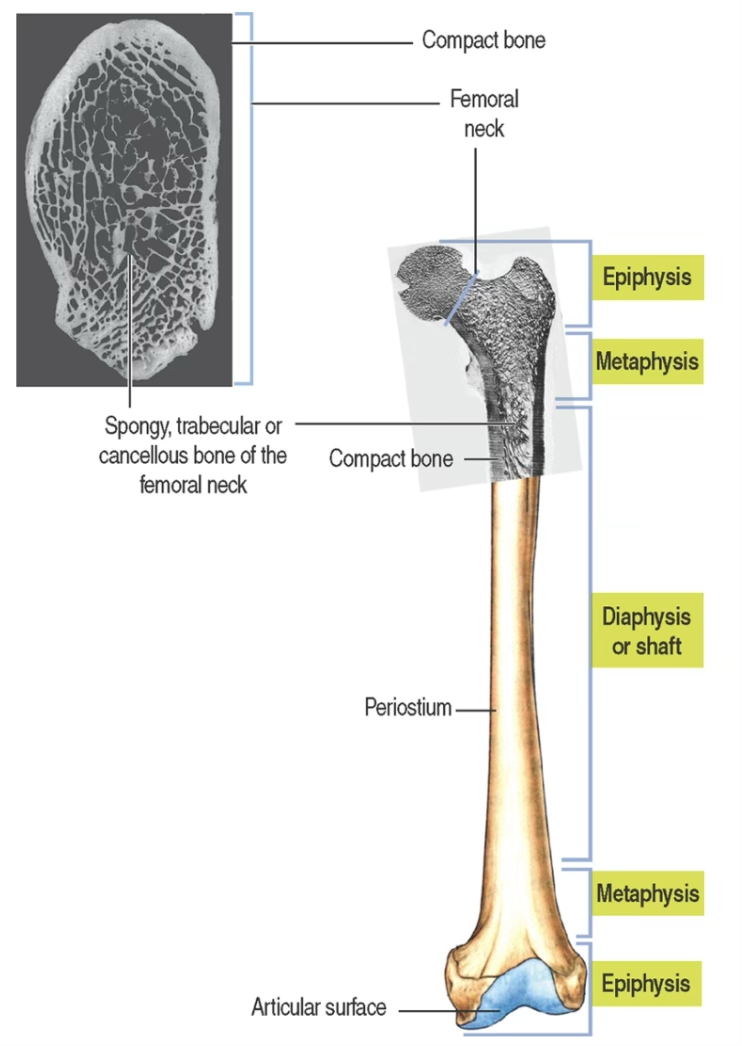
- Diaphysis:
- mainly compact bone forming a hollow cylinder; central marrow cavity, hosting the bone marrow.
- Epiphyses:
- spongy bone covered by a thin layer of compact bone; covered by articular cartilage (hyaline)
When classifying a bone, we always have to think about the development stage.
In the growing individuals, we will identify a special region separating the epiphysis from the diaphysis by a cartilaginous space → epiphyseal plate.
The growing zone is mainly made by cartilage.
- Epiphyseal plate:
- cartilaginous plate separating epiphysis from diaphysis in growing individuals; the growth zone
- Metaphysis:
- transitional region connecting epiphysis and diaphysis
- Periosteum:
- covers most of the outer bone surface (except articular surfaces and tendon/ligament insertions)
- Endosteum:
- lines the marrow cavity of the diaphysis and spaces within spongy bone; contains osteoprogenitor cells with osteogenic potential
Anki cloze
The {1:epiphyseal plate} is the cartilaginous growth zone responsible for increase in length of growing long bones.
Microscopic Structure of Mature Bone
Two Microscopic Types (based on collagen 3D orientation)
| Type | Collagen Arrangement | Mechanical Strength/Resilience | Formation/Development Rate | Typical of… |
|---|---|---|---|---|
| Lamellar (compact) bone | Regular, alternating helix orientation in successive lamellae | Strong | Slow | Mature bone |
| Woven bone | Irregular, random | Weak | Rapid | Developing bone; fracture repair |
