TARGET DECK: MED::I::Signaling Pathways in Health and Disease::Metabolic Biochemistry::10 - Pentose Pathway
Pentose Phosphate Pathway – Overview
Main Products
The two main products of the PPP are NADPH and ribose-5-phosphate.
NADPH (electron donor)
- Used in biosynthesis: fatty acids, cholesterol, steroids
- Used for repair of oxidative damage: reduction of glutathione
Ribose-5-phosphate (biosynthetic precursor of nucleotides)
- Used in DNA and RNA synthesis
- Used in the synthesis of some coenzymes
What are the two main products of the Pentose Phosphate Pathway?
NADPH and ribose-5-phosphate
Anki cloze
NADPH is used in {1:biosynthesis (fatty acids, cholesterol, steroids)} and for {2:repair of oxidative damage via reduced glutathione}.
NADH vs NADPH
Key Ratios
- NADPH/NADP⁺ ratio kept high → reflects role in biosynthesis and cellular protection from oxidation
- NADH/NAD⁺ ratio kept low → NADH used in respiration, much of it in mitochondria
| Coenzyme | Ratio maintained | Primary role |
|---|---|---|
| NADPH/NADP⁺ | High | Biosynthesis & antioxidant protection |
| NADH/NAD⁺ | Low | Cellular respiration (mitochondria) |
Malate-Aspartate Shuttle (context)
The malate–aspartate shuttle transfers NADH equivalents from the cytosol into the mitochondrial matrix:
- Intermembrane space (P side): malate-α-ketoglutarate transporter, glutamate-aspartate transporter
- Matrix (N side): malate dehydrogenase, aspartate aminotransferase
- Net effect:
Why is the NADPH/NADP⁺ ratio kept high while the NADH/NAD⁺ ratio is kept low?
NADPH/NADP⁺ is high because NADPH is needed for biosynthesis and antioxidant defense. NADH/NAD⁺ is kept low because NADH is rapidly consumed by the mitochondrial respiratory chain.
Pentose Phosphate Pathway – Alternative Pathway of Glucose Oxidation
General Position in Metabolism
