1. Introduction to Physiology
Physiology is the science that studies the functions of living organisms.
In this course.. we’ll be studying… cell physiology >:)
The cell is a biological system that maintains its characteristics unchanged thanks to:
- interaction with the external environment
- renewal of its components
- regulation of its functions
Homeostasis: The maintenance of a “normal” range of the “fundamental” variables of the extracellular and intracellular compartments.
Multicellular organisms: cells differentiate and develop specific functions (mechanical, biochemical, information processing), leading to the development of systems; they present refined regulatory and interaction mechanisms with the external environment to obtain energy, preserve the internal environment, and respond to changes in the external environment (allostasis) → Systems physiology
2. Eukaryotic Cells → Plasma Membrane (Mb)
Eukariotic cells differ from prokaryotic cells because of mb delimited nucleus.
All eukaryotic cells, apart human red blood cells and cells within the lens of the eye, have a nucleus and cytoplasm.
Eukaryotic Cell Components Table
idfc
| Component | Primary Function |
|---|---|
| Cytosol | Metabolism, protein synthesis (via free ribosomes). |
| Cytoskeleton | Cell shape and movement, intracellular transport. |
| Nucleus | Genome (22 autosomes and 2 sex chromosomes), DNA and RNA synthesis. |
| Mitochondria | ATP synthesis by oxidative phosphorylation, storage. |
| Smooth ER | Synthesis of lipids, storage. |
| Free Ribosomes | Translation of mRNA into cytosolic proteins. |
| Rough ER | Translation of mRNA into membrane-associated proteins or for secretion. |
| Lysosome | Intracellular degradation. |
| Endosome | Cellular uptake of cholesterol, receptor removal, and internalization of particles. |
| Golgi Apparatus | Modification, sorting, and packaging of proteins and lipids. |
| Proteasome | Degradation of intracellular proteins. |
| Peroxisome | Detoxification of substances. |
Main functions
Plasma membrane separates intracellular from extracellular env.
- Selective transport of molecules in and out of the cell (mb transport proteins)
- Cell recognition through the use of surface antigens
- Cell communication through neurotransmitter or hormone receptors and through signal transduction pathways
- Tissue organization, such as temporary or permanent cell junctions or interaction through extracellular matrix
- Mb dependent enzymatic activity
- Determination of cell shape by linkage with cytoskeleton
Structure and Composition
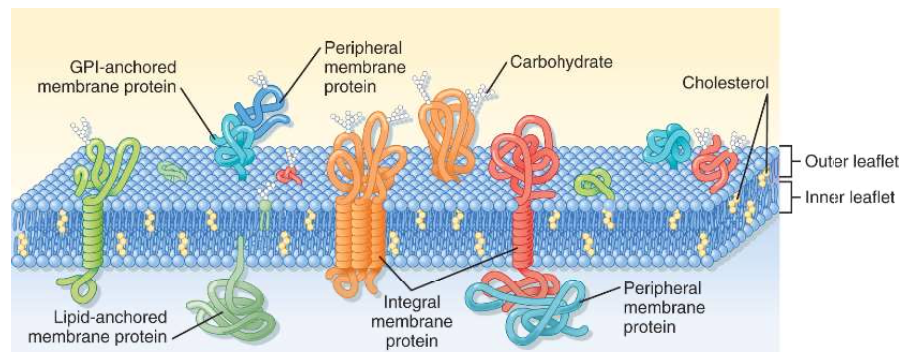
5nm-thick lipid bilayer with associated proteins integrated or loosely attached to the inner or outer surface
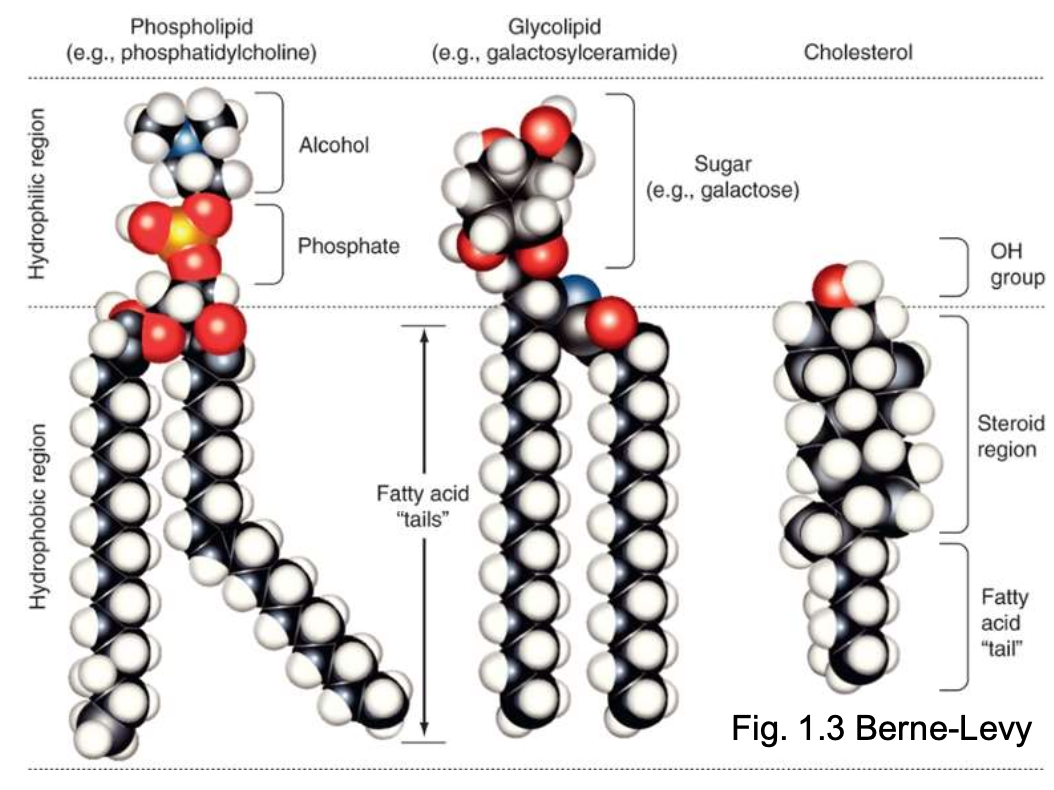
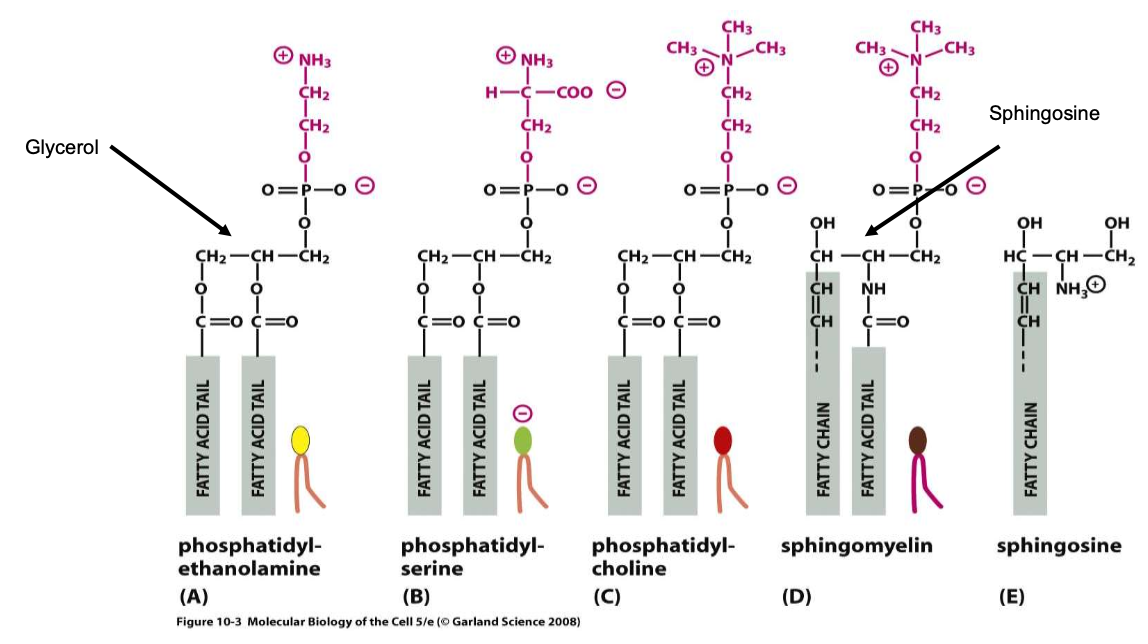
The major part of lipids of the cell mb are phospholipids, and in particular glycerophospholipids (e.g. phosphatidylcholine).