How does woven bone differ from lamellar bone microscopically?
Woven bone has irregularly (randomly) oriented collagen fibers and is mechanically weak; lamellar bone has regularly aligned collagen fibers in alternating helical orientations and is mechanically strong.
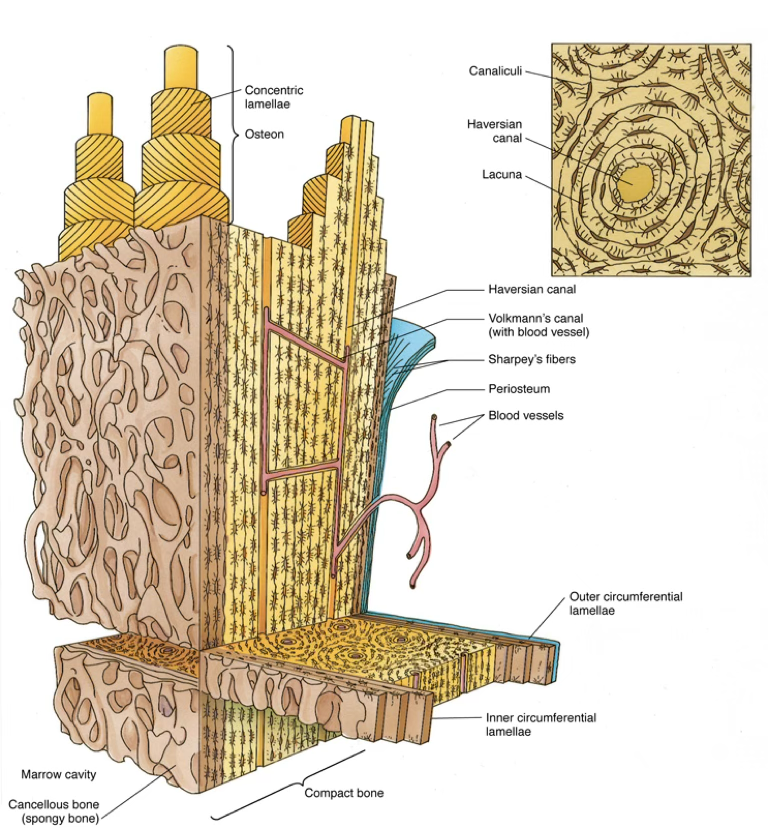
Lamellar Bone — Four Patterns
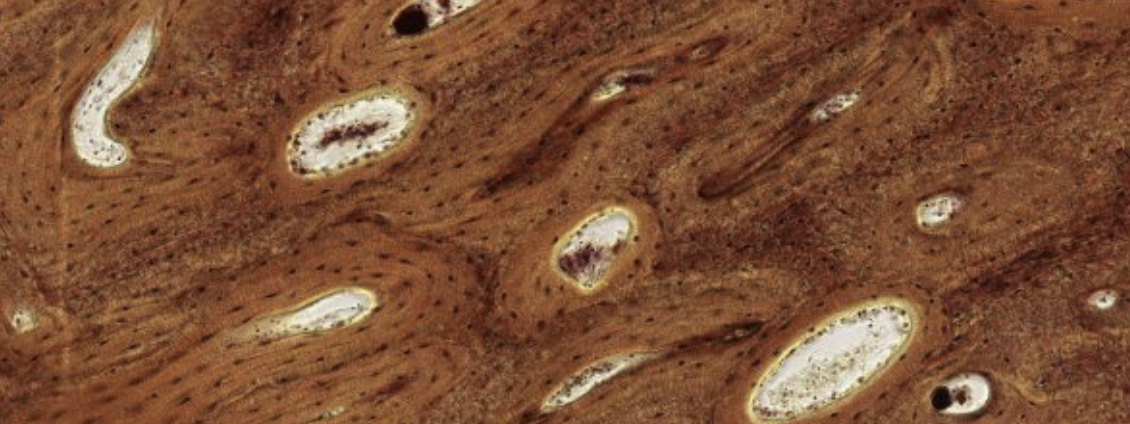
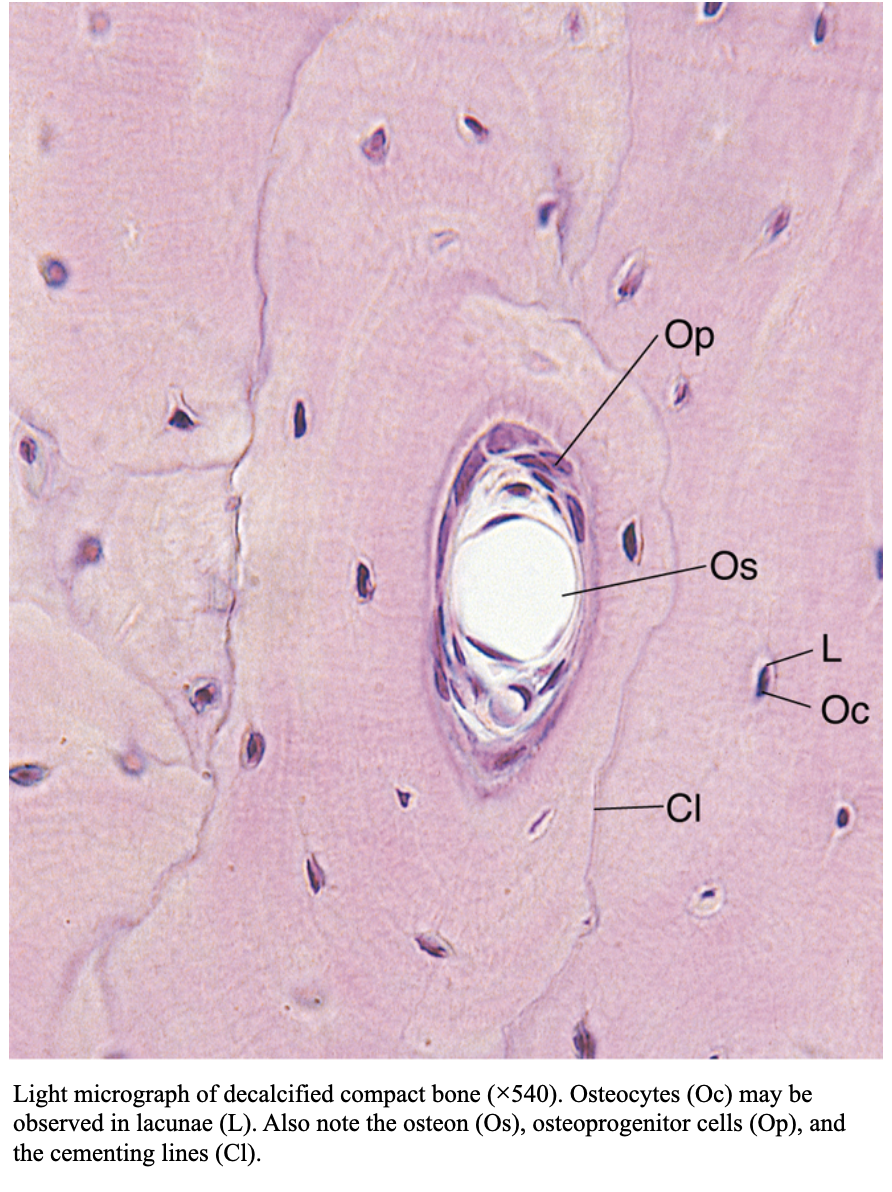
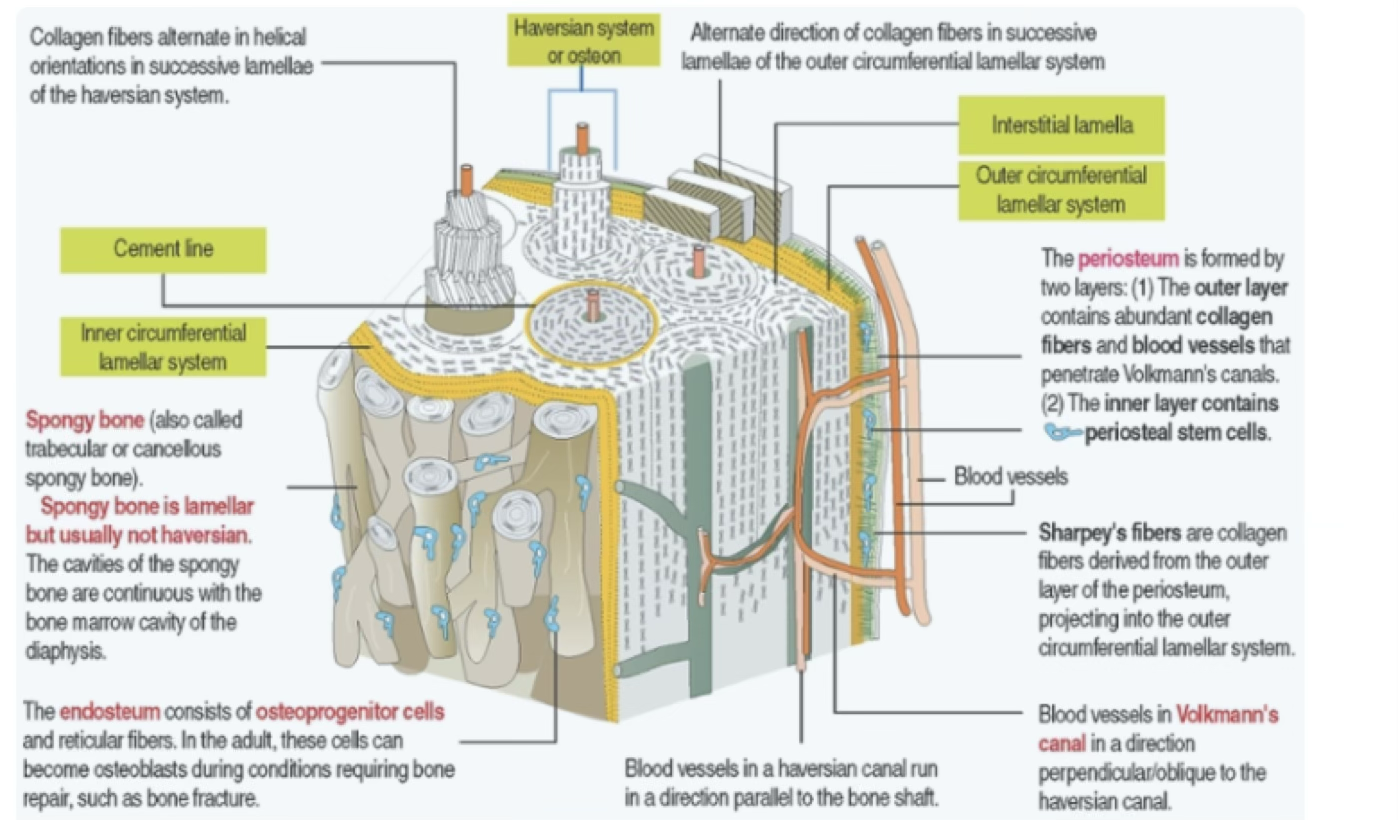
- Osteons (Haversian systems):
- concentrically arranged lamellae (4–20) around a central longitudinal vascular channel (haversian canal)
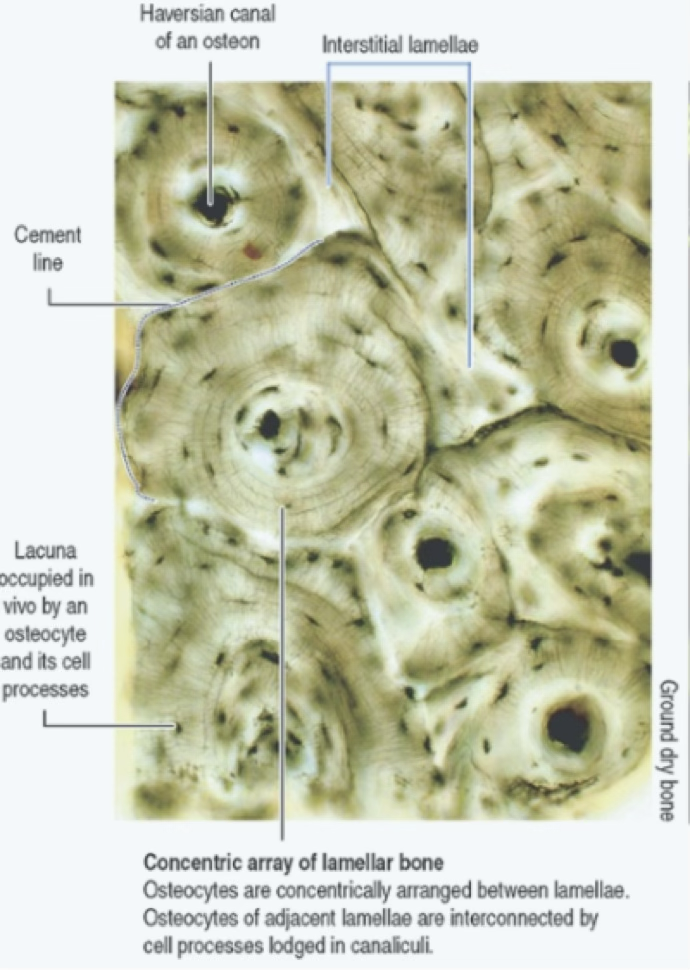
- Interstitial lamellae:
- remnants of old osteons between current osteons; separated by a thin layer called cement line
- Outer circumferential lamellae:
- at the external surface of compact bone, beneath the periosteum
- Inner circumferential lamellae:
- at the internal surface, adjacent to the endosteum
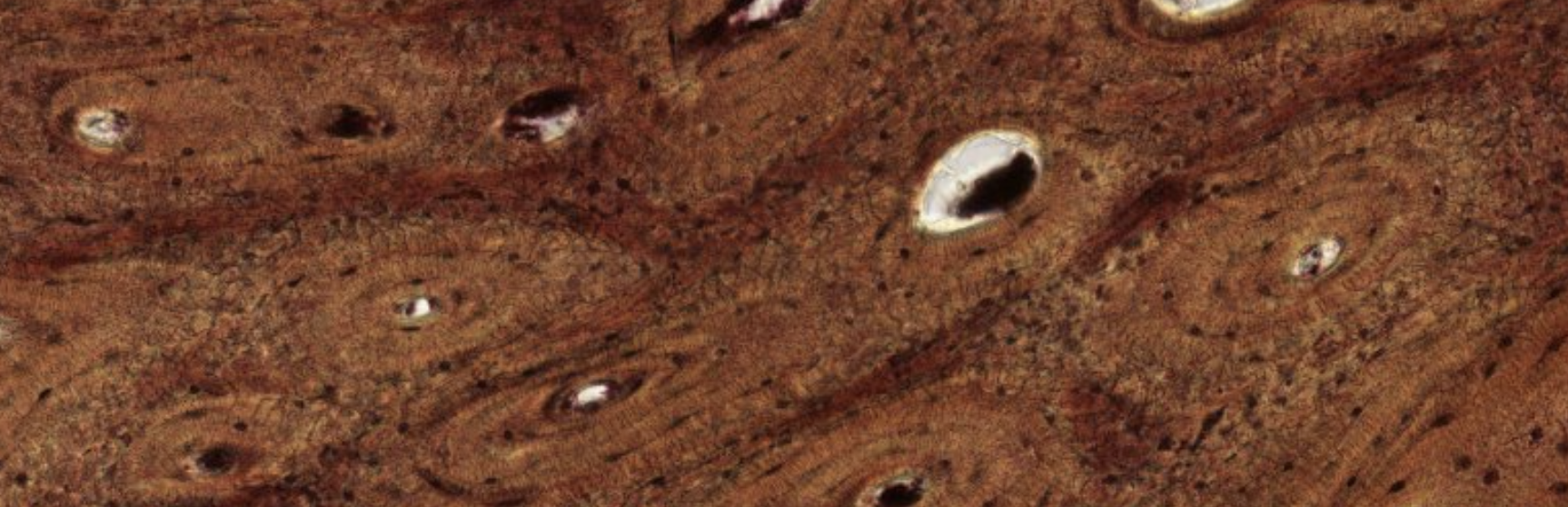
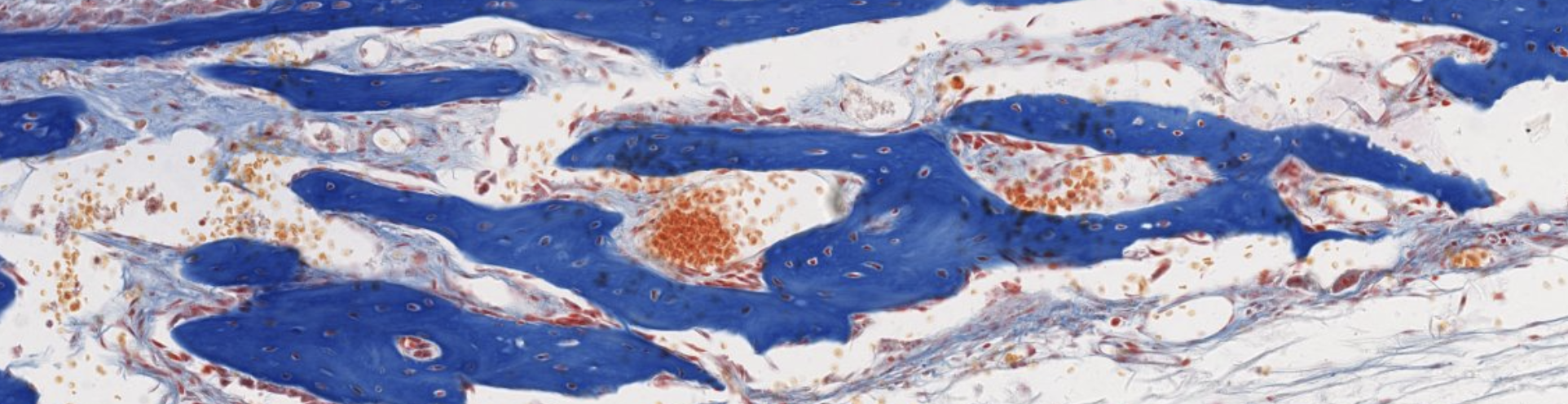
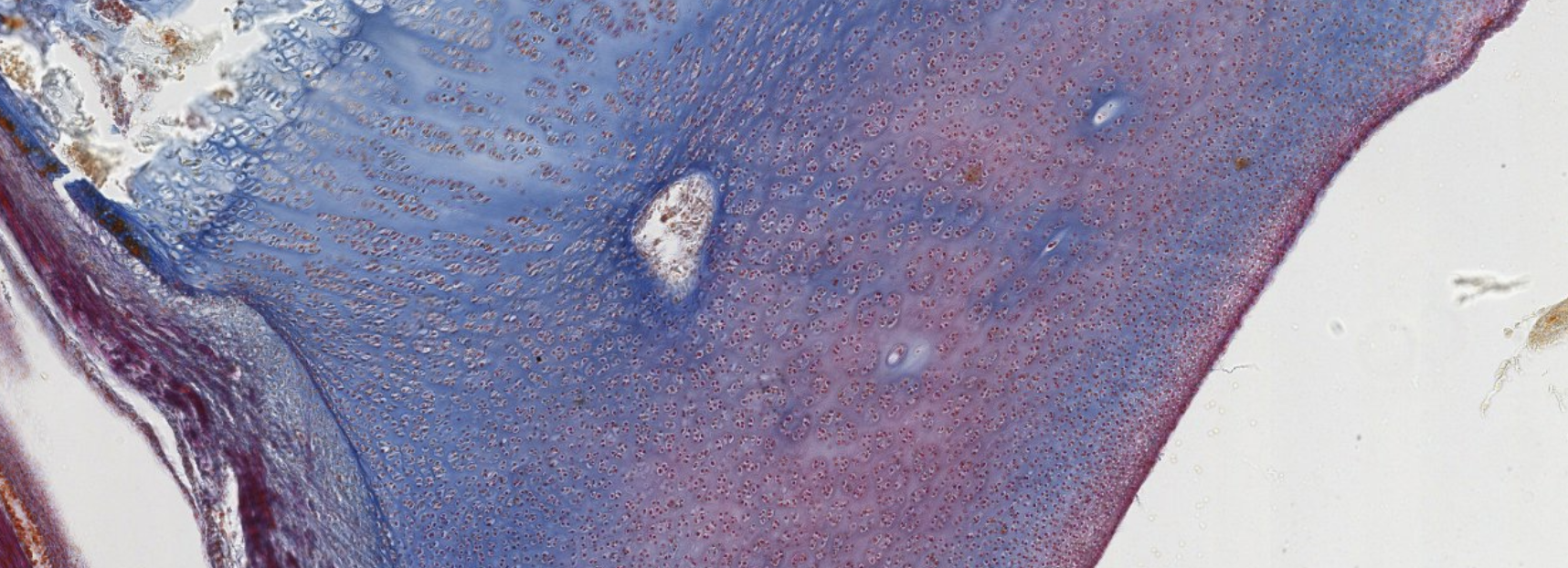
Classifying... https://unibo.smartzoom.com/s1241/course1776/f1841/i1847/
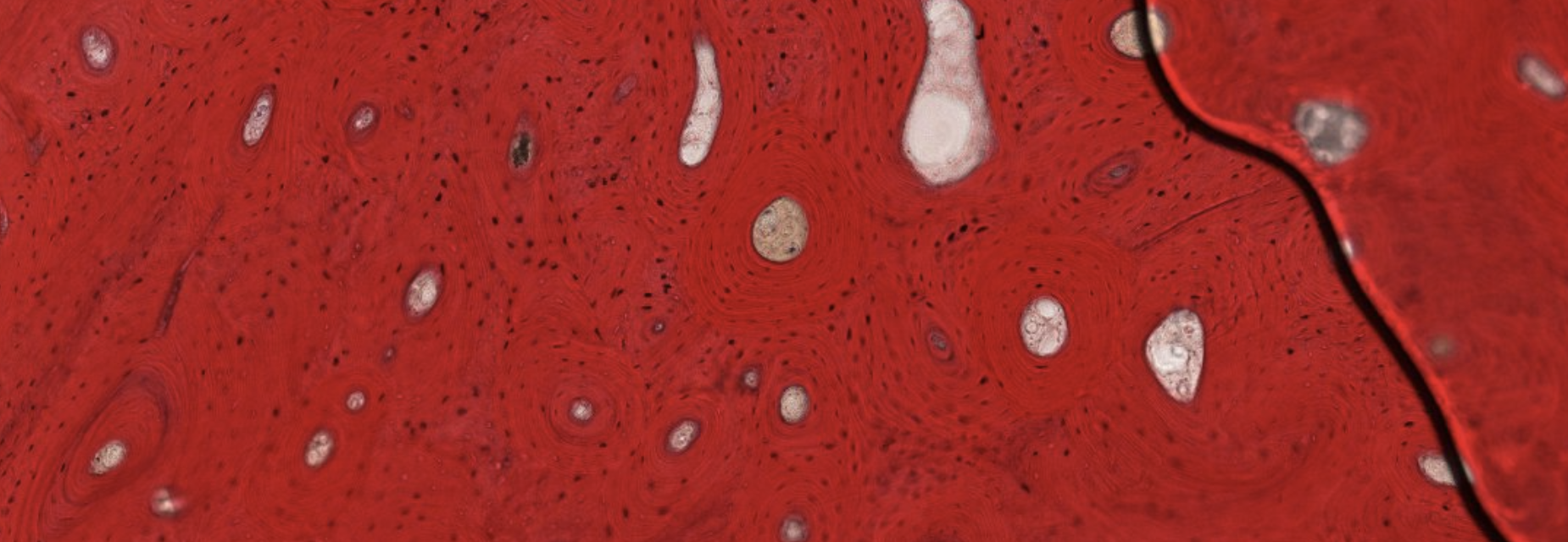
Compact bone → Hardest tissue in our body.
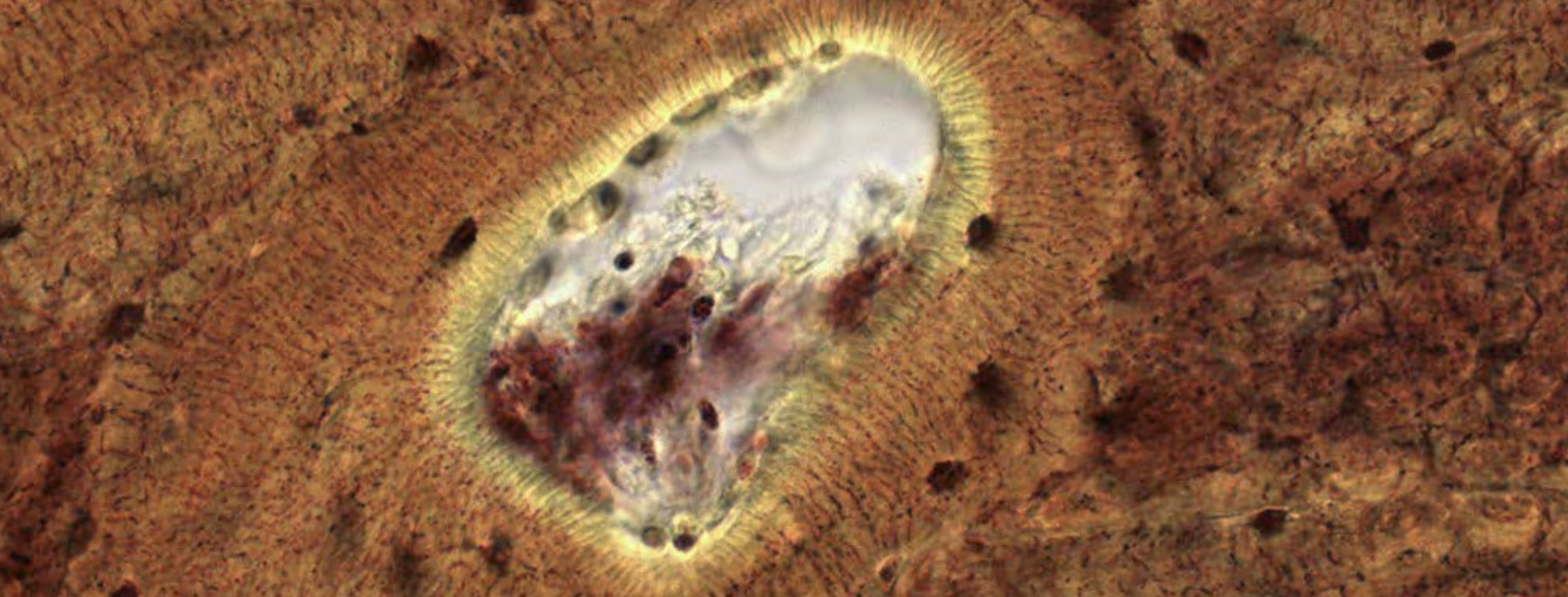
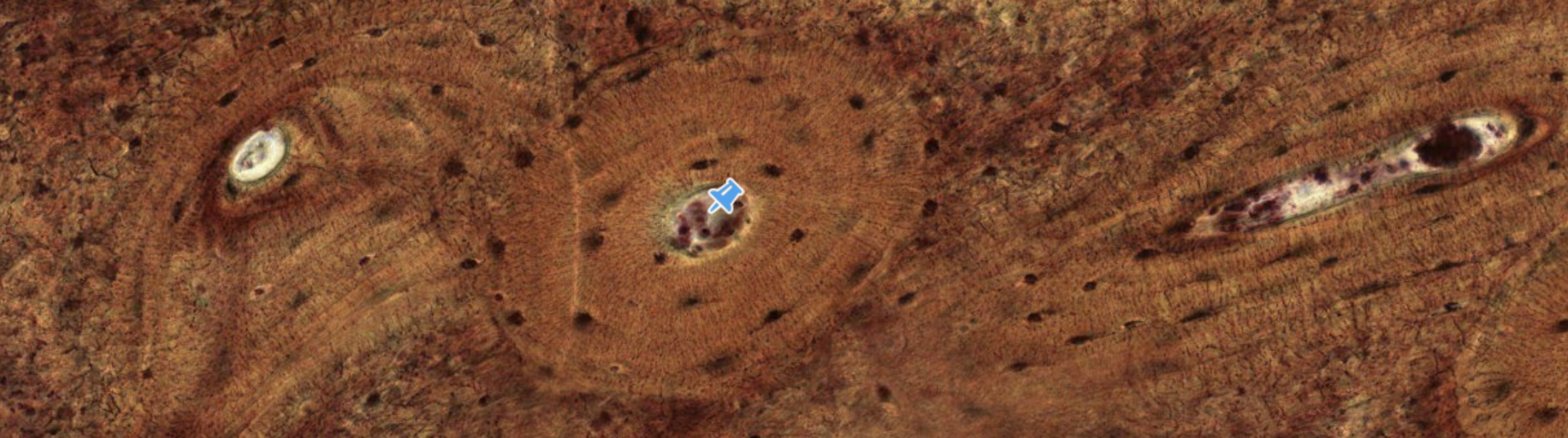
The round structures are osteons. Let’s focus on one:
We can see, at the center of the osteon, the Haversian canal, with blood flow in the middle. It is lined by a layer of osteoprogenitor cells.