The PPP is an alternative pathway of glucose oxidation running parallel to glycolysis:
Glucose-6-P can be used in glycolysis to obtain ATP and NADH or it can be used in the PPP to obtain NADPH and Ribose-5-P.
Pentose Phosphate Pathway – Two Phases
Two Phases
- Oxidative phase – irreversible (up to ribulose-5-P, a ketose molecule); generates NADPH
- Non-oxidative phase – reversible (interconversions of sugar phosphates. made only of reversible reactions associated with a positive .)
Anki cloze
The oxidative phase of the PPP is {1:irreversible} and produces {2:NADPH and ribulose-5-phosphate}. The non-oxidative phase is {3:reversible} and performs sugar phosphate interconversions.
Pentose Phosphate Pathway – General Scheme
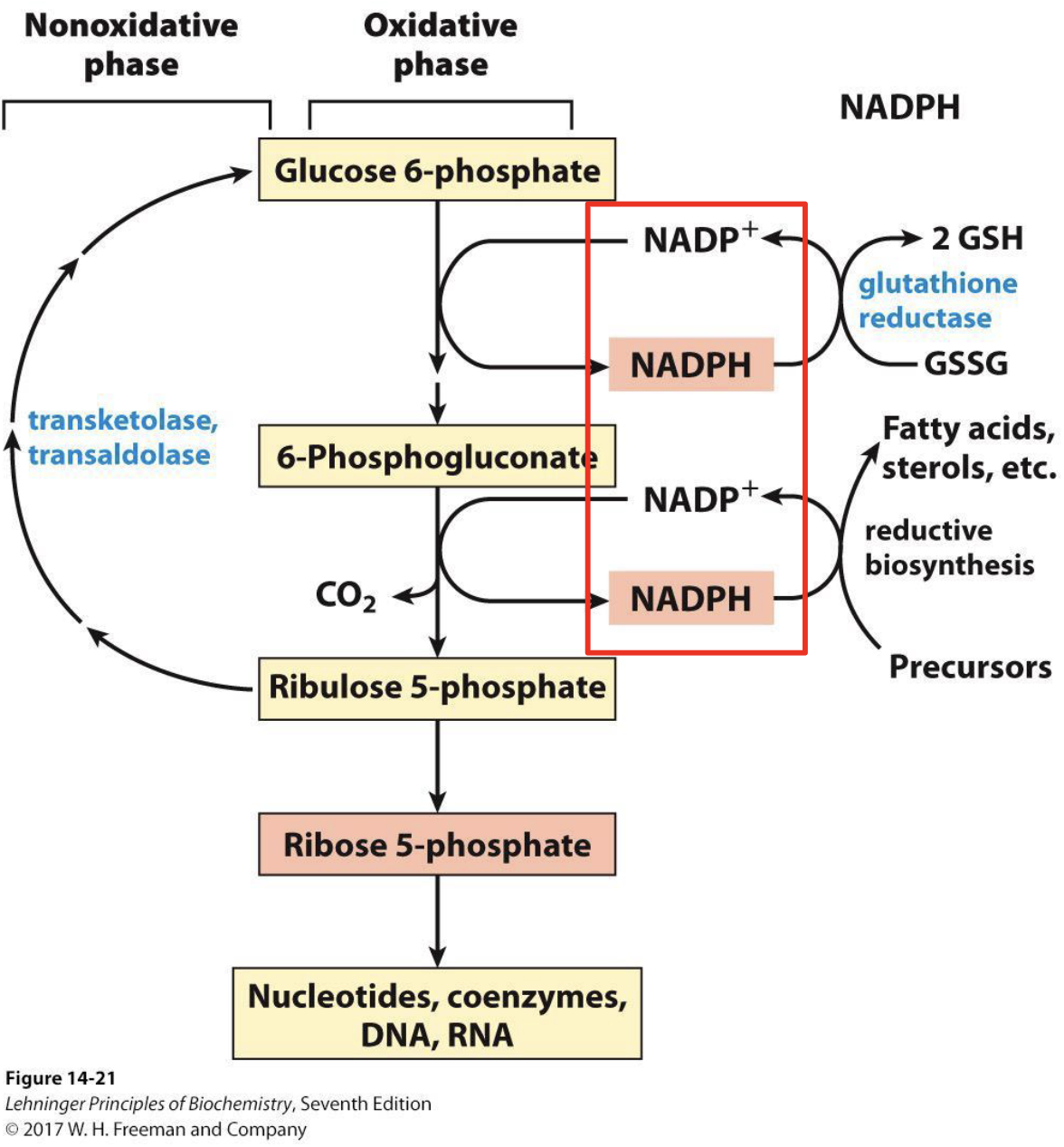
Overview of Both Phases
Oxidative phase:
Glucose-6-P → 6-Phosphogluconate (intermediate, then further oxidised) → Ribulose-5-phosphate
Non-oxidative phase (transketolase + transaldolase reactions):
- Converts 5-carbon sugars back to 6-carbon sugars (→ regenerates G6P for NADPH-demanding tissues)
- OR
- generates glycolytic intermediates (G3P, F6P) for ATP synthesis when ribose-5-P is not needed
Glucose-6-P
↓ (oxidative phase)
6-Phosphogluconate
↓
Ribulose-5-P + CO₂ + 2 NADPH
↓ (non-oxidative phase)
Ribose-5-P ←→ Xylulose-5-P
↓ transketolase / transaldolase
G3P + F6P → glycolysis
NADPH fates
- Reductive biosynthesis: fatty acids, sterols
- Glutathione reductase: 2 GSH regeneration (antioxidant defense)
Oxidative Phase – Reactions
Step 1: Glucose-6-phosphate → 6-Phosphogluconolactone
- Enzyme: Glucose-6-phosphate dehydrogenase (G6PDH)
- Rate-limiting step of the PPP
Step 2: 6-Phosphogluconolactone → 6-Phosphogluconate
Step 3: 6-Phosphogluconate → Ribulose-5-phosphate
Net of Oxidative Phase
What is the rate-limiting enzyme of the PPP oxidative phase?