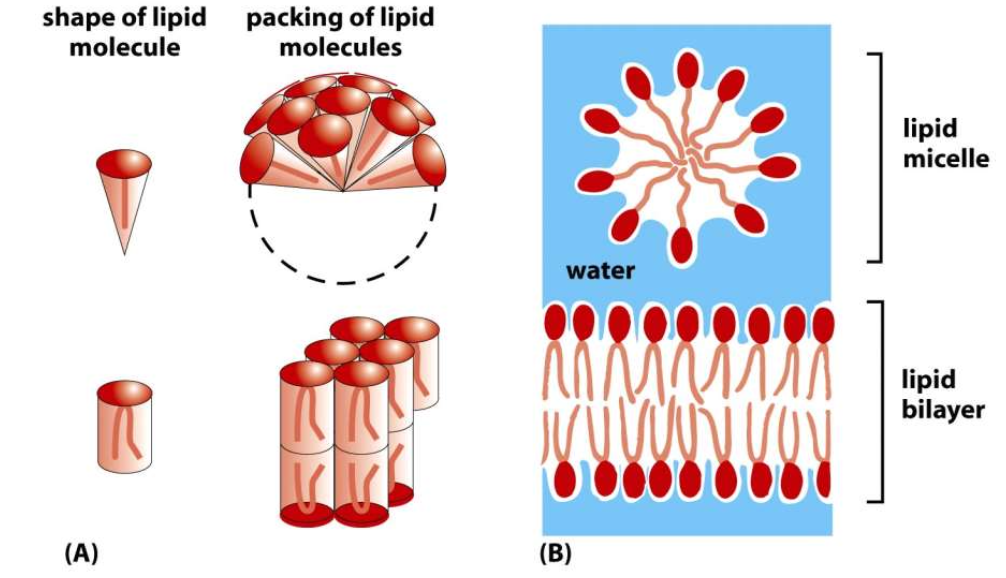
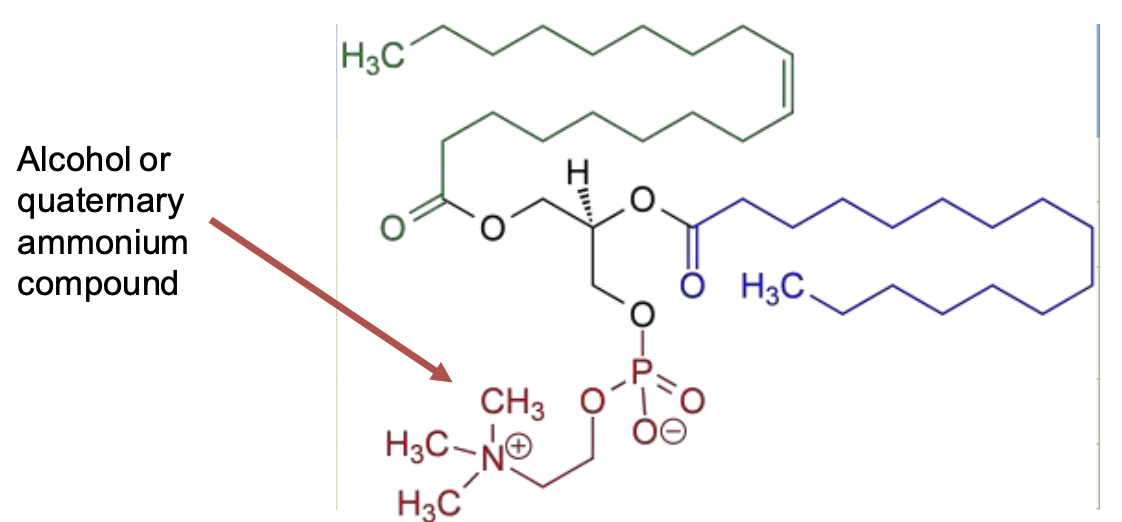
Phospholipids are amphipathic molecules containing a polar hydrophilic head and two non polar hydrophobic fatty acid chains.
Majority of phospholipids have a glycerol backbone to which are attached 2 fatty acyl chains (14-20C) and, via a phosphate group, an alcohol.
Sphingomyelin has the amino-alcohol sphingosine as its backbone instead of glycerol.
In one of the two fatty acyl chain there is a double bond (unsaturated fatty acyl chain) that produces a kink in the 3D structure of the molecule.
Glycolipids contain 2 apolar fatty acyl chains linked to a polar head that consists of a carbohydrate.
Glycosylphosphatylinositol (GPI) anchors proteins to the outer leaflet of the mb.
Cholesterol is a critical component of the bilayer and serves to stabilize the mb structure at 37°C. We need it to regulate the fluidity of the mb.
50% of the lipids found in mb can be cholesterol.
 |  |
|---|
| Phospholipid | Primary Location in Membrane |
|---|---|
| Phosphatidylcholine | Outer leaflet |
| Sphingomyelin | Outer Leaflet |
| Phospatidylethanolamine | Inner Leaflet |
| Phosphatidylserine | Inner leaflet |
| Phosphatidylinositol | Inner Leaflet |
Cholesterol can be found pretty much everywhere.
The 3D structure of the lipid has consequences in physiology.
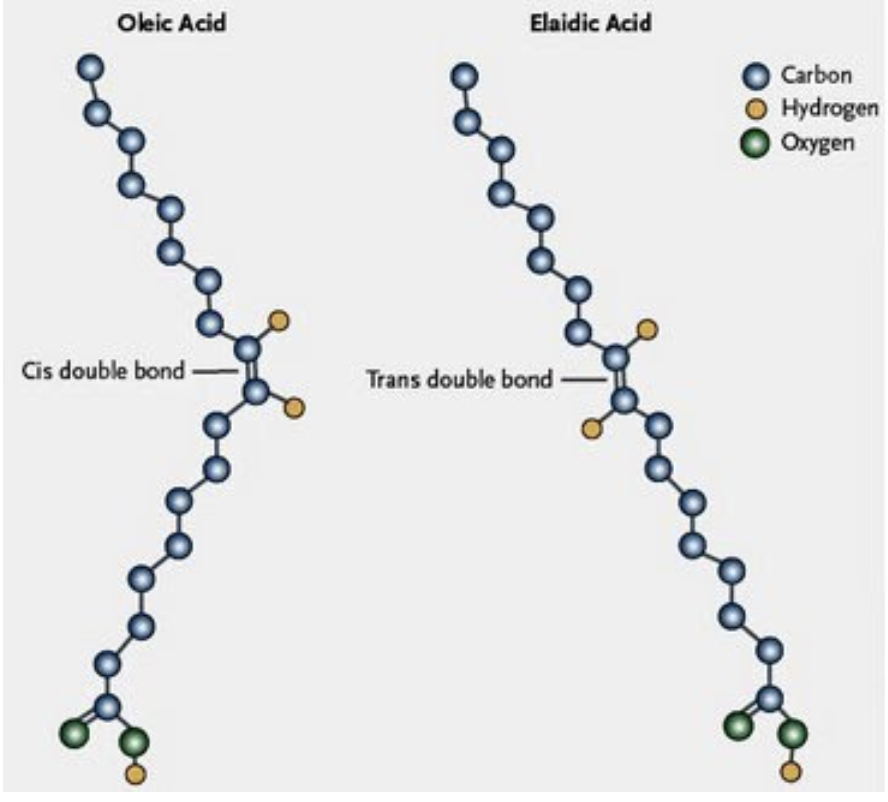
The fatty acid chains can be found in two ways:
- Cis double bond
- H Atoms on opposite sides.