Since we’re dealing with an already formed bone, we can’t see the osteoprogenitor layer become osteoblasts, which would secrete the ECM in a circularly organized manner.
As we can see here. Counting the lamellae will give us an approximate age of the bone, like a tree.
What about all the vertical filaments inside the circles? Those are all the projections that exit from the osteocytes. These projections are all forming channels connecting the osteocytes in the same lamellae.
Which is the oldest lamellae? Obv the outer one is the oldest since it was the first one to be pushed out be the newer layers.
Are all these osteons separated units?
No they’re all connected by the Wolkmann’s canals.
Such as this one. They’re not always visible, as it depends on how the cut was made.
What about the darker parts, they’re interstitial lamellae that haven’t been rearranged by the osteon yet. ??
What are the four patterns of lamellar bone?
- Osteons (Haversian systems)
- Interstitial lamellae
- Outer circumferential lamellae
- Inner circumferential lamellae
Vascular Channels in Compact Bone
| Canal | Orientation | Contents |
|---|---|---|
| Haversian canal | Longitudinal (parallel to bone shaft) | Capillaries and postcapillary venules |
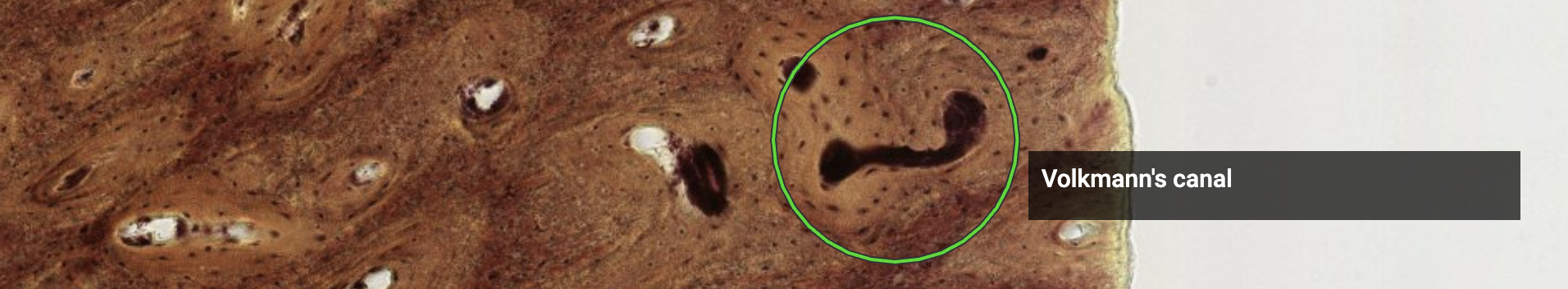
| Volkmann’s canal | Transverse/oblique | Blood vessels from periosteum and bone marrow; connect haversian canals |
Anki cloze
{1:Volkmann’s canals} run transversely/obliquely and connect haversian canals with one another.
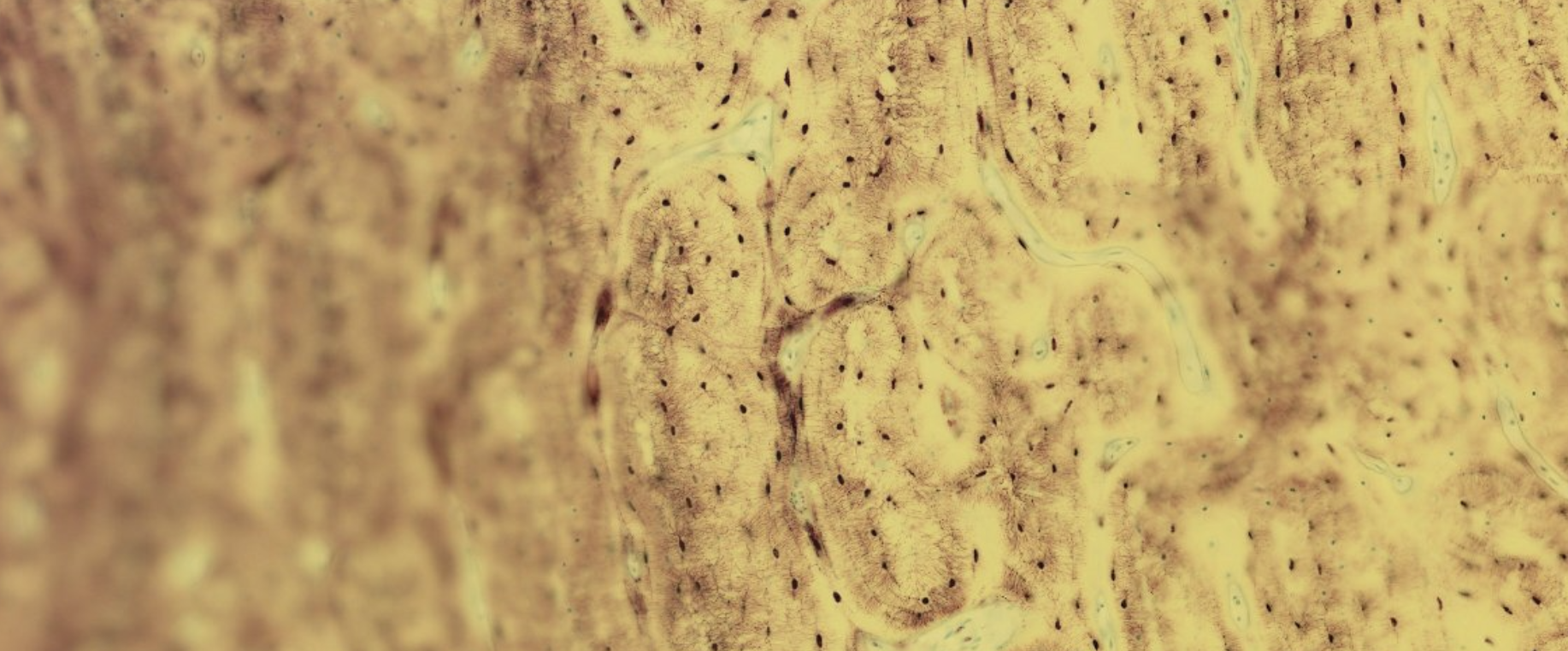
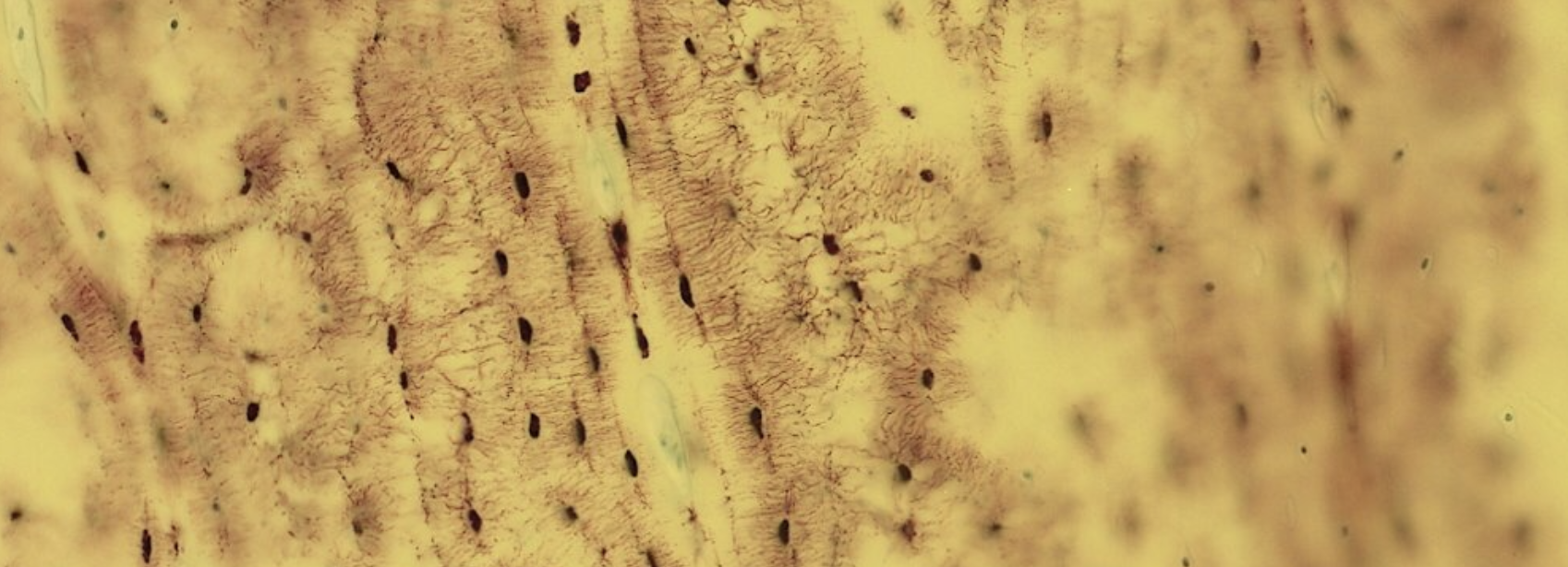
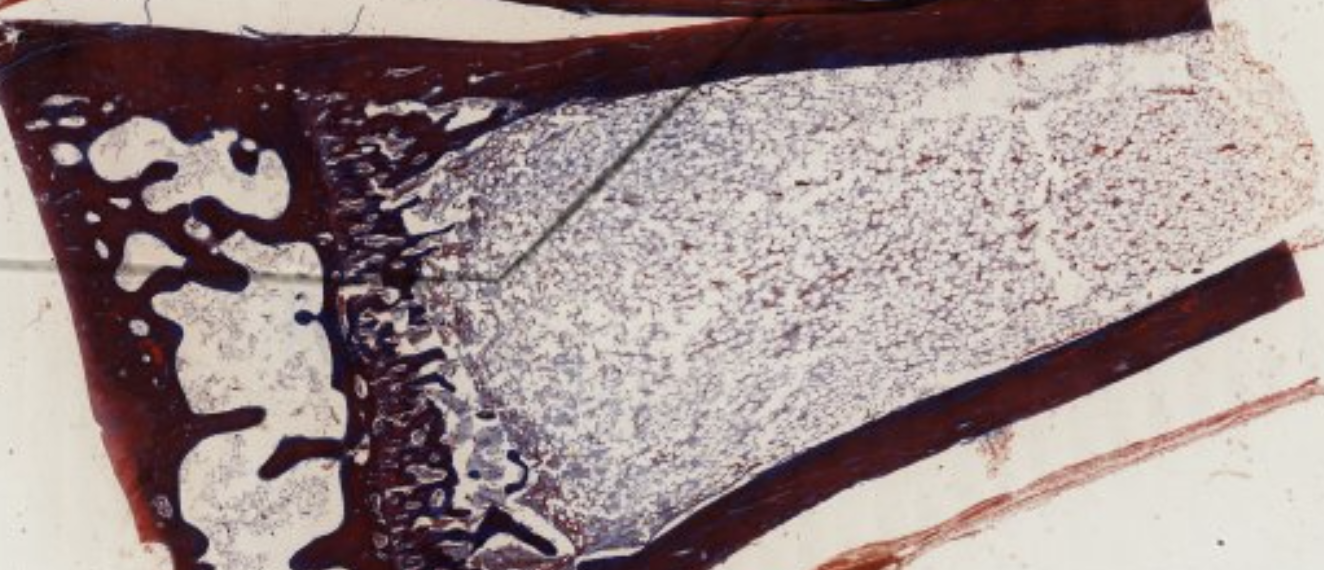
Classifying... https://unibo.smartzoom.com/s1241/course1776/f1841/i7755/
Often, some parts are completely out of focus. That is because it’s extremely hard to cut a thin slice of the sample. This is an indicator that you’re looking at a thick compact bone.
Here you can clearly see the think oprojectors of the osteocytes from adjacent lamellae.