Glucose-6-phosphate dehydrogenase (G6PDH)
Anki cloze
The oxidative phase of the PPP generates {1:2 NADPH} and {2:CO₂} per glucose-6-phosphate oxidized, yielding {3:ribulose-5-phosphate}.
Non-Oxidative Phase – Part 1: Isomerizations
First Reactions:
Epimerase and Isomerase Ribulose-5-phosphate is converted by two enzymes:
- Epimerase → Xylulose-5-phosphate
- Isomerase → Ribose-5-phosphate
What are the two products of ribulose-5-phosphate isomerization/epimerization in the PPP non-oxidative phase?
Xylulose-5-phosphate (via epimerase) and Ribose-5-phosphate (via isomerase)
Non-Oxidative Phase – Part 2: Transketolase Reaction 1
First Transketolase Reaction Transfer of a 2-carbon unit from a ketose to an aldose
Cofactor: Thiamine Pyrophosphate (TPP) Mechanism: nucleophilic attack by the TPP carbanion on the carbonyl group of the ketose donor. TPP is a coenzyme derived from thiamine (vitamin B1).
Anki basic
What cofactor is required by transketolase in the PPP non-oxidative phase? Thiamine Pyrophosphate (TPP), derived from thiamine (vitamin B1)
Anki cloze
Transketolase transfers {1:2-carbon} units from a {2:ketose} to an {3:aldose}, using the cofactor {4:TPP (thiamine pyrophosphate)}.
Non-Oxidative Phase – Part 3: Transaldolase Reaction
Transaldolase Reaction (Reaction 3) Transfer of a 3-carbon unit
Anki cloze
Transaldolase transfers {1:3-carbon} units, converting sedoheptulose-7-P + G3P into {2:erythrose-4-P + fructose-6-P}.
Non-Oxidative Phase – Part 4: Transketolase Reaction 2
Second Transketolase Reaction (Reaction 4) Transfer of another 2-carbon unit
Non-Oxidative Phase – Summary of Interconversions
Starting Material:
3 Ribulose-5-P To obtain: 2 Fructose-6-P + 1 Glyceraldehyde-3-P
Conversion chain:
- 3 Ribulose-5-P → 1 Ribose-5-P + 2 Xylulose-5-P (epimerase/isomerase)
- Xylulose-5-P + Ribose-5-P → G3P + Sedoheptulose-7-P (transketolase)
- Sedoheptulose-7-P + G3P → Erythrose-4-P + F6P (transaldolase)
- Xylulose-5-P + Erythrose-4-P → G3P + F6P (transketolase)
Carbon Accounting:
6C₅ → 5C₆
Detailed interconversion balance:
What is the net carbon conversion of the complete PPP non-oxidative phase?
6 × C₅ → 5 × C₆ (six pentose phosphates yield five hexose phosphates)
Non-Oxidative Phase – Regeneration of G6P
Tissues Requiring More NADPH Than Ribose-5-P (e.g., liver and adipose tissue) The non-oxidative phase regenerates glucose-6-phosphate from ribose-5-phosphate via the reverse reactions of transketolase and transaldolase, allowing continuous cycling through the oxidative phase for maximal NADPH production.
Alternative Sources of NADPH
Beyond G6PDH – Other Cytosolic/Mitochondrial NADPH Sources
- Residual activity of glucose-6-phosphate dehydrogenase
- Cytosolic isocitrate dehydrogenase (requires NADP⁺ → produces NADPH)
- Nicotinamide nucleotide transhydrogenase (mitochondrial):
- Malic enzyme (cytosolic):
Name four sources of cytosolic/mitochondrial NADPH other than G6PDH.
- Residual G6PDH activity; 2. Cytosolic isocitrate dehydrogenase; 3. Nicotinamide nucleotide transhydrogenase; 4. Malic enzyme
Regulation of the PPP
Rate-Limiting Step Glucose-6-phosphate dehydrogenase (G6PDH) is the rate-limiting enzyme.
- Regulated by substrate availability: NADP⁺ allosterically activates G6PDH
- When NADPH is consumed (↑ NADP⁺), G6PDH is activated → more NADPH produced
Anki cloze
The rate-limiting enzyme of the PPP is {1:glucose-6-phosphate dehydrogenase (G6PDH)}, which is allosterically activated by {2:NADP⁺}.