- → sharp kink in the molecule → occupies MORE SPACE.
- H Atoms on opposite sides.
- Trans double bond
- H Atoms on same side.
- → still an angle, not as much as cis. similar to a saturated fat.
- H Atoms on same side.
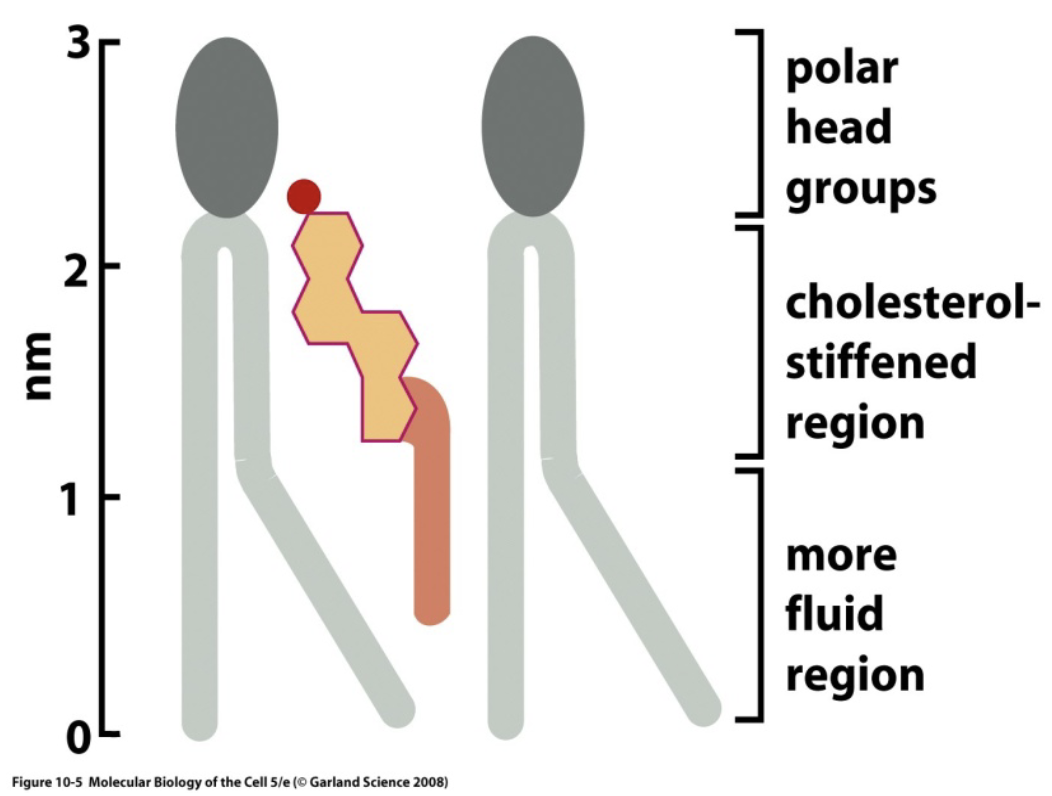
Cholesterol increases the viscosity of the mb, limiting it’s movement.
This happens at 37°C.
Whenever the temperature is lower, cholesterol has the opposite effect, increasing the fluidity of the mb.
This as a consequence sees the more important core organs like the heart and the brain strictly regulated by homeostasis, while other organs like the hands can be more loosely regulated.
Membrane fluidity depends on:
- The number of cis configurations.
- The number of cholesterol molecules.
Differences
Between types of cells
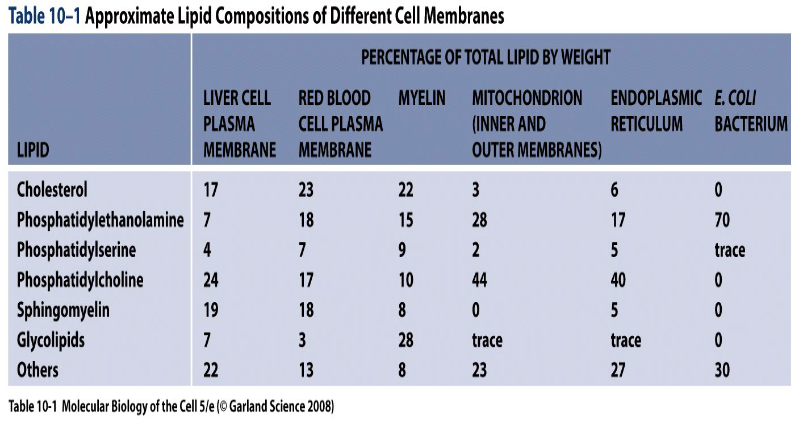
Between the sides of the mb
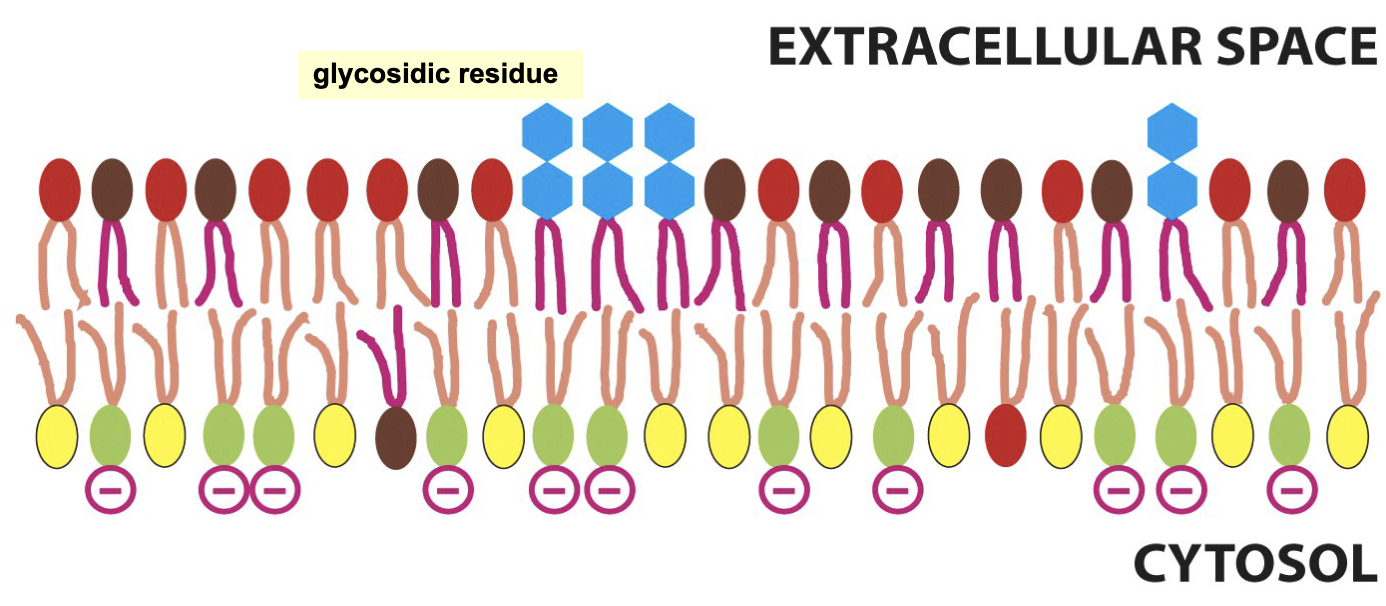
The composition of the two lipid layers is different.