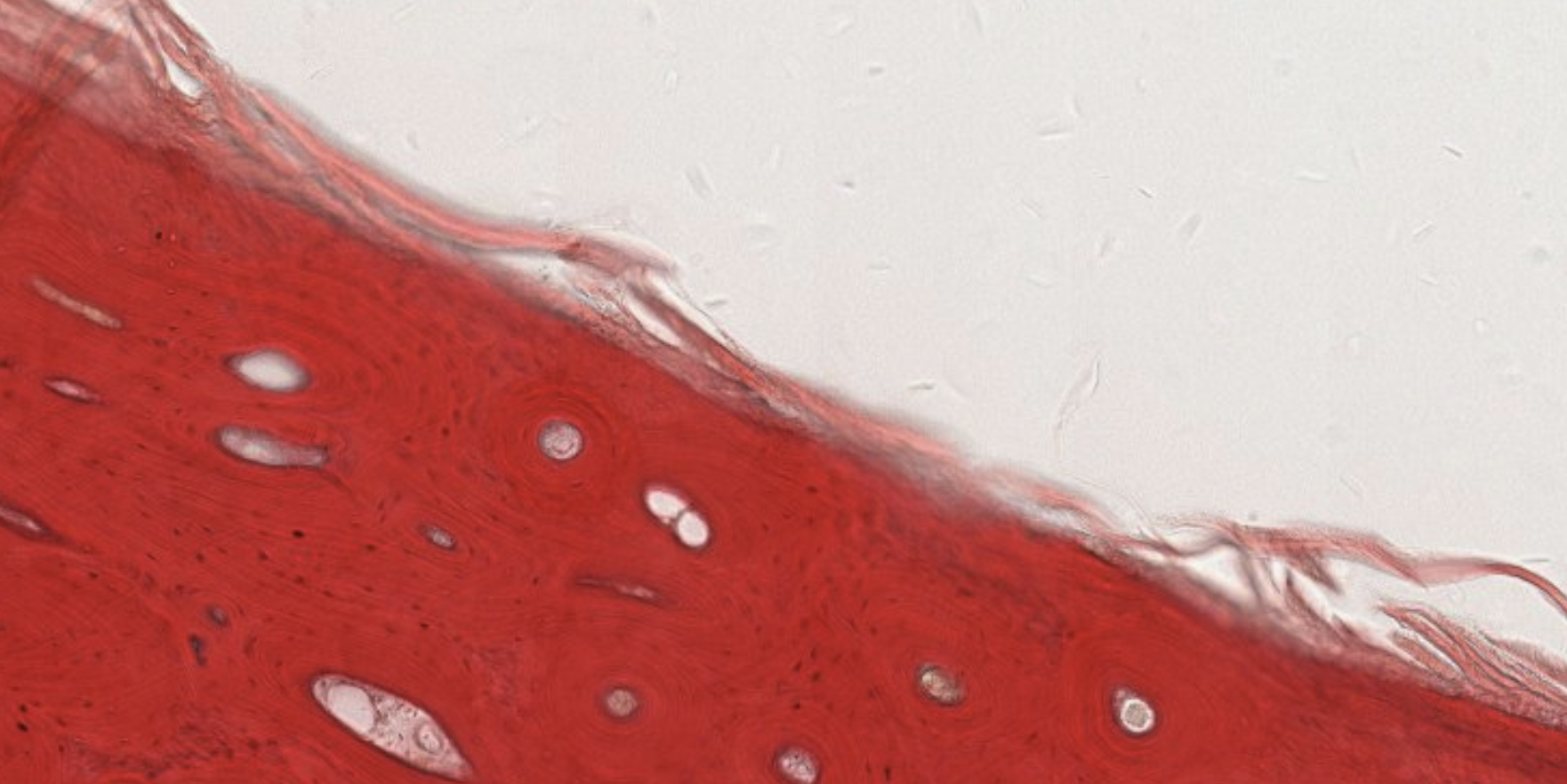
Periosteum and Endosteum — Detailed
Periosteum
- Inner layer:
- osteoprogenitor cells in direct contact with bone. In adults → periosteal stem cells (PSCs) with clonal multipotency and self-renewal; give rise to bone-forming osteoblasts in response to injury
- Outer layer:
- rich in blood vessels (some entering Volkmann’s canals) and thick anchoring collagen fibers called Sharpey’s fibers, which penetrate the outer circumferential lamellae (to the surface?)
Endosteum
- Covers spongy bone walls; extends into all bone cavities (haversian and Volkmann’s canals)
- Consists of: osteoprogenitor cells, reticular stromal cells of bone marrow, connective tissue fibers
- Preosteoblasts and osteoblasts in the endosteum contribute hematopoietic cytokines to the endosteal niche → essential for hematopoietic stem cell (HSC) proliferation and maturation
Endosteal Niche
The endosteal niche provides the microenvironment essential for hematopoietic stem cell proliferation and maturation through cytokines secreted by preosteoblasts and osteoblasts.
What are Sharpey's fibers?
Collagen fibers derived from the outer layer of the periosteum that project into and anchor into the outer circumferential lamellar system of bone.
Osteogenesis (Bone Development / Ossification)
Two Routes to Bone Formation
Bone can develop via:
- Intramembranous ossification — directly from mesenchymal cell condensation (no cartilage template)
- Endochondral ossification — by gradual replacement of a pre-existing cartilage template
Common Initial Mechanism
Regardless of type, bone formation begins with laying down a primary spongiosa (initial trabecular network), which is then transformed into mature bone.
What are the two types of ossification?
Intramembranous ossification (direct from mesenchyme) and endochondral ossification (replacement of a cartilage template)
Intramembranous Ossification
Bones Formed This Way
Frontal, parietal, parts of occipital, temporal, mandible, maxilla bones, and the clavicle
Sequence
- Embryonic mesenchyme becomes highly vascularized; mesenchymal stem cells aggregate within ECM (collagen + proteoglycans)
- Process is controlled by patterning signals from: Wnt, Hedgehog, FGF, TGF-β family polypeptides
- Aggregated mesenchymal stem cells directly differentiate into osteoblasts (no cartilage intermediate) that secrete osteoid (unmineralized organic bone matrix)
- A bone blastema is formed; osteocytes within the core interconnect via cell processes forming a functional syncytium
- Osteoblasts line the surface of the blastema
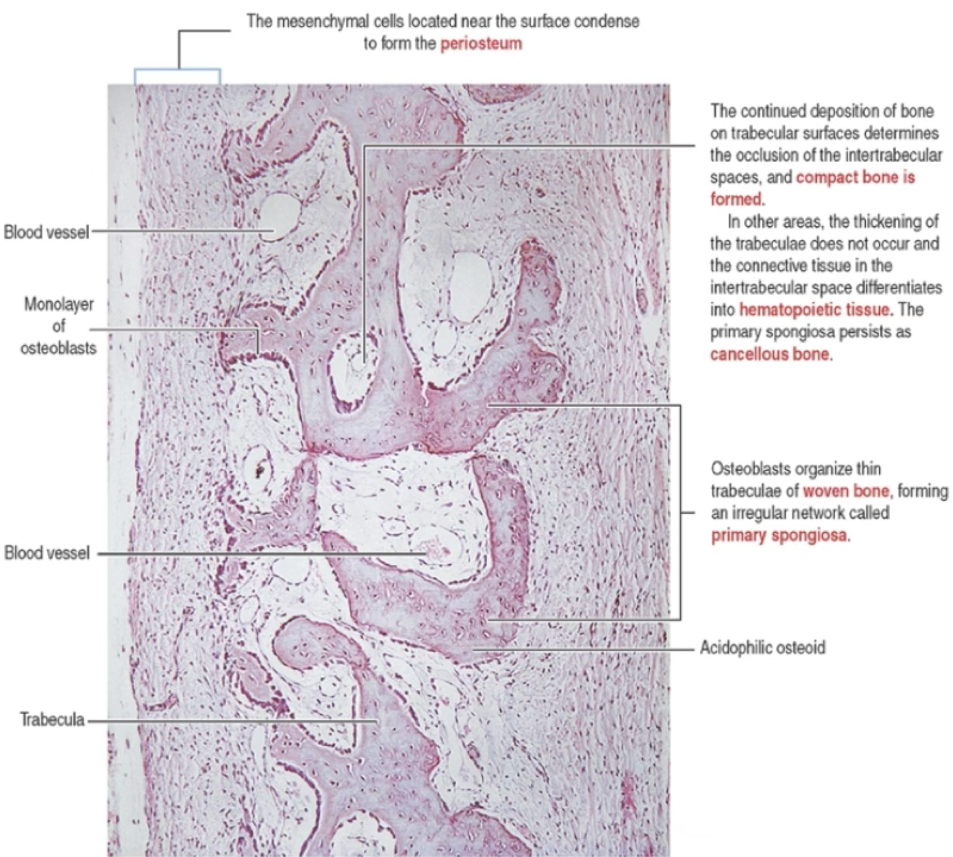
- Multiple osteogenesis centers develop → fuse → form primary spongiosa (anastomosing trabeculae = spongy bone)
- Collagen fibers are randomly oriented → early bone = woven bone
- (transported by blood vessels) deposits in osteoid → mineralization (calcification)
- Osteoid is laid down by apposition (no interstitial growth)
- Osteoblasts become trapped → osteocytes; perivascular channels assume hematopoietic role as mesenchymal stem cells convert to blood-forming cells; osteoclasts initiate modeling
- Mesenchymal cells near surface condense → form periosteum
- Continued appositional deposition on trabecular surfaces → closes intertrabecular spaces → compact bone forms
- Alternatively, connective tissue in intertrabecular space differentiates into hematopoietic tissue → cancellous bone
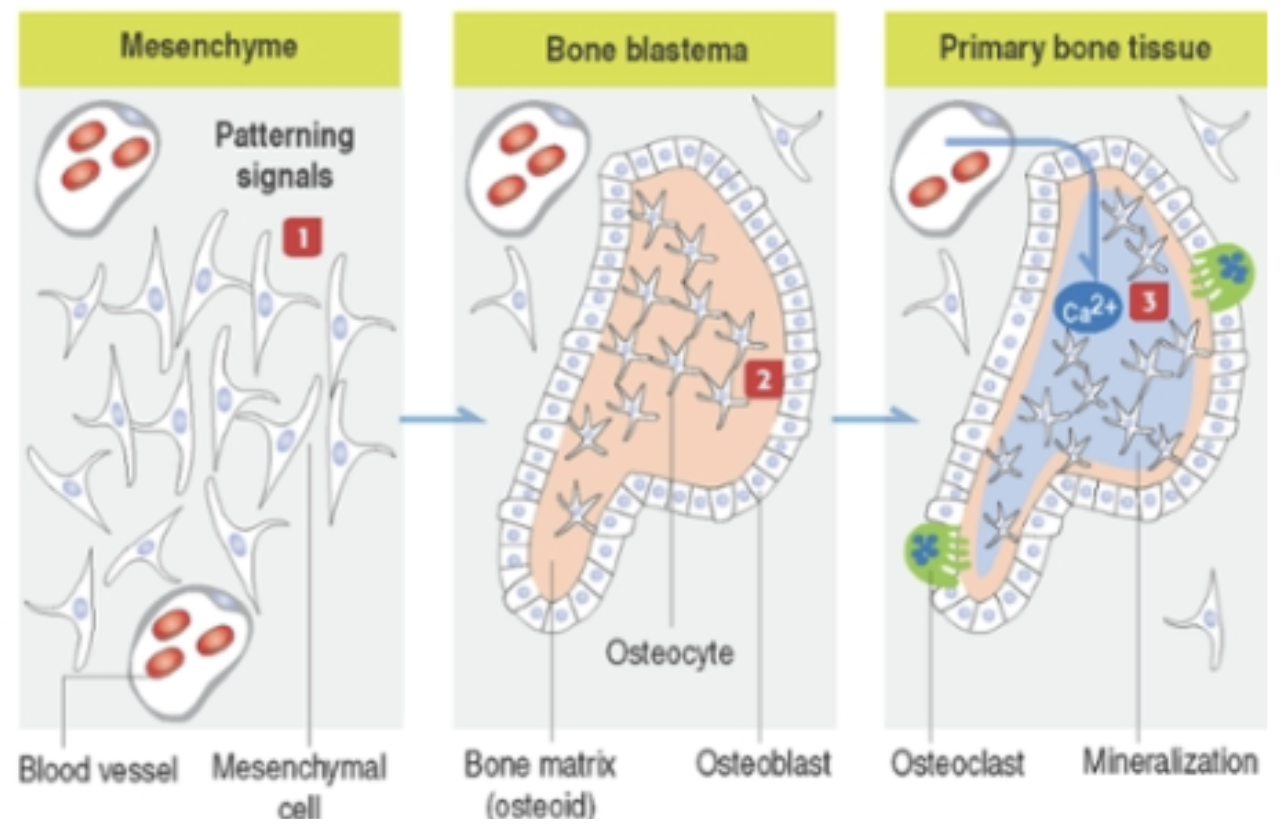
Organization of a primary center of ossification:
Anki cloze
In intramembranous ossification, mesenchymal stem cells differentiate {1:directly into osteoblasts} without forming a cartilage intermediate.
What is osteoid?
Osteoid is the unmineralized, organic portion of the bone matrix secreted by osteoblasts before mineralization occurs.