Hormonal Regulation of the PPP
Hormones Act Mainly by Gene Expression
| Hormone | Effect on PPP enzymes | Mechanism |
|---|---|---|
| Insulin | Induces (↑) G6PDH | Activates fatty acid & cholesterol biosynthesis → ↓ NADPH/NADP⁺ → activates G6PDH |
| Glucagon | Represses (↓) PPP enzymes | Opposes insulin actions |
How does insulin regulate the PPP? Insulin induces PPP enzymes (especially G6PDH).
By activating fatty acid and cholesterol biosynthesis (which consume NADPH), insulin decreases the NADPH/NADP⁺ ratio, thereby activating G6PDH.
Functions of the PPP – Metabolic Modes
Four Functional Modes of the PPP
Mode 1: Only Oxidative Phase (reducing power + pentoses)
Mode 2: Oxidative + Non-Oxidative Phase (reducing power only)
Simplifies to:
Mode 3: Only Non-Oxidative Phase Backwards (pentose synthesis from glucose)
Mode 4: Only Non-Oxidative Phase Forwards (pentose catabolism)
How many NADPH can be produced from one glucose-6-phosphate when both oxidative and non-oxidative phases run together?
12 NADPH (with complete oxidation to 6 CO₂)
Uses of NADPH
1. Reductive Biosynthesis
- Fatty acid synthesis
- Cholesterol synthesis
2. Hydroxylation Reactions (mono-oxygenase / cytochrome P-450)
- Several biosynthetic processes
- Catabolism of xenobiotics (drug metabolism)
Cytochrome P-450 System
- Cytochrome P-450 reductase (Fe-S) transfers electrons from NADPH to cyt P-450
- Other reductants used in hydroxylations:
- α-ketoglutarate → proline hydroxylation
- Tetrahydrobiopterin → catecholamine hydroxylation
3. Reduction of Glutathione (detoxification of hydroperoxides)
What are the three main uses of NADPH in the cell?
- Reductive biosynthesis (fatty acids, cholesterol); 2. Hydroxylation reactions (cytochrome P-450, xenobiotic metabolism); 3. Regeneration of reduced glutathione (GSH) for antioxidant defense
Glutathione Structure and Function
Glutathione (GSH) – Structure A tripeptide: γ-glutamyl – cysteinyl – glycine
- Linked by an unusual γ-carboxyl linkage (not α) between glutamate and cysteine
- The thiol (-SH) group of cysteine is the reactive moiety
OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS)C(=O)NCC(=O)OGlutathione Redox Cycle
Critical Tissues for Glutathione
- Erythrocytes: full of iron and oxygen; susceptible to Fenton reaction
- •OH is the most damaging of all ROS
- Brain: most aerobically active tissue; ROS may contribute to Alzheimer’s disease
Anki cloze
Glutathione is a tripeptide of {1:γ-glutamate – cysteine – glycine}. Its oxidized form {2:GSSG} is regenerated to {3:2 GSH} by glutathione reductase using {4:NADPH} as electron donor.
Glutathione and ROS Defence
Reactive Oxygen Species (ROS) Cascade
Reduction potentials:
Key Antioxidant Enzymes
- SOD (Superoxide dismutase):
- Catalase:
- Glutathione peroxidase (selenium-dependent): reduces using GSH
ROS Sources Mitochondrial respiration, ionizing radiation, sulfa drugs, herbicides, antimalarials, divicine
What is the Fenton reaction and why is it dangerous in erythrocytes?
Fe²⁺ + H₂O₂ → Fe³⁺ + •OH + OH⁻. The hydroxyl radical (•OH) is the most damaging ROS, and erythrocytes are rich in both iron and oxygen, making them especially vulnerable.