Some lipids in the outside layer are connected to glycosidic residue, that allow for cell recognition.
Yellow → Phosphatidylethonolamine
Green → Phosphatidylserine
Red → Phosphatidylcholine
Brown → Sphingomyelin
Movement and Separation
Mb is not a static structure but rather fluid even if movement of proteins can be constrained or limited.
There are functional separated zones of the mb, e.g. tight junctions can separate (functionally) the plasma mb of epithelial cells in apical and basolateral mb that carry different roles in the transport of substances from one side of the epithelium to the opposite site.
The apical membrane looks to the lumen, the basolateral membrane looks at the rest of the body.
It is also possible to find regions of the mb where lipids like sphingomyelin and cholesterol aggregate into what are called lipid rafts, important to segregate signaling molecules.
Lipids can constantly move laterally.
Such movement is temperature dependent.
Since we can assume the temperature of the body as constant, the membrane composition is the key factor.
Lipids can also interchange (flip-flop), but it rarely occurs.
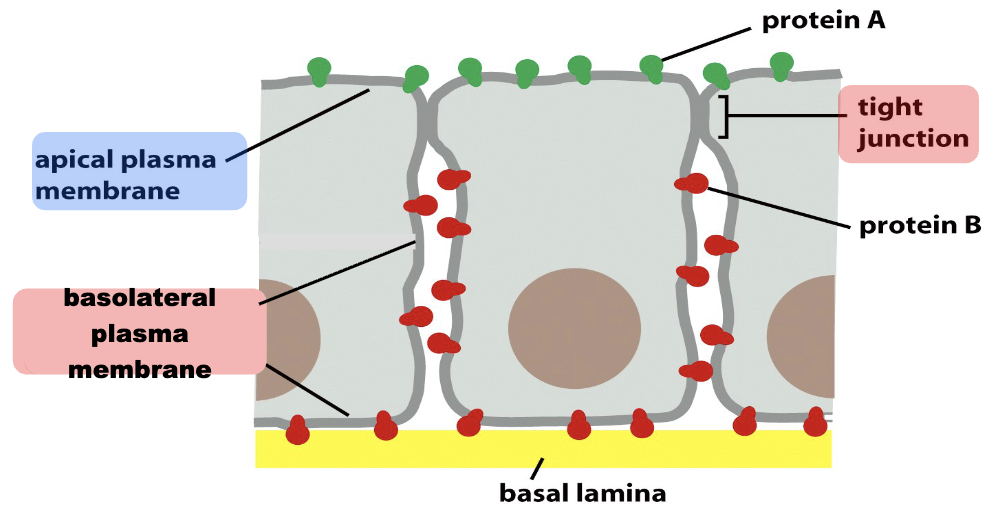
The lateral movement of transmembrane proteins is limited by cellular junctions, especially by the tight junctions.
Such compartmentalization of membrane proteins is at the base of the different functional role of the apical portion of the cell compared to the basolateral one.
Proteins in the Mb
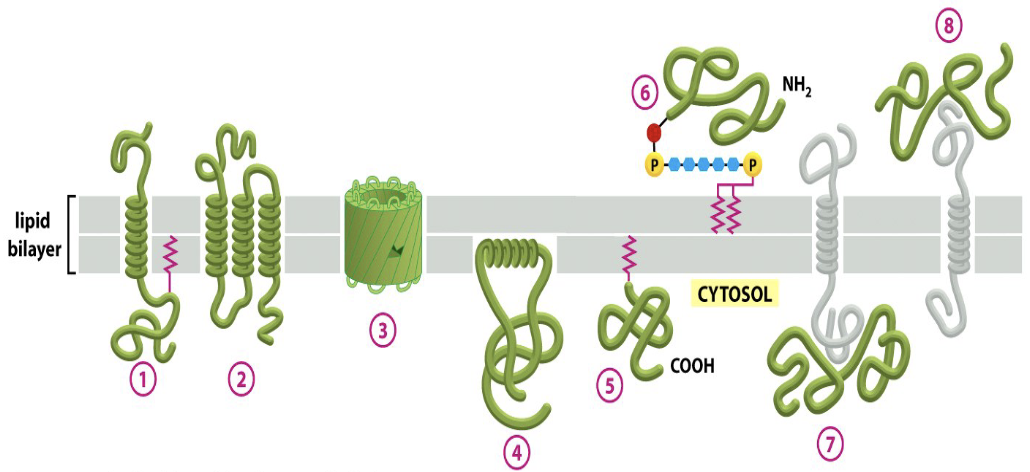
Protein interact with cellular membranes in different ways.
- Transmembrane protein
- =
- =
- Hydrophobic interaction
- Covalent binding with the lipid chain, docking the protein to the membrane or by binding to phosphatidylinositol bonded oligosaccharides
- =
- Non covalent binding with other proteins
- =
Integral mb proteins are embedded in the lipid bilayer, some of them span the bilayer and are termed transmembrane proteins.
Example
Transmembrane proteins → G protein coupled receptors (7x helix domains)
Proteins can also be covalently attached to a lipid moelcule (lipid anchors) but also to GPI-
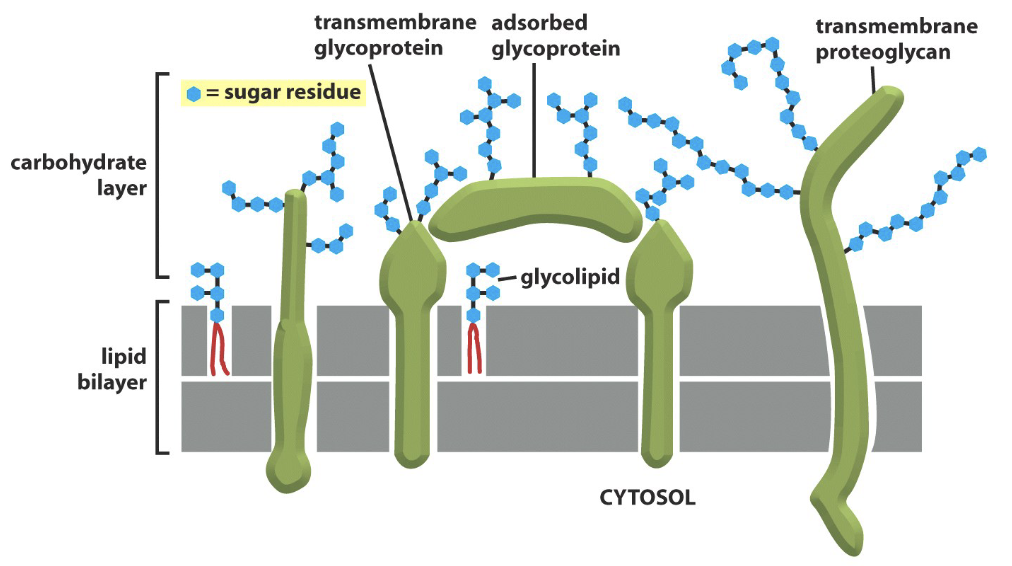
In many cells the outer leaflet lipids, as well as many proteins exposed to the outer surface of the mb, are glycosilated (oligosaccharides attached) that together form the glycocalyx.
The glycocalyx is involved in cell recognition and formation of cell-cell interactions.