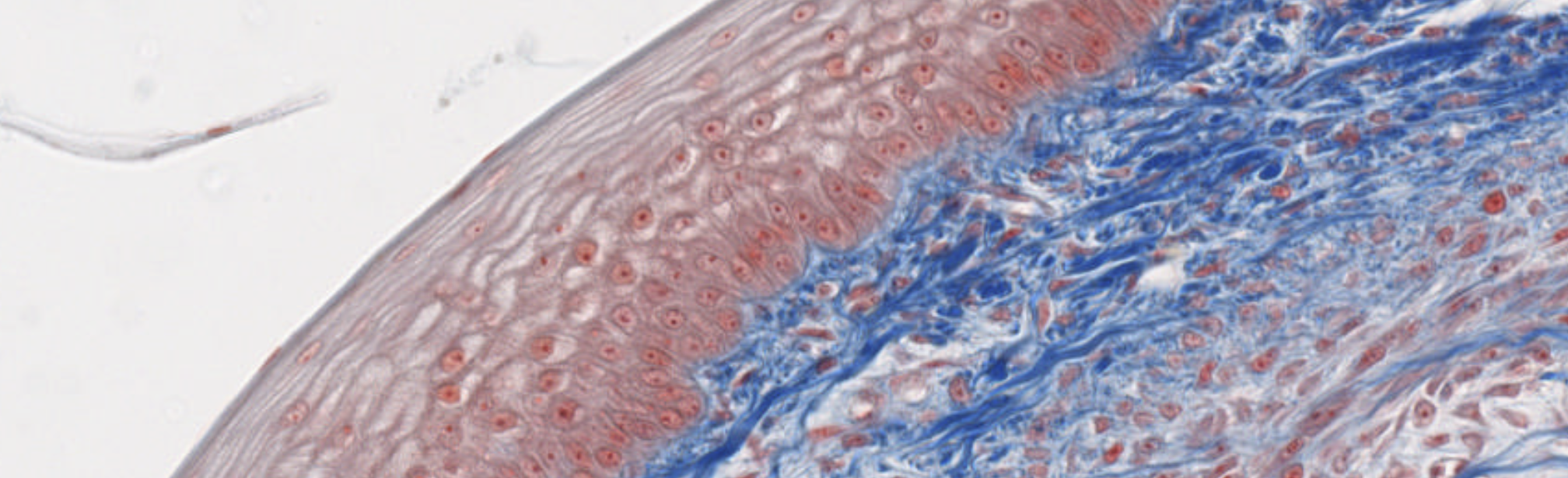
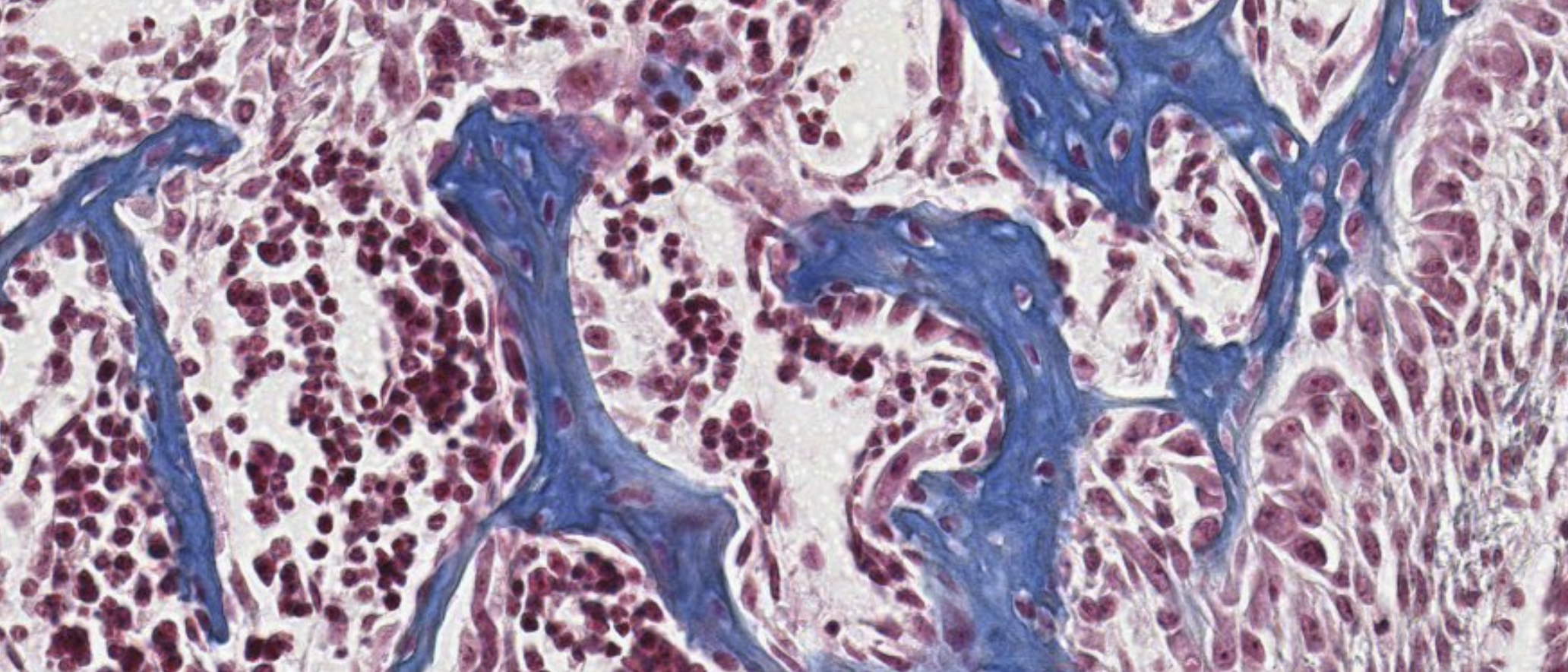
Classifying... https://unibo.smartzoom.com/s1241/course1776/f1841/i7758/
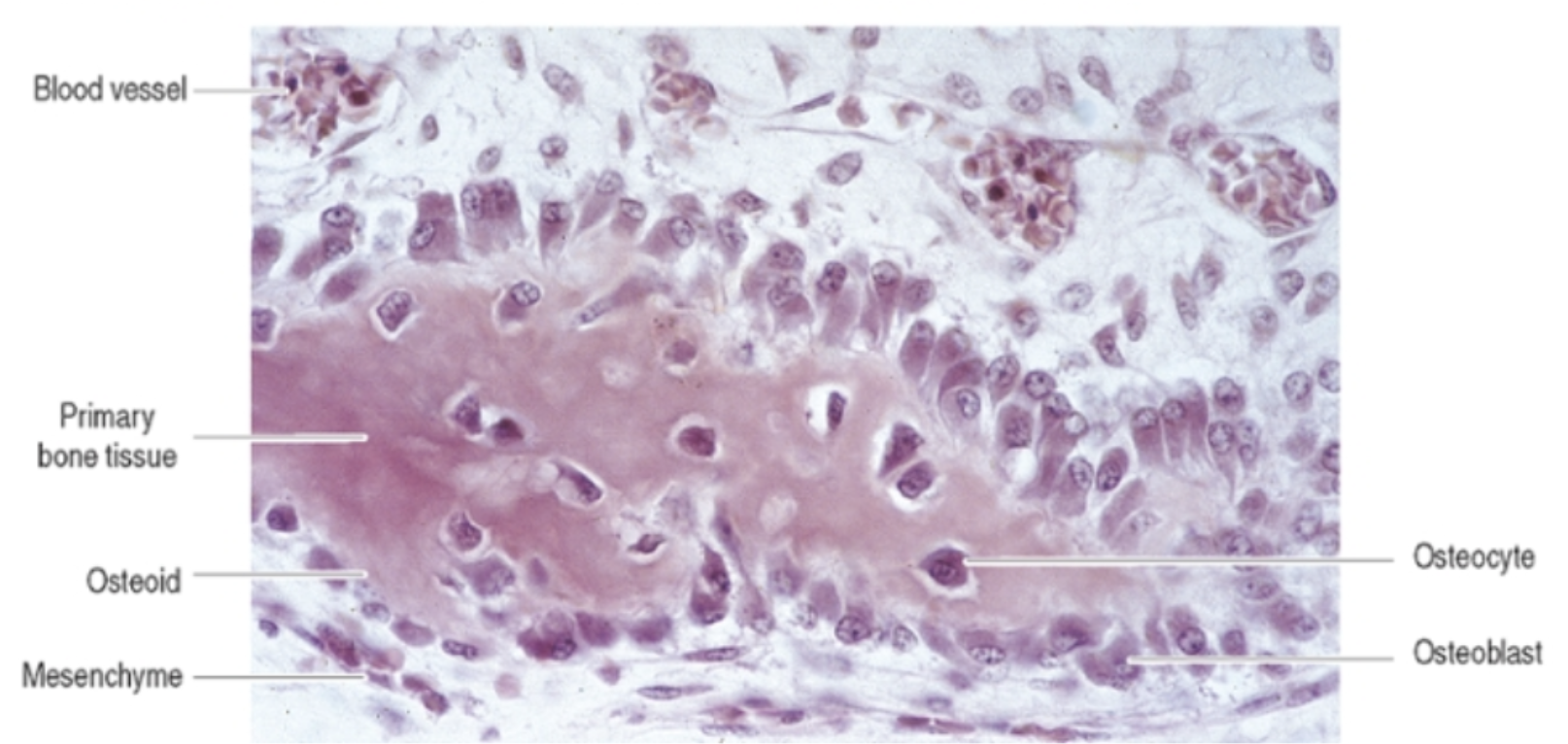
Stratified squamous? and loose connective tissue…
Compound glandular exocrine gland. Tubulo-alveolar, nuclei squished at the periphery, so mucus secretion.
Where that weird part is → Serous secretion.
On the surface of the blue structure → Layer of the osteoblasts. Above it? Progenitors… (?).
The osteoblasts are surrounding the newly formed bones, secreting the matrix.
Here, when they will complete that secretion, they will become surrounded by their own newly produced matrix.
These are trapped osteocytes, once were osteoblasts.
Eventually, all these blue structure will be interconnected, forming the structure of the spongy bone.
What are these? Mesenchymal cells. We’re inside the embryo (?).
Classifying... https://unibo.smartzoom.com/s1241/course1776/f1841/i7756/
Vabbe you can see the osteoblasts… The trabecolae already fused…
We can see the osteoblasts sitting on top of the newly found bone. They are secreting!!
After being surrounded, they can’t reach the nutrients found in the superficie so lacking them, they’re lead to differentiation.
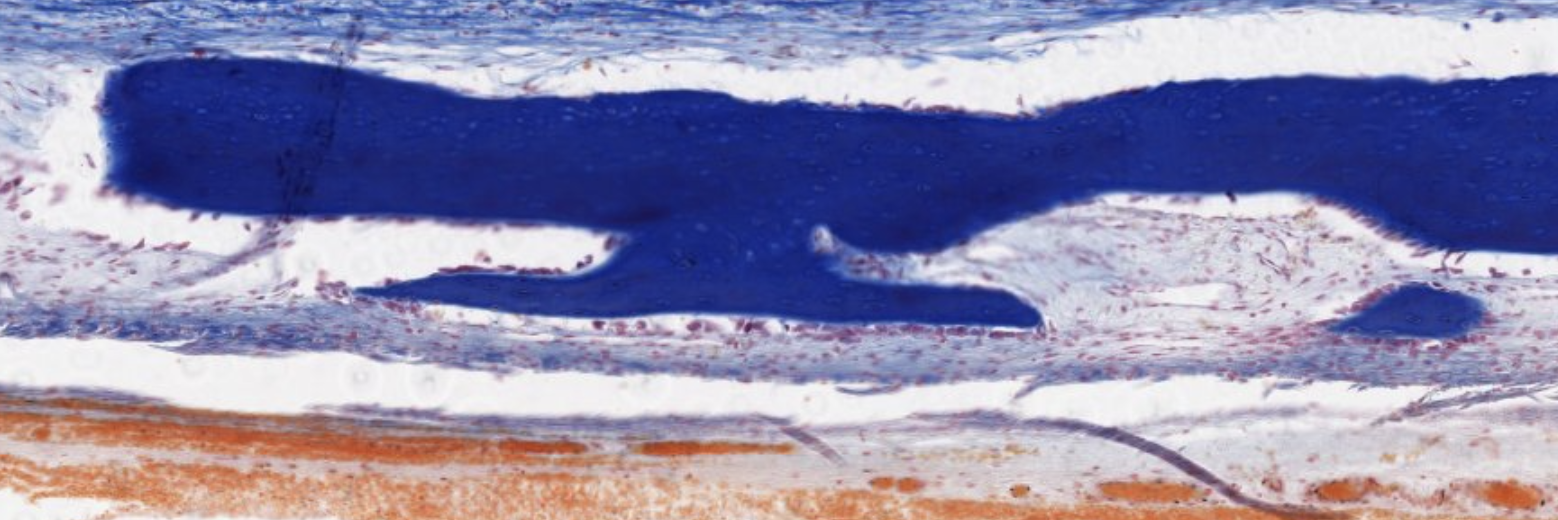
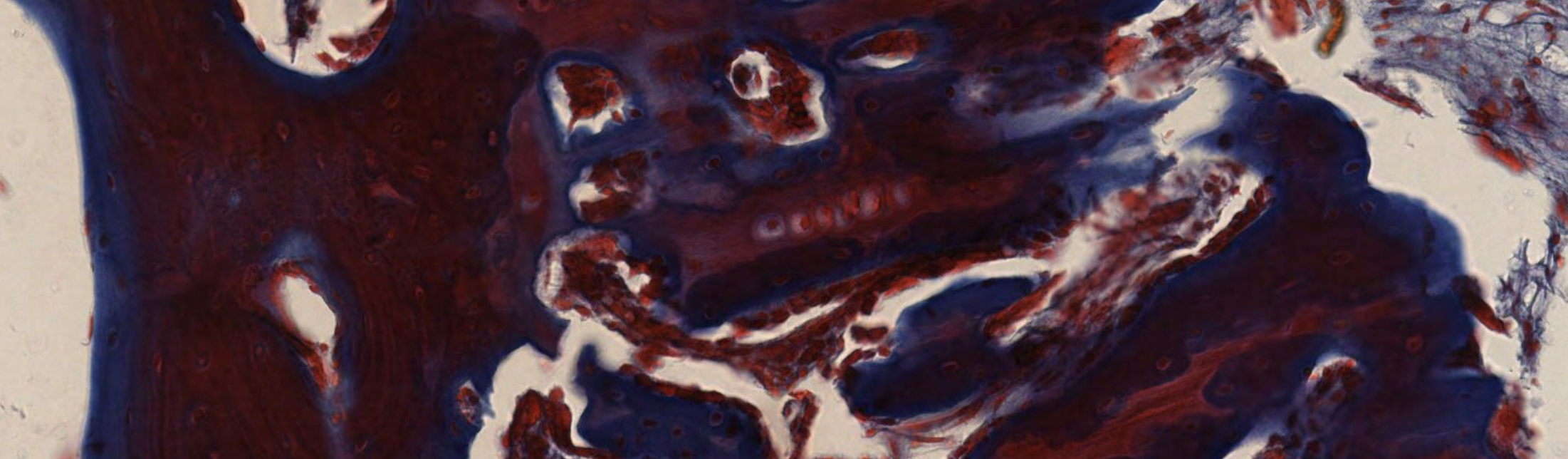
Classifying... https://unibo.smartzoom.com/s1241/course1776/f1841/i1846/
Clearly identified by the central canal… surrounding, progenitor cells forming the endostium.
We can see the concentric structure called lamellae, produced by the osteoblasts producing the osteoid (inorganic ECM), once the osteoid is produced, it calcifies with the osteoblast inside, where it will differentiate into an osteocyte.
Periosteum, other precursor cells located here…
You can’t always see a Wolkmann’s canal in a transversal section, but here it is: what keeps all the different lamellae connected.
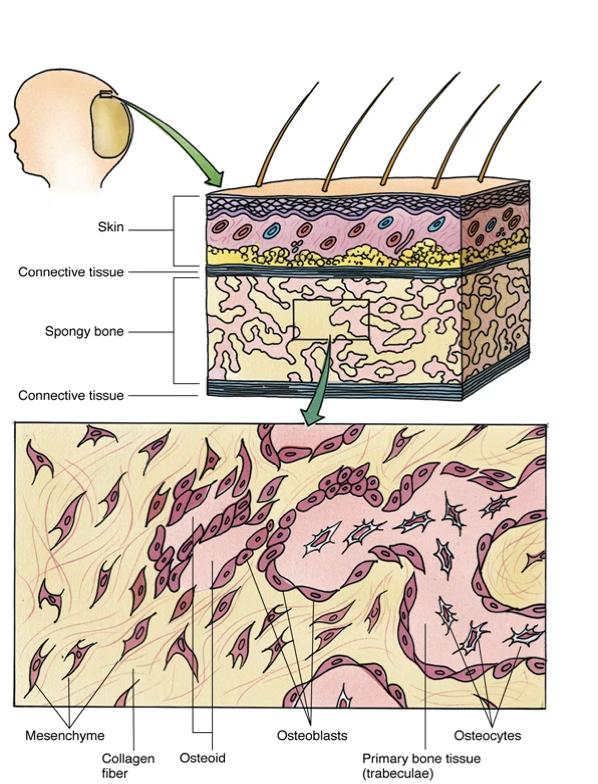
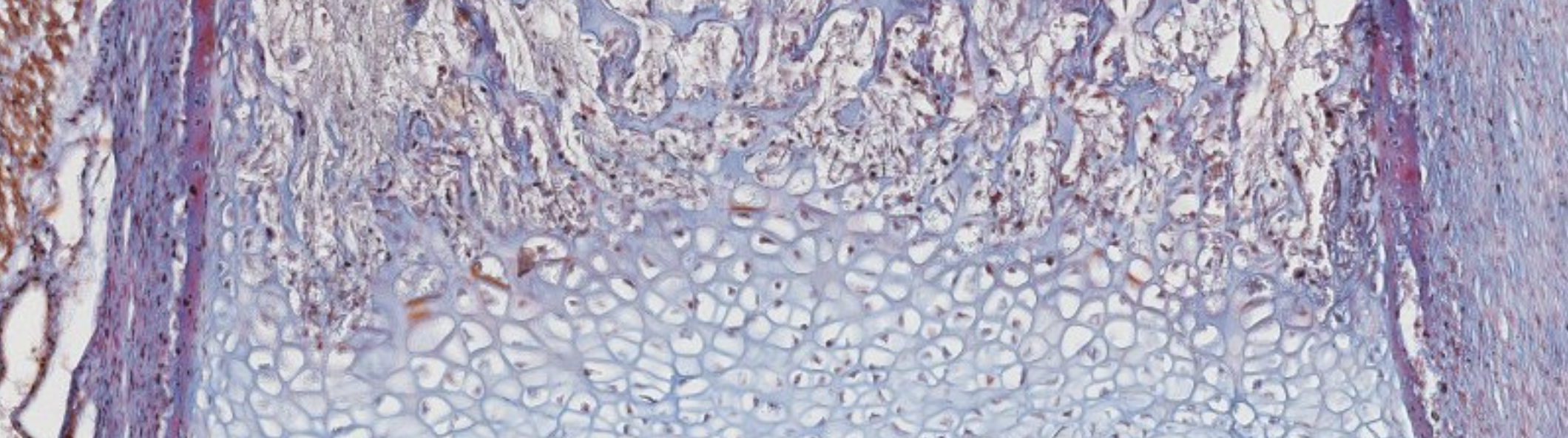
Endochondral Ossification
Bones Formed This Way
Most long and short bones of the body
Overview
Bone forms inside a pre-existing hyaline cartilage template that acts as a scaffold.