Glucose-6-Phosphate Dehydrogenase Deficiency
G6PD Deficiency – Erythrocyte Vulnerability Erythrocytes are uniquely vulnerable because they:
- Are formed in bone marrow; live 90–120 days; old ones removed by liver and spleen
- Have no nuclei or mitochondria → cannot make or replace enzymes
- Have limited metabolism: only glycolysis and PPP
- Depend on glutathione to deal with ROS
- Depend on NADPH (via PPP/G6PDH) to replenish reduced glutathione
Hemolytic Anemia in G6PD Deficiency
- Active hemoglobin (; ferrous) oxidized to methemoglobin (; ferric)
- ↑ ROS → depletion of reduced glutathione
- Erythrocytes lyse, releasing contents into bloodstream
- Oxidation triggered by: antimalarial drugs (e.g. primaquine), fava beans
- Can be treated with transfusions
Primaquine (triggering agent) Structure: 8-aminoquinoline antimalarial
CCCCNc1ccnc2cc(OC)ccc12Why do erythrocytes have no capacity to replace G6PD enzyme once it is inactivated?
Erythrocytes have no nuclei or mitochondria, so they cannot synthesize new proteins or replace enzymes once they are lost or damaged.
G6PD Deficiency – Epidemiology and Genetics
Most Common Human Genetic Deficiency
| Feature | Detail |
|---|---|
| Prevalence | ~400,000,000 people worldwide |
| Origin | Emerged >10,000 years ago |
| Known mutations | >300 mutations that decrease enzyme activity |
| Inheritance | X-linked → males affected most often |
| Geographic distribution | Closely coincides with malaria-endemic regions |
| Evolutionary advantage | Lowered G6PD activity confers some protection from malaria (reduces suitability of erythrocytes for hosting malarial parasite) |
G6PD deficiency is {1:X-linked}, affects approximately {2:400 million} people worldwide, and confers partial protection against {3:malaria}.
PPP and Human Diseases – Wernicke-Korsakoff Syndrome
Wernicke-Korsakoff Syndrome Results from severe deficiency of thiamine (vitamin B1), a component of the cofactor TPP required by transketolase in the non-oxidative phase of the PPP.
Consequences
- PPP is slowed → attenuated NADPH production
- Symptoms: memory loss, mental confusion, partial paralysis
- Common in alcoholics: ethanol inhibits thiamine (B1) uptake
TPP Requirement Distinction Oxidative decarboxylations that require TPP:
- Pyruvate dehydrogenase
- α-Ketoglutarate dehydrogenase
- Branched-chain α-ketoacid dehydrogenase
Oxidative decarboxylations that do NOT require TPP (hydroxy acids):
- Malic enzyme
- Isocitrate dehydrogenase
- 6-Phosphogluconate dehydrogenase
Why does thiamine (B1) deficiency impair the PPP?
Thiamine is a precursor of TPP (thiamine pyrophosphate), the essential cofactor for transketolase. Without TPP, the non-oxidative phase of the PPP is blocked, reducing NADPH production.
Anki cloze
Wernicke-Korsakoff syndrome is caused by deficiency of {1:thiamine (vitamin B1)}, which impairs {2:transketolase} in the PPP non-oxidative phase; it is common in {3:alcoholics} because ethanol inhibits {4:thiamine uptake}.
Synthesis of Polysaccharides
Mucopolysaccharides (Glycosaminoglycans)
- E.g. Hyaluronic acid, chondroitin sulfate
- Contain: glucuronic acid, N-acetylglucosamine or N-acetylgalactosamine
- Enzymes: glycosyl transferases; activated sugar donors: UDP-sugars
Glycoproteins
- Isoprenoid lipid dolichol transfers oligosaccharide chains to protein
UDP-Galactose Reactions
- Non-lactating tissues: UDP-galactose + N-acetyl-D-glucosamine → D-galactosyl-N-acetyl-D-glucosamine (→ glycoprotein)
- Lactating mammary gland: UDP-galactose + D-glucose D-lactose
Glucuronic Acid
Functions of Glucuronic Acid
| Function | Detail |
|---|---|
| Structural | Component of mucopolysaccharides (e.g. hyaluronic acid = glucuronate + N-acetylglucosamine polymer) |
| Detoxification | Glucuronidation of physiological and xenobiotic compounds (e.g. bilirubin from heme catabolism) |
| Biosynthetic precursor | Precursor for synthesis of vitamin C (ascorbic acid) in non-primates |
Biosynthetic Pathway
From UDP-glucuronate:
- → Insertion of glucuronate residues into glycosaminoglycans (hyaluronate, chondroitin sulfate)
- → Glucuronidation of drugs and toxins
Vitamin C synthesis pathway (non-primates):
Humans lack gulonolactone oxidase → cannot synthesize vitamin C
What are the two main roles of glucuronic acid in human metabolism?