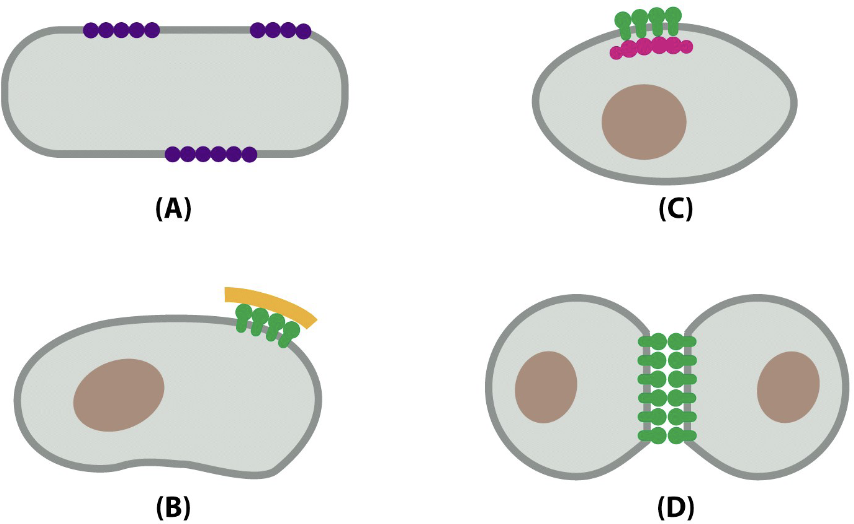
Transmembrane proteins can freely move along the lipid layers (lateral movement) or can regroup in clusters, less mobile because bound with:
- Other transmembrane proteins
- Proteins of the extracellular matrix
- Proteins of the cytoskeleton
- Other cell’s proteins
Many proteins are bound to glycosidic residue.
Moving Across the Membrane
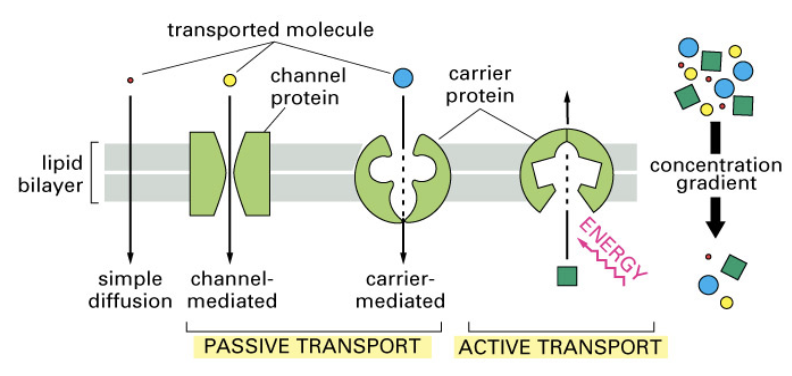
Transcellular passive transports move substances along a gradient by using a transmembrane protein (facilitated diffusion).
Active transports (primary and secondary) move substances against a gradient.
Energy is needed.
inizio buco
3. Transport Across Cell Membranes
Overview
- Passive transport: movement along the electrochemical gradient (no energy required)
- Active transport: movement against the gradient (energy required — ATP or coupled ion gradient)
Fluids and substances move by convection.
- Natural convection → Diffusion
- Forced convection → Advection
3.1 Diffusion
Diffusion is the spontaneous movement of molecules from high to low concentration until the gradient is dissipated.
Energy is needed to maintain the gradient.
Fick’s First Law:
Where:
- = flux density (mol s⁻¹ cm⁻²)
- = diffusion coefficient (cm² s⁻¹)
- = concentration gradient across membrane thickness
Einstein–Stokes equation (for spherical solutes much larger than solvent molecules):
Where:
- J K⁻¹ (Boltzmann’s constant)
- = absolute temperature (K)
- → D depends on the temperature! Remember.
- = molecular radius
- = viscosity
For Lipid Bilayer Diffusion
Introduce the partition coefficient :
- → apolar molecule (crosses easily)
- → hydrophilic molecule (crosses poorly)
Permeability coefficient:
Modified Fick for membrane:
3.2 Electrochemical Gradient
For charged molecules (ions), the driving force is the electrochemical gradient:
- J mol⁻¹ K⁻¹
- = valence (Na⁺: 1; Ca²⁺: 2)
- C mol⁻¹
Glucose only uses the concentration term (uncharged).
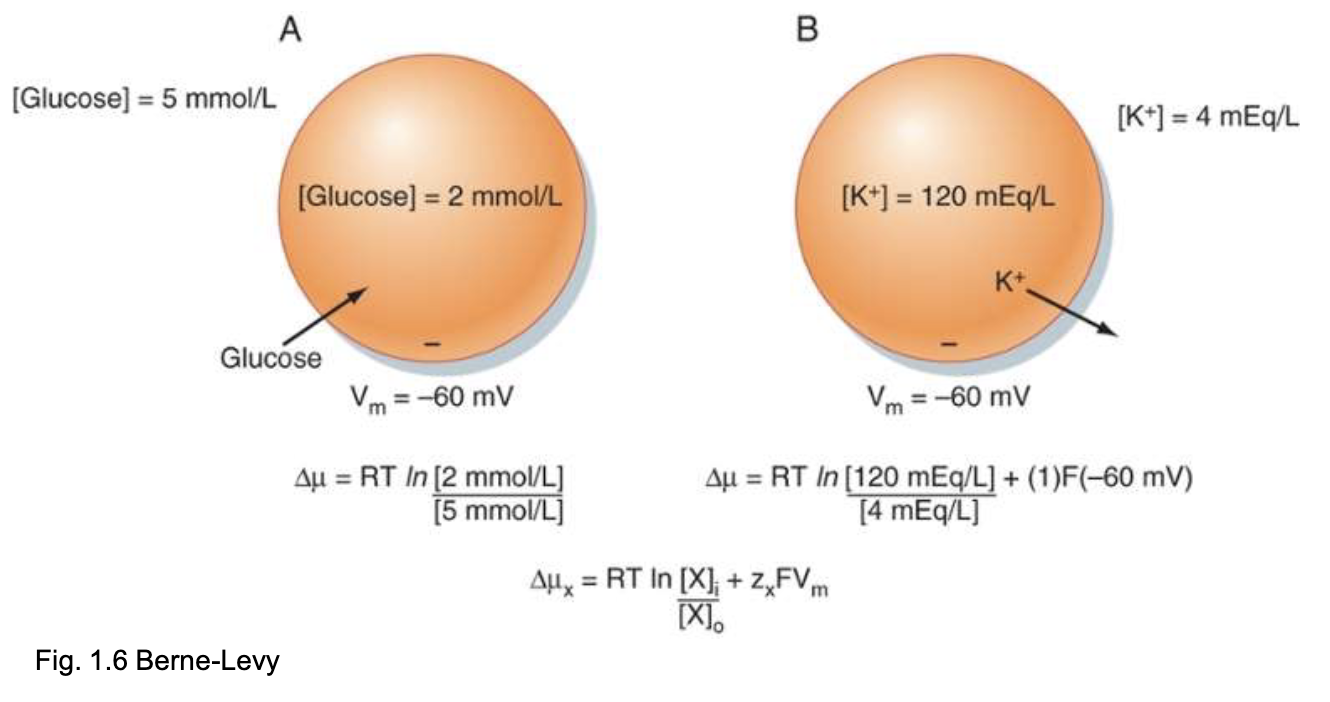
3.3 Nernst Equation
At equilibrium ():
At 37°C:
Example — K⁺:
With mV, net driving force = mV → drives K⁺ out of the cell.