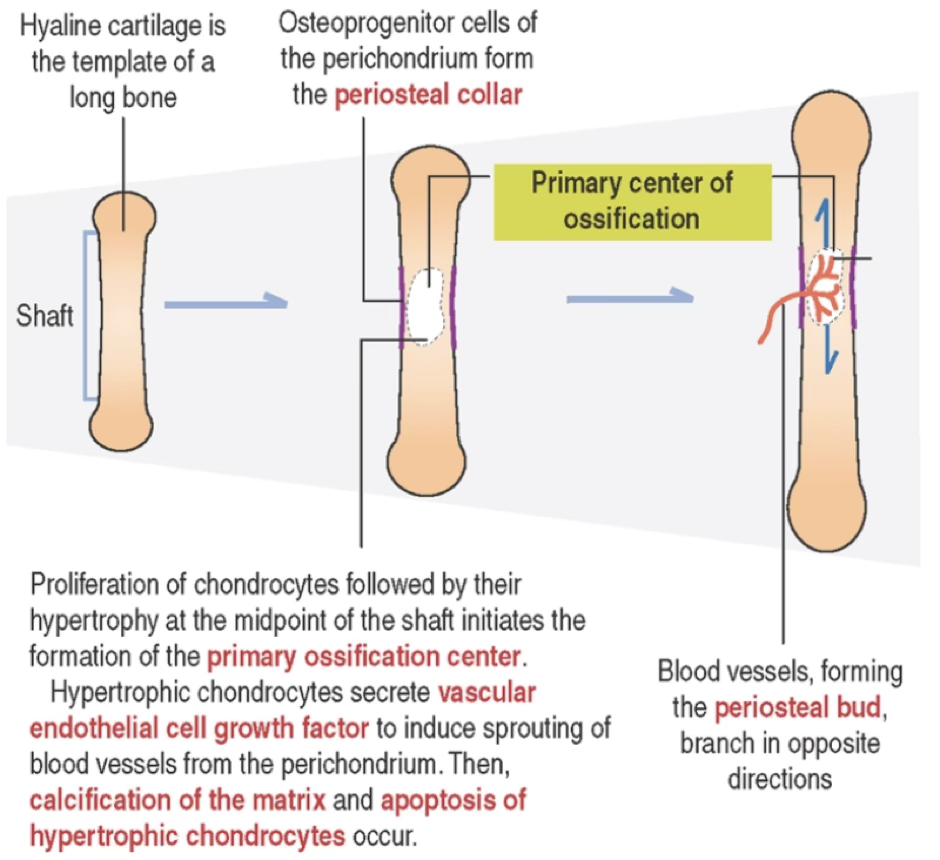
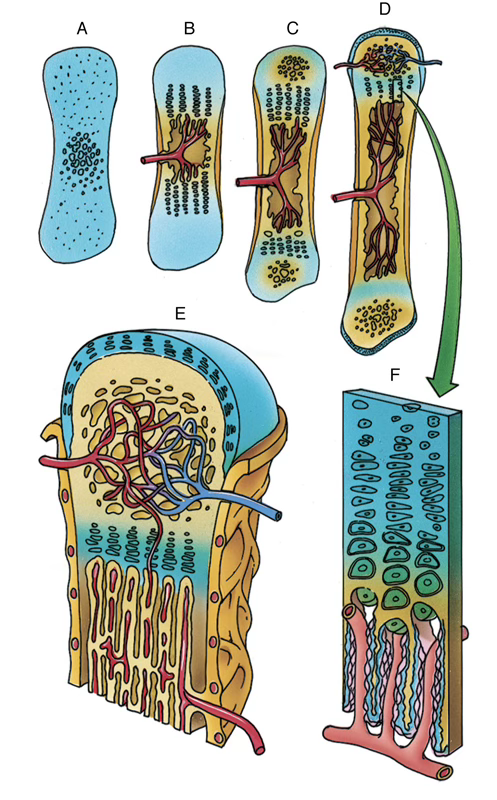
Sequence
- A hyaline cartilage model of the future bone develops and grows both appositionally and interstitially
- Chondrocytes in the center of the diaphysis undergo hypertrophy; lacunae enlarge; intervening cartilage matrix septa become calcified
- Perichondrium at the diaphyseal midriff becomes vascularized → chondrogenic cells become osteoprogenitor cells→ differentiate into osteoblasts; overlying perichondrium → periosteum
- Hypertrophic chondrocytes secrete VEGF → induce vascular sprouting from perichondrium
- New osteoblasts secrete bone matrix → form subperiosteal bone collar (by intramembranous bone formation) on the surface of the cartilage template
- Hypertrophied chondrocytes die → empty confluent lacunae → future marrow cavity
- Osteoclasts etch holes in bone collar → periosteal bud (osteogenic bud) enters cartilage concavities
- Osteoprogenitor cells divide → osteoblasts deposit bone matrix on calcified cartilage → calcified cartilage/calcified bone complex
- Osteoclasts resorb this complex, enlarging the marrow cavity
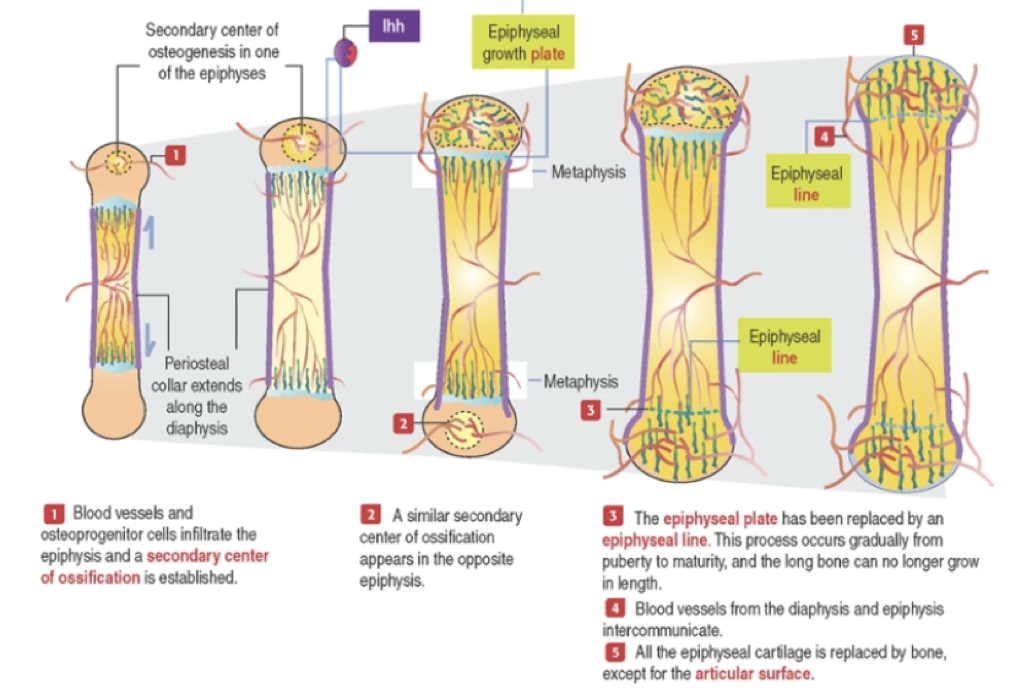
Primary vs. Secondary Centers of Ossification
| Feature | Primary Center | Secondary Centers |
|---|---|---|
| Location | Diaphysis (mid-shaft) | Epiphyses |
| Timing | ~3rd month of fetal life | After birth |
| Result | Marrow cavity + bone collar | Spongy bone replaces most epiphyseal cartilage |
| Cartilage retained | Articular cartilage + epiphyseal growth plate | Articular cartilage |
What triggers vascular invasion of the cartilage model during endochondral ossification?
Hypertrophic chondrocytes secrete VEGF (vascular endothelial cell growth factor), inducing sprouting of blood vessels from the perichondrium.
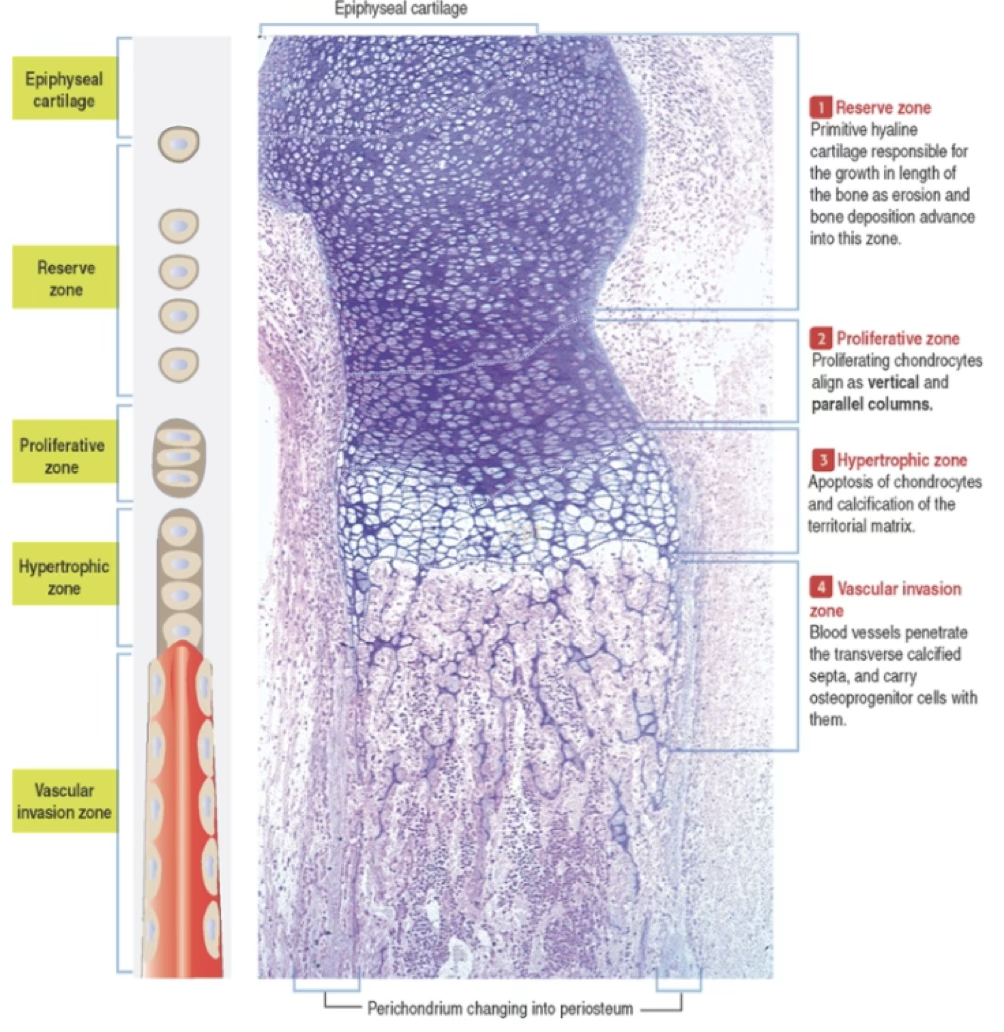
Epiphyseal Growth Plate Zones
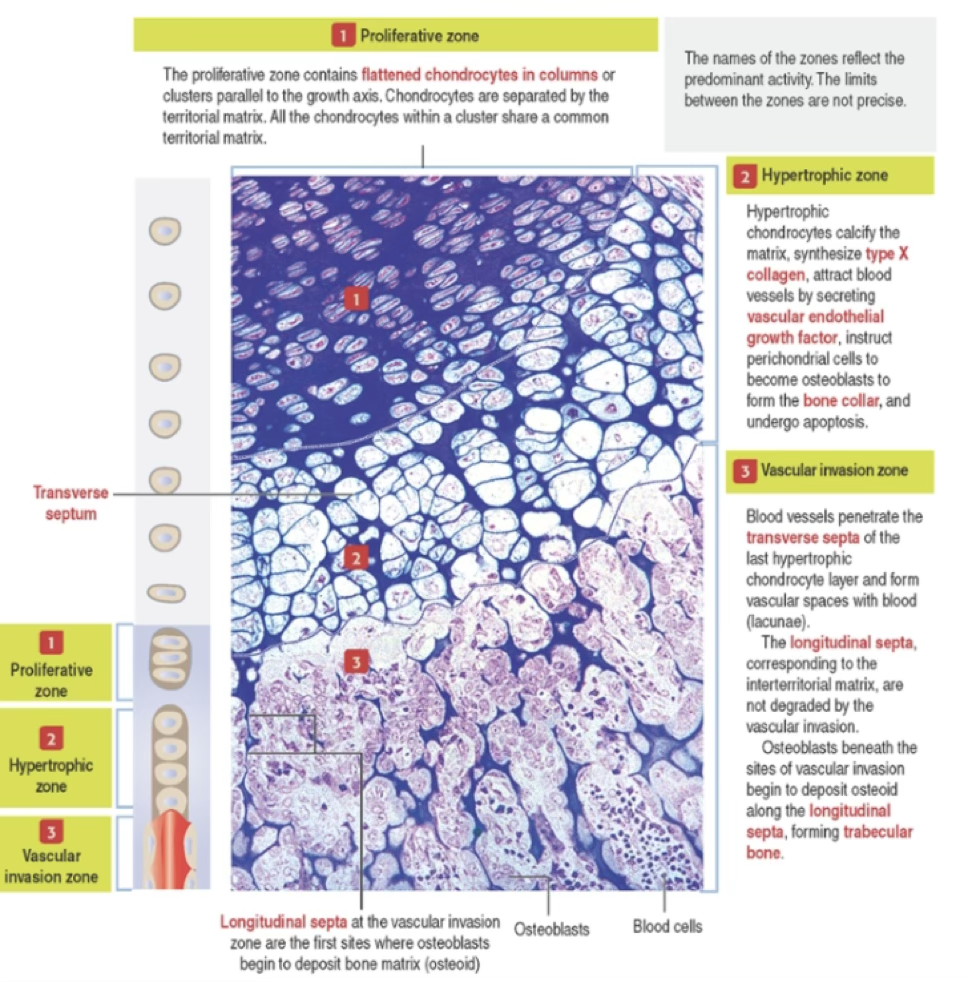
| Zone | Activity |
|---|---|
| Proliferative zone | Flattened chondrocytes in columns parallel to growth axis; separated by territorial matrix; active mitosis |
| Hypertrophic zone | Chondrocytes enlarge; calcify matrix; synthesize type X collagen; secrete VEGF; instruct perichondrial cells to become osteoblasts; undergo apoptosis |
| Vascular invasion zone | Blood vessels penetrate transverse septa; osteoblasts deposit osteoid on longitudinal septa → trabecular bone |
Important
The epiphyseal growth plate is responsible for growth in length of bone.
Gradual replacement from puberty to maturity → epiphyseal line → long bone can no longer grow in length.