- Structural component of glycosaminoglycans (e.g. hyaluronic acid, chondroitin sulfate); 2. Glucuronidation of xenobiotics and physiological compounds (e.g. bilirubin) for detoxification
Mnemonics
Mnemonic: PPP Non-Oxidative Phase Enzymes "Every Intelligent Transporter Trips And Tumbles"
- Epimerase
- Isomerase
- Transketolase (reaction 1)
- Transaldolase
- Transketolase (reaction 2)
Mnemonic: TPP-requiring decarboxylations (α-KETOacids only) "Pretty Keto? Better Take Two Pills" → Pyruvate DH, Ketoglutarate DH, Branched-chain DH → all need TPP × 2 (α-keto structure) Hydroxy-acids (malate, isocitrate, 6-PG) → NO TPP needed.
Mnemonic: Glutathione tripeptide "Glu-Cys-Gly" = "Good Cells Guard" (γ-Glutamate – Cysteine – Glycine)
TLDR - 10 - Pentose Phosphate Pathway
Pentose Phosphate Pathway – Comprehensive Summary
- The PPP is an alternative glucose oxidation pathway producing NADPH and ribose-5-phosphate; runs in cytosol parallel to glycolysis.
- NADPH/NADP⁺ kept high (biosynthesis, antioxidant); NADH/NAD⁺ kept low (respiration).
- Two phases: (1) irreversible oxidative phase (G6P → Ribulose-5-P + 2 NADPH + CO₂); (2) reversible non-oxidative phase (pentose interconversions via transketolase/transaldolase).
- Oxidative phase enzymes: G6PDH (rate-limiting) → 6-phosphogluconolactonase → 6-phosphogluconate dehydrogenase.
- Non-oxidative phase: epimerase (→ Xyl-5-P), isomerase (→ Rib-5-P), transketolase (2C transfer, TPP-dependent), transaldolase (3C transfer).
- Carbon balance: ; non-oxidative phase regenerates G6P or feeds glycolysis (G3P, F6P).
- Regulation: G6PDH allosterically activated by NADP⁺; insulin induces (via ↓NADPH/NADP⁺), glucagon represses.
- Alternative NADPH sources: cytosolic isocitrate DH, malic enzyme, nicotinamide nucleotide transhydrogenase.
- NADPH uses: (1) reductive biosynthesis (fatty acids, sterols); (2) cytochrome P-450 hydroxylations; (3) glutathione reductase (regenerates GSH from GSSG).
- Glutathione (γ-Glu–Cys–Gly): protects against ROS; especially critical in erythrocytes (Fenton reaction: Fe²⁺ + H₂O₂ → •OH) and brain (Alzheimer’s).
- G6PD deficiency: most common human genetic deficiency (~400M); X-linked; causes hemolytic anemia triggered by antimalarials (primaquine), fava beans; protective against malaria.
- Wernicke-Korsakoff syndrome: thiamine (B1) deficiency → impaired transketolase (TPP-dependent) → ↓ NADPH; common in alcoholics.
- TPP only required for α-ketoacid oxidative decarboxylation (pyruvate DH, α-KG DH, branched-chain DH); NOT for hydroxy-acid decarboxylations (malic enzyme, isocitrate DH, 6-PG DH).
- Glucuronic acid: UDP-glucose → UDP-glucuronate (via UDP-glucose dehydrogenase, 2 NAD⁺); used in glycosaminoglycans (hyaluronate, chondroitin sulfate) and glucuronidation (drug/bilirubin detox); precursor for vitamin C in non-primates (humans lack gulonolactone oxidase).
- Polysaccharide synthesis uses UDP-sugars + glycosyl transferases; lactose synthesis in mammary gland requires α-lactalbumin to redirect UDP-galactose transferase to glucose.