4. Membrane Transport Proteins
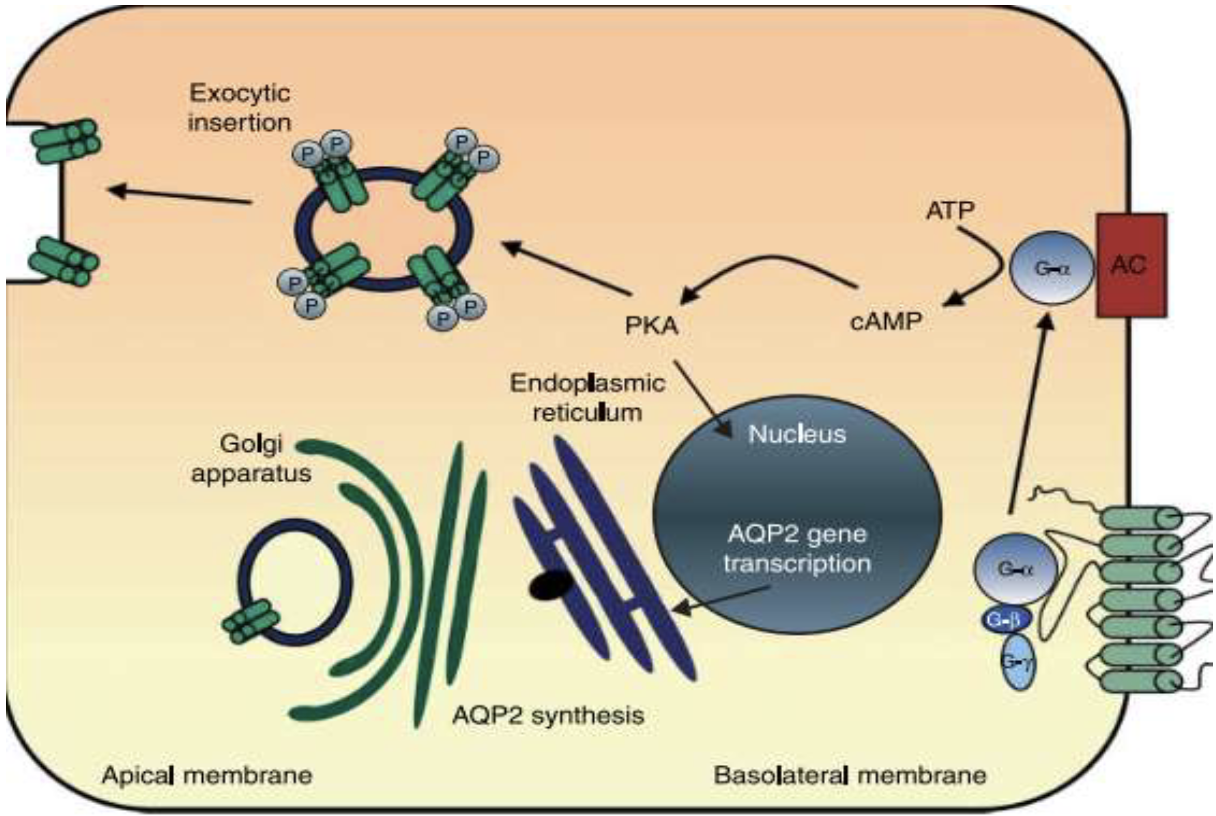
4.1 Water Channels — Aquaporins (AQPs)
- Allow passive water movement
- Widely distributed; different isoforms (e.g. AQP2 apical/AQP3–4 basolateral in kidney collecting duct)
- AQP2 regulated by ADH
- Aquaglyceroporins: also transport glycerol, urea, mannitol, purines, Ca²⁺, NH₃
- Structure: 6 transmembrane domains + central pore; form tetramers
4.2 Ion Channels
Important in excitable cells (neurons, muscle, cardiac). Classified by:
- Selectivity — which ions pass
- Conductance — ions per unit time (pS); inward/outward rectifiers
- Gating — voltage, extracellular ligands, intracellular messengers, mechanical stretch
4.3 Solute Carriers (SLCs)
Integral membrane proteins with hydrophobic TM α-helices. No ATP required.
| Type | Direction | Example |
|---|---|---|
| Uniporter | Downhill, one solute | GLUT1 (glucose) |
| Symporter | Downhill + coupled uphill | NKCC2 (kidney) |
| Antiporter | Opposite directions | NHE-1 (Na⁺/H⁺) |
Active vs Passive
- Passive: along — water channels, ion channels, uniporters (e.g. GLUT1)
- Primary active: ATP-dependent pumps (e.g. Na⁺/K⁺-ATPase)
- Secondary active: movement against coupled to downhill movement of another ion (e.g. SGLT-1, 1Ca²⁺/3Na⁺ antiporter)
4.4 Primary Active Transport — ATPases
Three ATPase families:
| Family | Feature | Example |
|---|---|---|
| P-type | Phosphorylated during transport | Na⁺/K⁺-ATPase, Ca²⁺ pumps, H⁺/K⁺ pump |
| V-type | Vacuolar; intracellular | H⁺-ATPase (lysosome acidification) |
| F-type | Proton-driven ATP synthesis | ATP synthase (mitochondria) |
Na⁺/K⁺-ATPase
Pumps 3 Na⁺ out and 2 K⁺ in per ATP hydrolysed. Blocked by ouabain (used therapeutically at very low concentrations for hypotension and arrhythmias).
Ca²⁺ pump families:
- SERCA — sarco/endoplasmic reticulum Ca²⁺-ATPase
- PMCA — plasma membrane Ca²⁺-ATPase
- SPCA — secretory pathway Ca²⁺-ATPase (Golgi)
H⁺/K⁺ ATPase: kidney, stomach, intestine — 2H⁺/2K⁺
4.5 ABC Transporters
- Present in prokaryotes and eukaryotes
- Use ATP binding/hydrolysis to transport ions or molecules
- MDR (multidrug resistance protein) — confers chemotherapy resistance
- CFTR (cystic fibrosis transmembrane conductance regulator):
- Low-conductance Cl⁻ channel on apical membrane of epithelial cells
- ATP binding to 2 NBDs → channel opening; ATP hydrolysis + ADP release → closure
- Mutation → defective Cl⁻ transport → reduced anion/water flow → thickened mucus → impaired respiration and digestion (Cystic Fibrosis)
Cystic Fibrosis
CFTR mutation impairs Cl⁻ secretion → decreased water secretion → viscous mucus in airways and pancreas.