Anki cloze
Hypertrophic chondrocytes in the growth plate synthesize {1:type X collagen} and secrete {2:VEGF} to attract blood vessels.
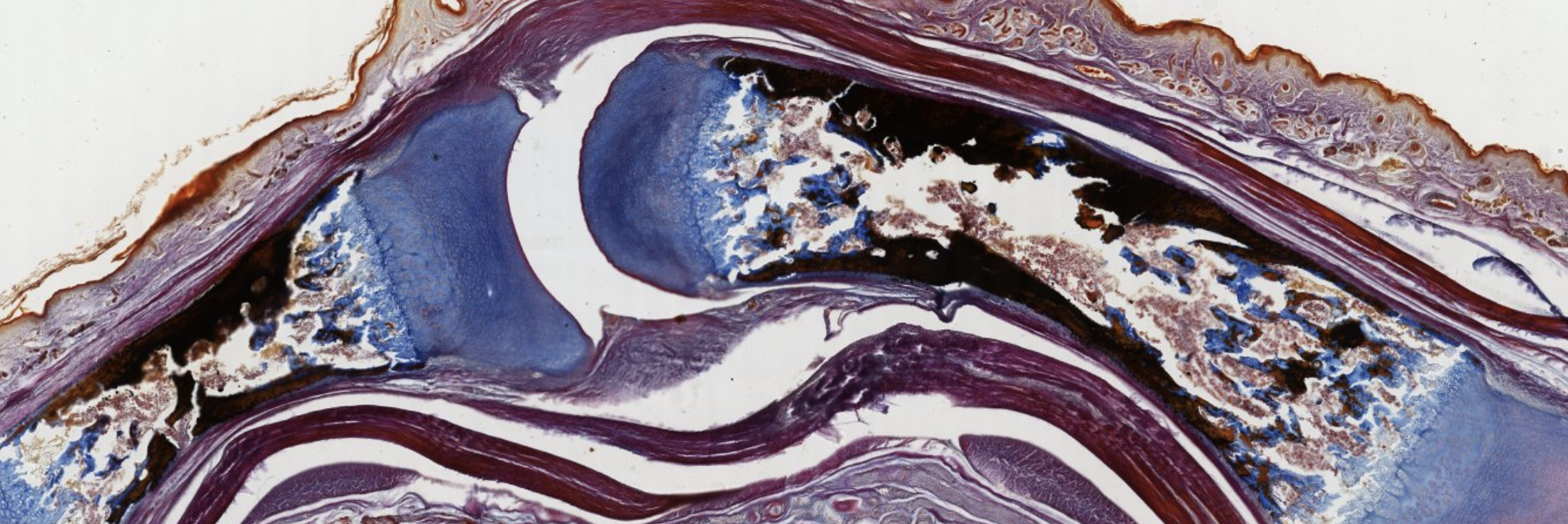
Classifying... https://unibo.smartzoom.com/s1241/course1776/f1841/i1852/
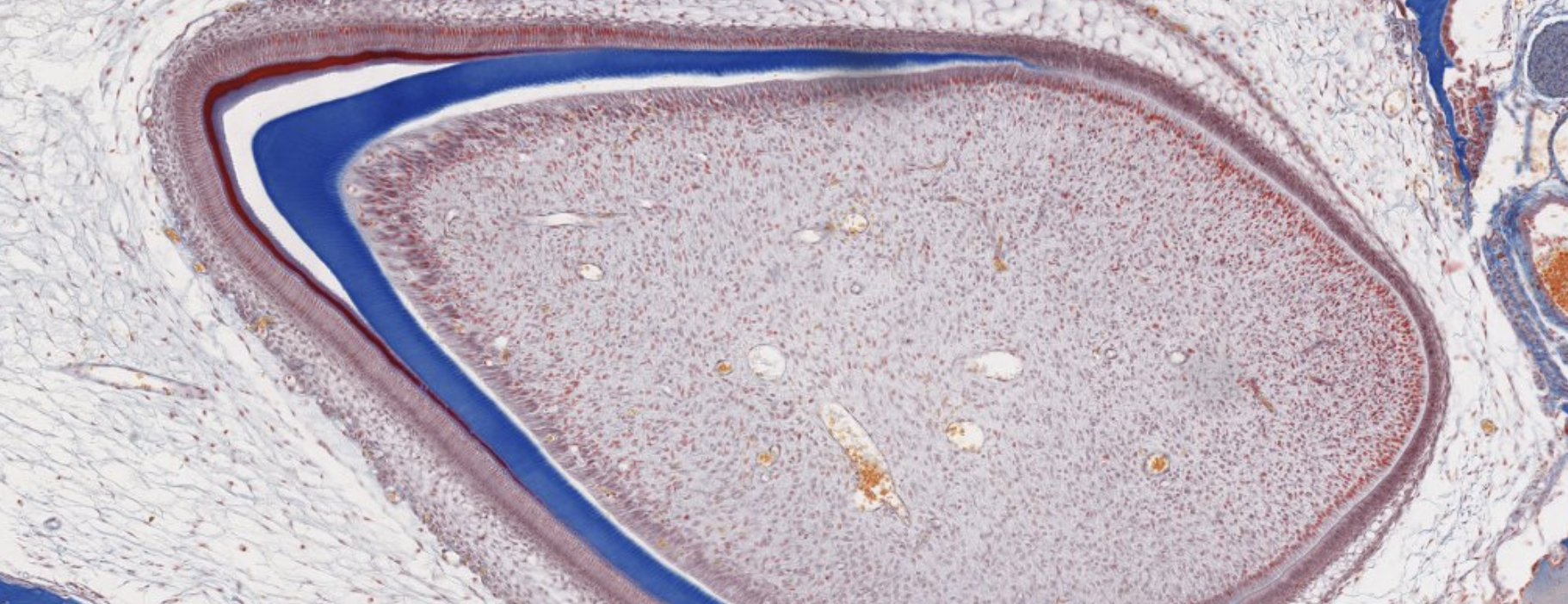
Primary center of ossification at the level of the diaphysis, where the cartilage is being replaced.
You can still see leftover mineralized cartilage.
Here are a lot of osteoblasts.
Mesenchymal and blood cells → will become bone marrow.
Here are cartilage progenitor cells… they will go into columnar formation and differentiate into chondroblasts. Hyperthropy → last stage before cell death.
Here we see chondroblasts that are becoming hyperthropic then bam dying. Down → Up.
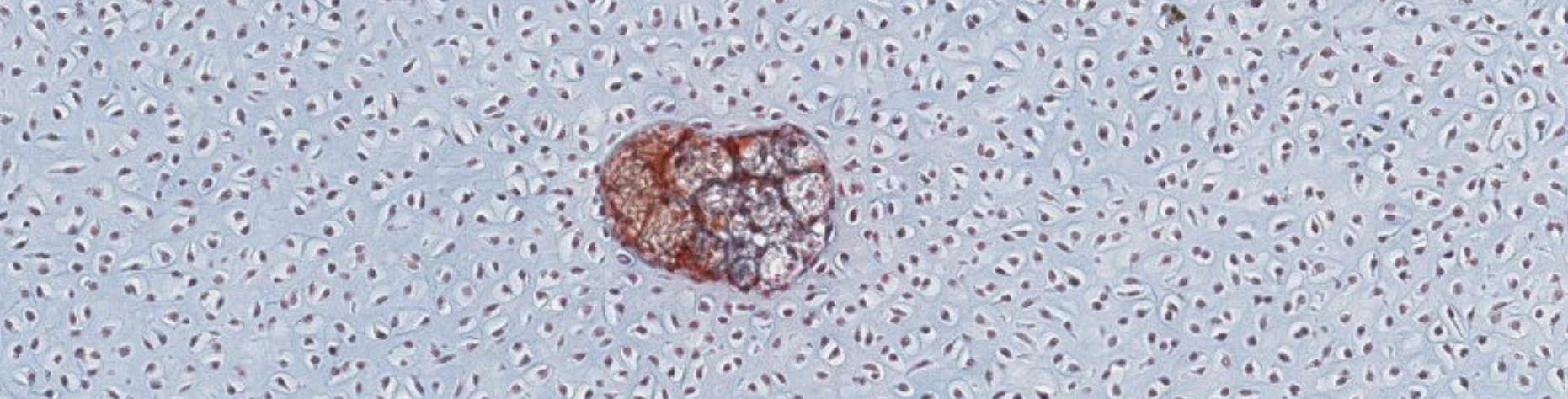
Hey look! This is the very start of a secondary center of ossification.
Very early stage of embryo development.
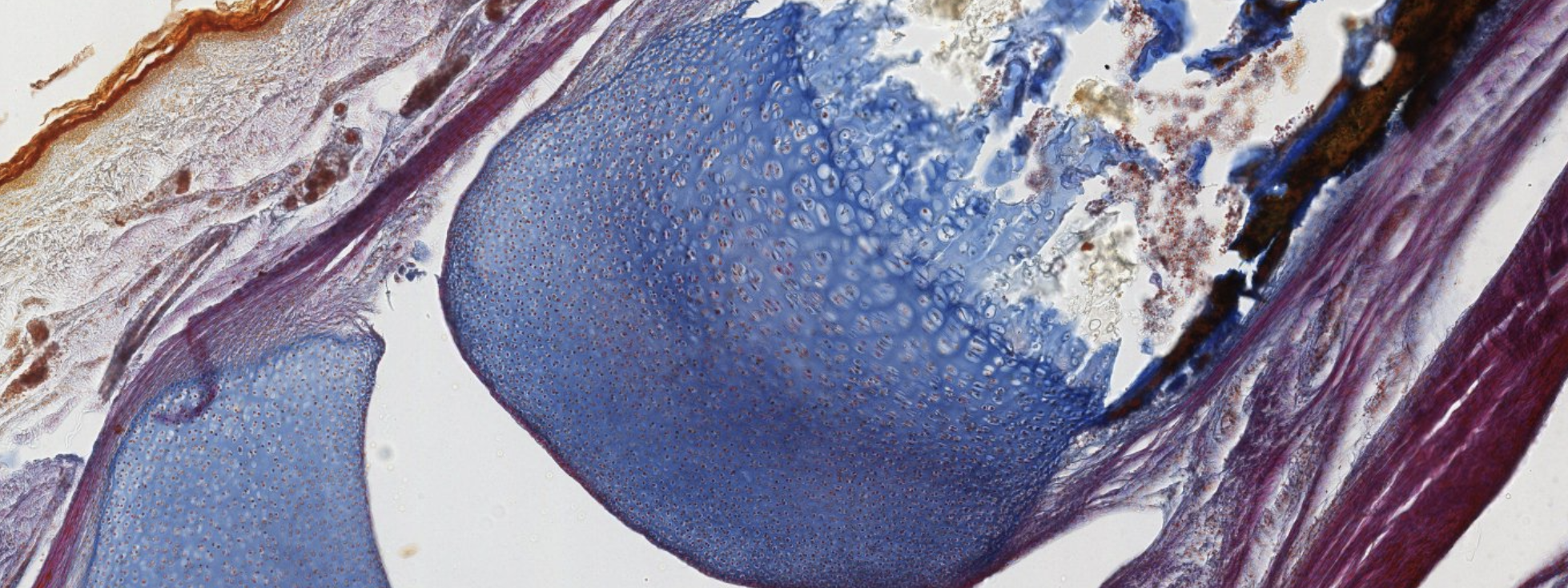
Classifying... https://unibo.smartzoom.com/s1241/course1776/f1841/i1853/
This is advanced. Most of the cartilage has already been replaced.
Dark stain? Mineralization already happened/happening there.
Elongation of the bone! Allowed by the proliferation of the osteoblasts on the proliferative zone of the cartilage. What do we see at the start of that elongation (closer to the bone): hypertrophic osteoblasts. Ready to die and become bone!!
This looks like a secondary center of ossification… At the epiphysis, we should see spongy bone!
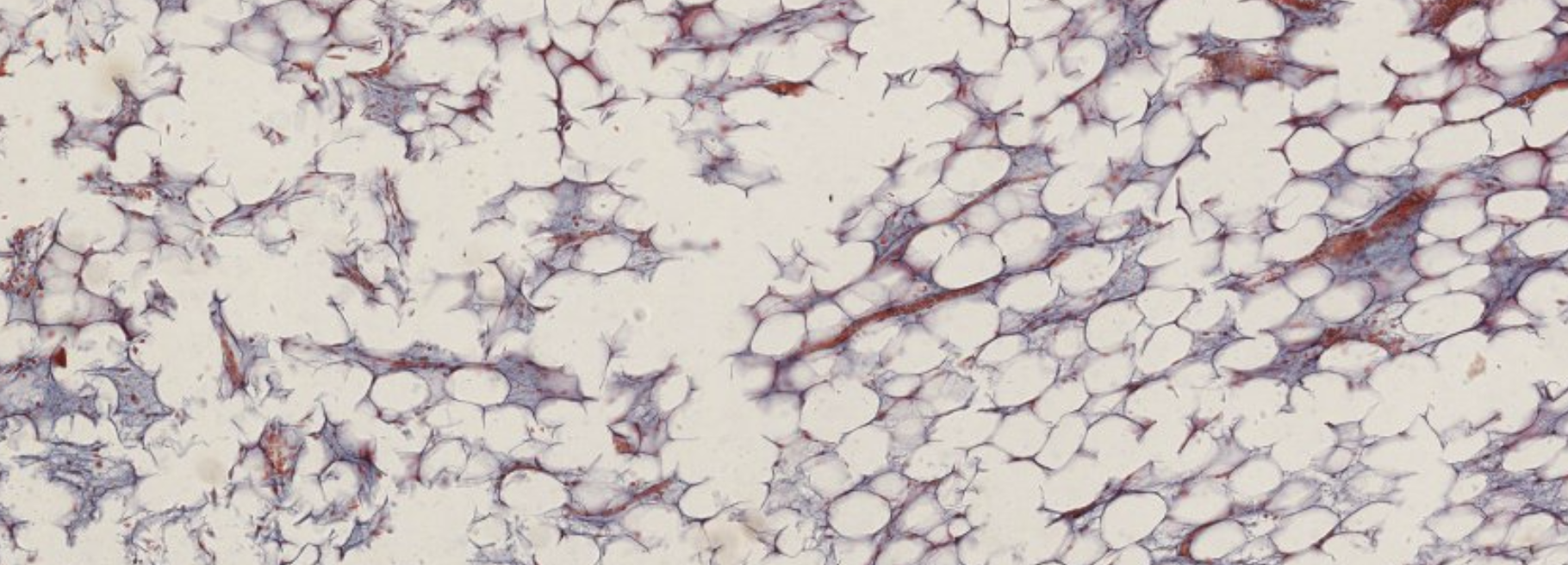
Classifying... https://unibo.smartzoom.com/s1241/course1776/f1841/i1854/
You cana lready identify the bone marrow cavity. At this higly advanced level you’re already suppoused to see the osteon.
Here’s some cartilage, only on the joint side, where the articulation will occurr. This cartilage is not going to help with the ossification.
Osteoblasts + hypertrophic chondocytes?? qualcosa del genere
Ma cosa ci fai qua white adipose tissue?? This is the yellow bone marrow, in the cavity of a long bone.