Regulation of transport: controlled by:
- Retrieval from / insertion into membrane (endocytosis/exocytosis) — fast
- Altered transporter kinetics (gating) — fast
- Synthesis of new transporters — slow (minutes–days)
5. Vesicular Transport (Bulk Transport)
| Process | Direction | Description |
|---|---|---|
| Endocytosis | Inward | Pinocytosis, phagocytosis, receptor-mediated |
| Exocytosis | Outward | Constitutive or regulated |
| Transcytosis | Across cell | Endocytosis + exocytosis in series |
Endocytosis Mechanisms
- Pinocytosis: small molecules + water; typical of endothelial cells
- Phagocytosis: large particles (bacteria, debris); typical of immune cells; often receptor-mediated
- Receptor-mediated endocytosis: specific uptake via surface receptors; requires adaptin, clathrin, dynamin (GTPase)
Endocytic Pathways
- Clathrin-mediated: coated pits → coated vesicles → endosomes → recycling or lysosomal degradation
- Caveolae-mediated: flask-shaped pits; cholesterol-binding caveolin; enriched in cholesterol and glycolipids
- Cargo is sorted in endosomes → recycled to surface or directed to MVBs/lysosomes
Exocytosis
- Constitutive: plasma cells (Ig), fibroblasts (collagen)
- Regulated: endocrine cells, neurons, exocrine glands
- Secretory product: synthesised in RER → Golgi → stored in granules → signal → membrane fusion → release
- Usually triggered by ↑ intracellular Ca²⁺
- Exceptions: renin (JG cells) and PTH (parathyroid) — triggered by ↓ Ca²⁺
fine buco
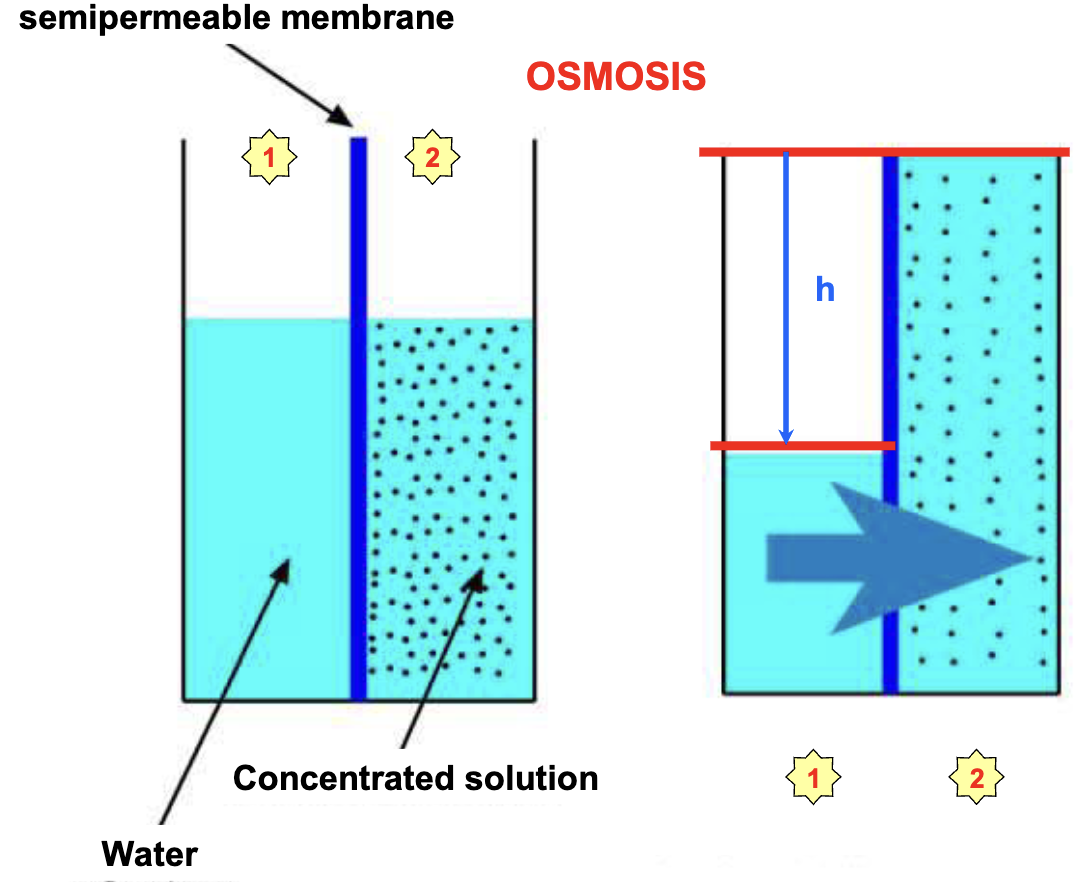
6. Osmosis & Osmotic Pressure
Osmosis: movement of water from low solute concentration to high solute concentration across a semipermeable membrane.
→ the membrane is permeable by the solvent but not by the solutes.
→ specific channels, aquaporins, allow transcellular passage of water. This is how the bulk of water moves through.
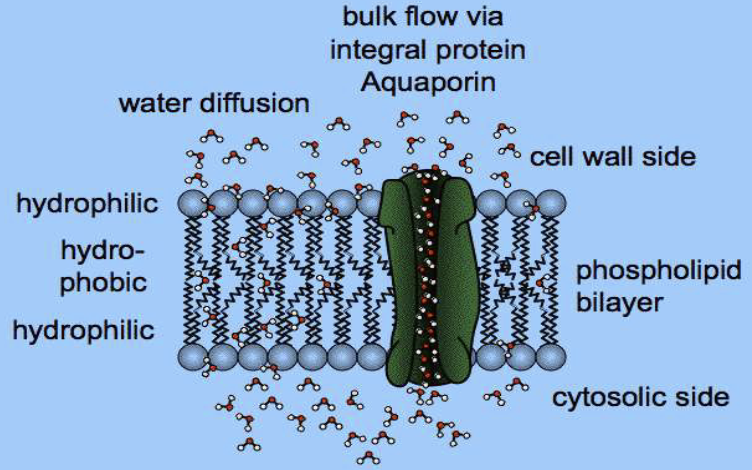
Aquaporins are regulated:
- on a short time scale, by kinases phosphorylating cytoplasmic aquaporins.
- on a longer time scale by activating the transcription and expression of the aquaporin genes.
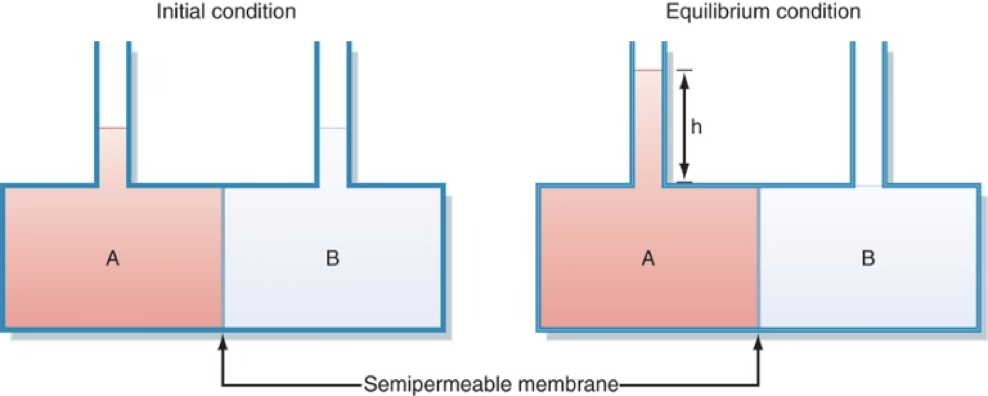
At equilibrium, hydrostatic pressure exerted by the column of water stops the net movement of water from to and is equal to the osmotic pressure exerted by the solute in .