Conversion of Trabecular Bone into Osteons
Spicule → Trabecula → Osteon
- Spicule: longitudinal core of calcified cartilage coated by osteoid produced by osteoblasts; blood vessels in contact with it
- Trabecula: lacks calcified cartilage core; contains an osteocyte lamellar core lined by osteoblasts depositing osteoid
- Osteon: bone cylinder with central longitudinal tunnel housing a blood vessel
Sequence: Trabecula → Osteon
- Longitudinal edges of a trabecula form the boundary of a groove containing a blood vessel; ridges and grooves lined by osteoblasts
- Ridges grow toward one another → groove → tunnel; blood vessel becomes trapped → longitudinal axis of the new osteon
- Blood vessels interconnect with adjacent tunnels via Volkmann’s canals
- Osteoblasts lining the tunnel wall deposit concentric lamellae by apposition → osteon formed
- Appositional growth adds lamellae under periosteum; inner circumferential lamellae develop from endosteal osteoblasts
- Interstitial lamellae fill spaces between osteons; outer circumferential lamellae form the boundary
What is the difference between a spicule and a trabecula?
A spicule has a longitudinal core of calcified cartilage coated by osteoid. A trabecula lacks the calcified cartilage core and instead has an osteocyte lamellar core lined by osteoblasts.
Bone Remodeling
Cortical (Haversian) Remodeling
- Osteoclast precursors recruited to haversian canal → differentiate into osteoclasts
- Osteoclasts line the bone lamella facing the canal → resorb inner lamella → progress outward, creating a Howship’s lacuna; interstitial lamellae = residuals of remodeled osteon
- Osteoclasts stop → osteoclast-to-osteoblast reversal
- Osteoblasts line the resorption cavity and start secreting osteoid; cement line marks the boundary of the newly organized lamella
- New bone lamellae deposited toward the center of the osteon
- Osteoblasts become trapped → osteocytes
- A new osteon (haversian system) is formed; interstitial lamellae are left behind
Trabecular Bone Remodeling
Occurs on the bone surface (vs. cortical remodeling which occurs within an osteon)
- Osteoclasts create a resorption space limited by a cement line
- Osteoblasts line the cement line surface → deposit osteoid → new bone closes the resorption space
Anki cloze
Trabecular bone remodeling occurs on the {1:bone surface}, whereas cortical bone remodeling occurs {2:within an osteon}.
Bone Fracture and Repair
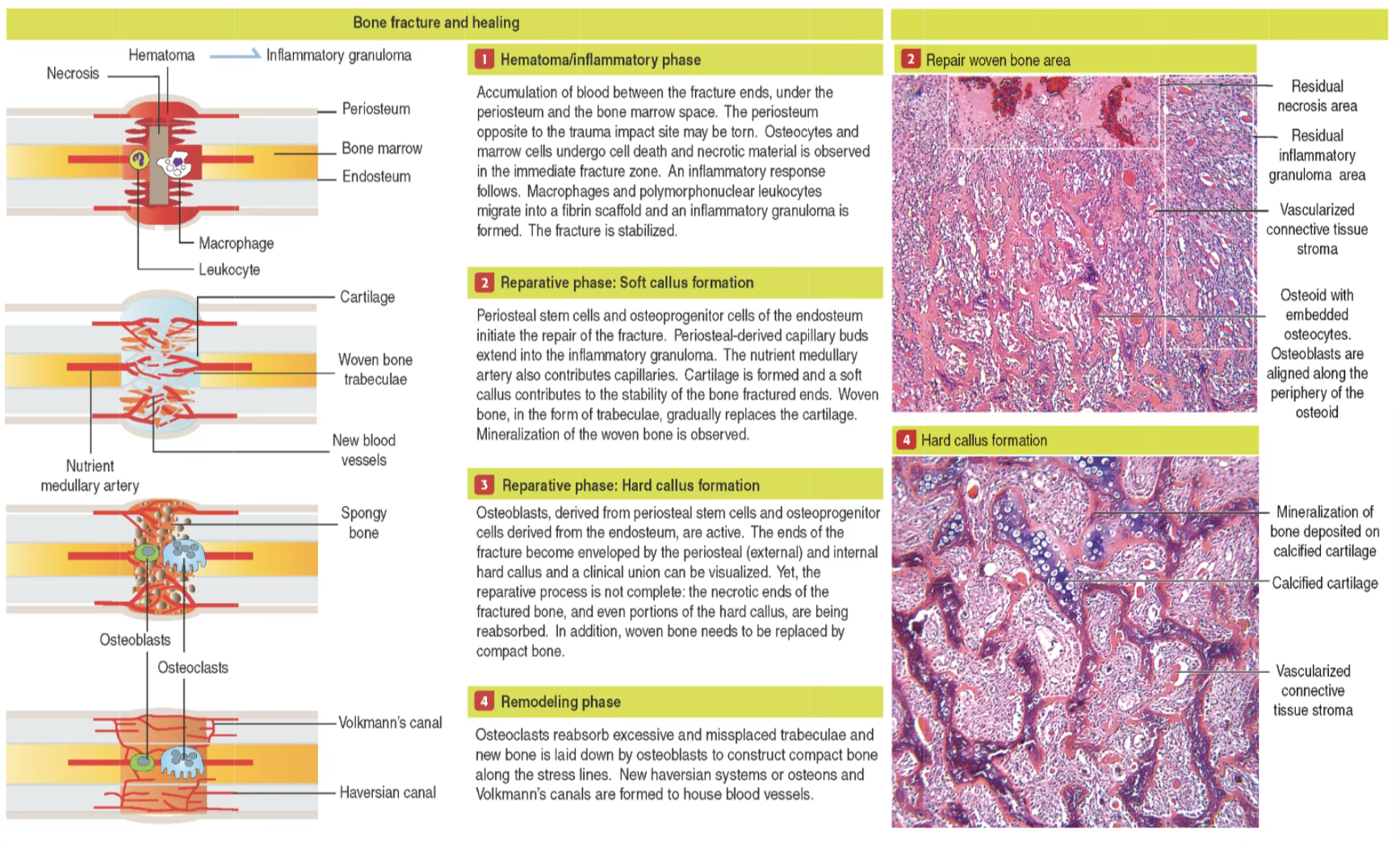
Phases of Fracture Repair
Phase 1 — Hematoma/Inflammatory Phase (Days 1–7)
- Massive disruption of blood vessels in haversian and Volkmann’s canals → hematoma
- Osteocytes and marrow cells undergo cell death; necrotic material in fracture zone
- Haversian systems without blood supply → osteocytes become pyknotic → lyse → empty lacunae
- Inflammatory response: macrophages, monocytes, lymphocytes, PMNs, fibroblasts recruited
- Fibrin scaffold forms → inflammatory granuloma (bulges over edges of fractured bone, connecting fragments)
- Cytokines from inflammatory cells and platelets recruit osteoprogenitor cells from periosteum and endosteum
- Blood clot invaded by capillaries and fibroblasts → granulation tissue; marrow clot invaded by osteoprogenitor cells → internal callus
Phase 2 — Reparative Phase: Cartilaginous Soft Callus (~Weeks 1–4)
- Phagocytic cells remove dead cells and damaged tissue
- Periosteal stem cells and osteoprogenitor cells of the endosteum initiate repair; periosteal-derived capillary buds and nutrient medullary artery contribute blood supply
- Soft callus (non-calcified cartilage) connects the two fractured ends → stability
- ~3–4 weeks after injury: osteoblasts (from periosteum and endosteum) penetrate and replace soft callus with woven bone
- Three zones in the collar:
- New bone cemented to the fragment
- Intermediate cartilage layer
- Proliferating osteogenic surface layer
- Osteoprogenitor cells in the center (avascular) → chondrogenic → chondroblasts → cartilage in outer collar
Phase 3 — Reparative Phase: Hard Bone Callus
- Collars from each fragment fuse → external callus → union of fragments
- Osteoblasts deposit osteoid → calcified → woven bone formed
- Fracture ends enveloped by periosteal (external) and internal hard callus → clinical union visible
- Necrotic ends and portions of hard callus are being reabsorbed; woven bone needs replacement by compact bone
Phase 4 — Remodeling Phase (~2–3 months post-injury)
- Osteoclasts reabsorb excessive and misplaced trabeculae
- Osteoblasts lay down compact bone along stress lines
- New haversian systems (osteons) and Volkmann’s canals form to house blood vessels
- Cartilage replaced by cancellous bone via endochondral bone formation
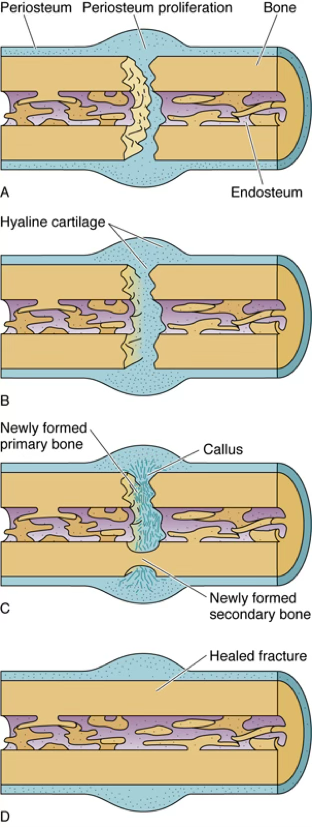
Mnemonic — Fracture Repair Phases
“Healing Calls Require Remodeling”
Hematoma/Inflammation → Cartilaginous soft callus → Reparative hard callus → Remodeling
Aging and Bone Loss
With aging, cortical bone remains stable until mid-life, when estrogen deficiency (women) and gradual sex steroid reduction (men) begins to drive cortical bone loss, increasing fracture risk.
List the four phases of bone fracture repair in order.
- Hematoma/Inflammatory phase
- Reparative phase: cartilaginous soft callus
- Reparative phase: hard bone callus
- Remodeling phase
Anki cloze
In bone fracture repair, osteoprogenitor cells situated away from capillaries in the proliferating periosteal mass become {1:chondrogenic cells} rather than osteoblasts.
What is woven bone and when is it produced?
Woven bone is mechanically weak bone with randomly oriented collagen fibers. It is produced rapidly during bone fracture repair (and during normal bone development) and is later replaced by lamellar bone.
Synovial Joints
She clearly said that we don't need to remember the types of joints. good.
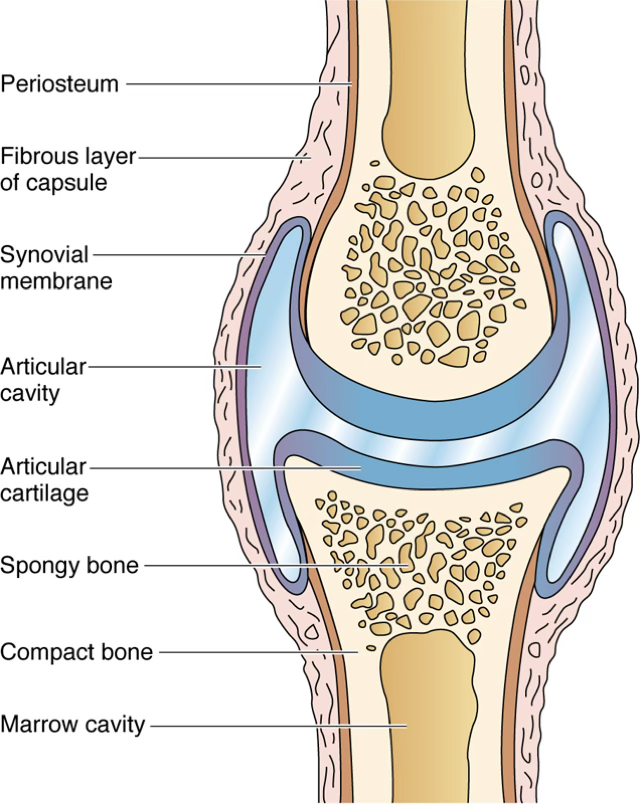
Classification of Joints
| Class | Movement | Examples |
|---|---|---|
| Synarthrosis | Minimal | Skull bones (adults), first rib-sternum, pubic symphysis |
| Diarthrosis | Wide range | Most limb joints |
Types of Synarthrosis
| Type | Uniting Tissue | Example |
|---|---|---|
| Synostosis | Bone | Skull bones in adults |
| Synchondrosis | Hyaline cartilage | First rib and sternum |
| Syndesmosis | Dense connective tissue | Pubic symphysis |
Diarthrodial (Synovial) Joint Structure
- Bones covered by persistent articular cartilage (hyaline cartilage)
- Ligaments maintain contact between bones
- Joint capsule:
- Outer fibrous layer: dense connective tissue; continuous with periosteum
- Inner cellular layer (synovial membrane / synovial layer): covers all non-articular surfaces
- Articular cavity between the cartilage-covered surfaces
TLDR
Comprehensive Summary — Bone Histology
Bone is a mineralized connective tissue (ECM + / salts) that provides structural support, mineral storage (~99% body calcium), and houses hematopoietic marrow.
Matrix composition: type I collagen fibers; ground substance with chondroitin sulfate, keratan sulfate proteoglycans; glycoproteins (osteonectin, osteocalcin, osteopontin, bone sialoprotein).
Four bone cell types:
- Osteoprogenitor: mitotic stem-like cells (mesenchymal origin) in periosteum, endosteum, haversian canals
- Osteoblasts: secrete osteoid (organic matrix); basophilic cytoplasm when active
- Osteocytes: trapped osteoblasts in lacunae; maintain bone; perform mechanotransduction via cAMP/osteocalcin/IGF release
- Osteoclasts: large, multinucleated (up to 50 nuclei), acidophilic; GM-CFU origin; resorb bone in Howship’s lacunae; have 4 zones: ruffled border, clear zone, vesicular zone, basal zone
Gross structure: compact bone (solid, diaphysis) vs. spongy/cancellous bone (trabeculae + marrow, epiphyses); covered by periosteum (outer fibrous + inner osteogenic layers); lined internally by endosteum.
Microscopic types: Lamellar bone (regular collagen, strong, slow, mature) vs. woven bone (random collagen, weak, rapid, developmental/repair).
Lamellar patterns: osteons (Haversian systems, 4–20 concentric lamellae around Haversian canal); interstitial lamellae; outer circumferential lamellae; inner circumferential lamellae. Haversian canals = longitudinal; Volkmann’s canals = transverse/oblique connections.
Osteogenesis — intramembranous: mesenchyme → direct osteoblast differentiation (no cartilage); forms flat skull bones, clavicle; produces woven then lamellar bone; Wnt/Hedgehog/FGF/TGF-β signaling.
Osteogenesis — endochondral: hyaline cartilage template → hypertrophic chondrocytes → VEGF → vascular invasion → osteoblasts deposit bone; primary ossification center (diaphysis, ~3rd fetal month); secondary centers (epiphyses, after birth); epiphyseal plate zones: proliferative → hypertrophic (type X collagen, VEGF, apoptosis) → vascular invasion.
Trabecular → osteon conversion: groove + blood vessel → tunnel → concentric lamellae deposited → osteon; interconnected by Volkmann’s canals.
Bone remodeling: cortical remodeling occurs within osteons (osteoclasts resorb → osteoblasts refill → new osteon); trabecular remodeling occurs on bone surfaces.
Fracture repair (4 phases):
- Hematoma/Inflammation (week 1): fibrin scaffold, granulation tissue, osteoprogenitor recruitment
- Soft callus (weeks 1–4): cartilage bridging, soft callus, early woven bone
- Hard callus: woven bone callus, clinical union
- Remodeling (months): osteoclasts remove excess, osteoblasts form compact bone along stress lines; new osteons and Volkmann’s canals
Synovial joints: synarthroses (synostosis/synchondrosis/syndesmosis) vs. diarthroses (articular cartilage, joint capsule with fibrous outer + synovial inner layers).