Van’t Hoff’s Law:
- = number of dissociable particles per molecule
- = total solute concentration
- atm L K⁻¹ mol⁻¹
- = temperature (K)
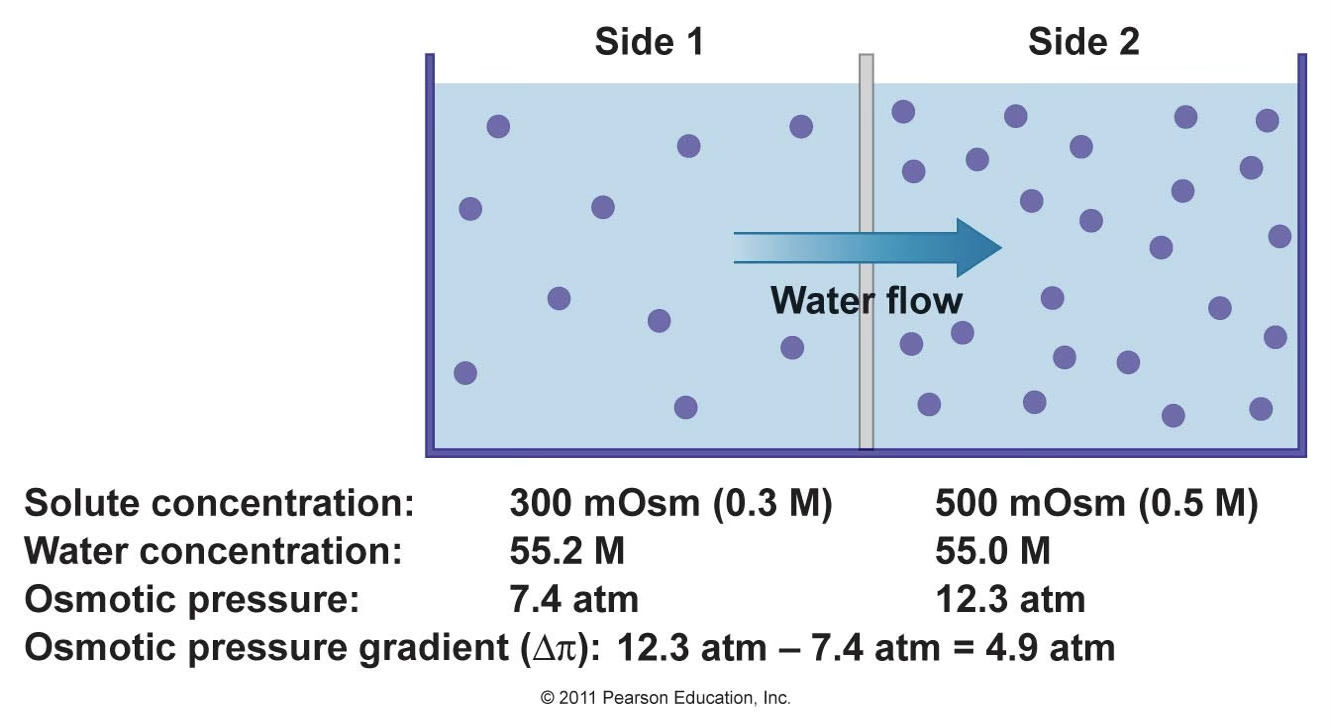
Osmolarity = (mOsm/L)
NaCl vs Glucose
- Glucose (1 mmol/L): , mmHg = 1 mOsm/L
- NaCl (1 mmol/L): , mOsm/L
Osmolarity vs Osmolality
- Osmolarity: osmoles per litre of solution (temperature-dependent)
- Osmolality: osmoles per kg of solvent (temperature-independent; preferred in physiology)
- At 37°C in dilute solutions they are approximately equivalent.
Example
Using vant’Hoff…
At , (considering ):Side 1 →
Side 2 →
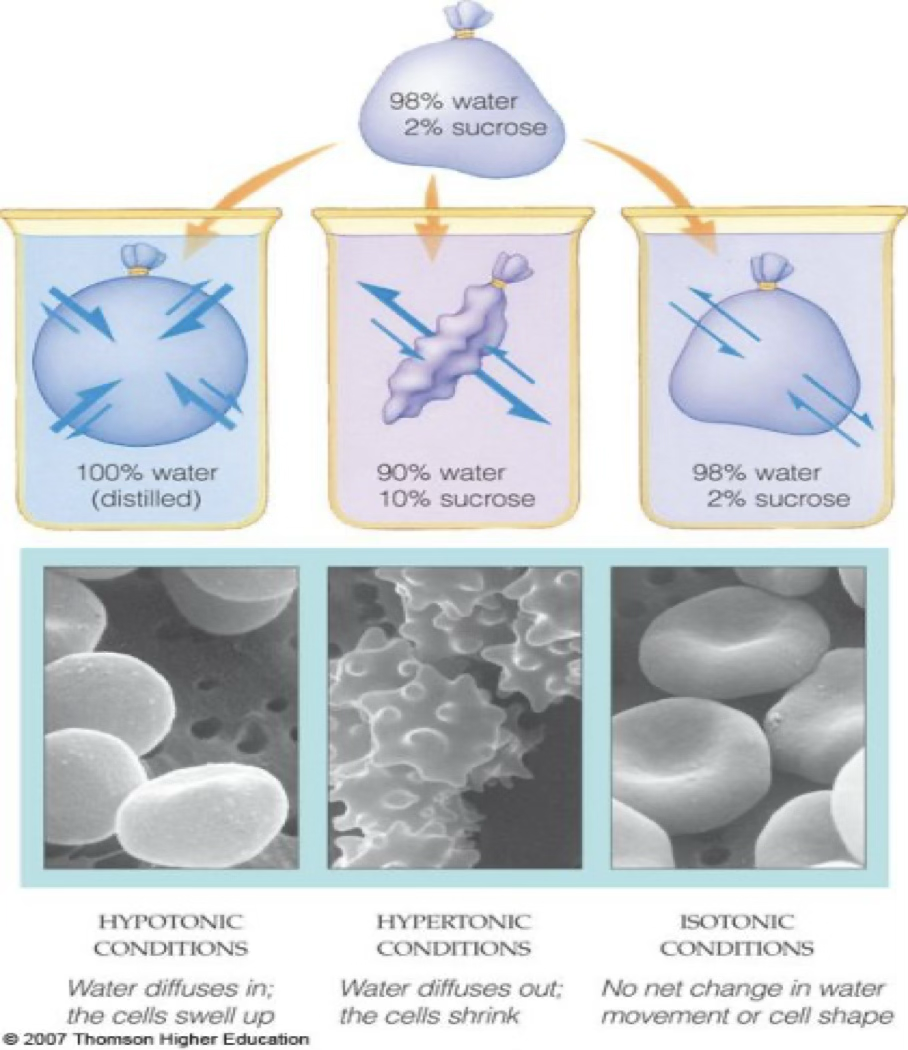
Tonicity
Tonicity is a qualitative measure.
Bodily fluid osmolality is about .
Tonicity relates to the effect of a solution on the volume of a cell.
| Solution | Effect on Cell Volume |
|---|---|
| Isotonic | No change |
| Hypotonic | Swelling |
| Hypertonic | Shrinking |
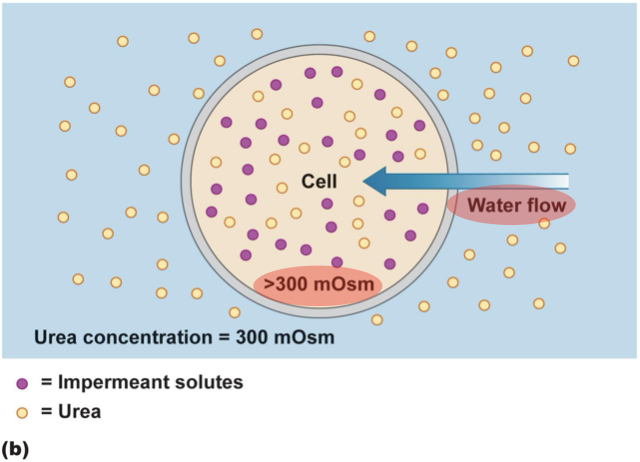
Effective vs Ineffective Osmoles
Tonicity accounts for membrane permeability.
) and causes cell swelling/lysis.
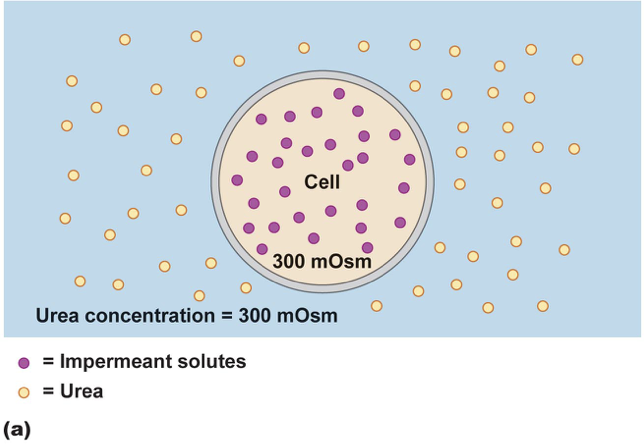
E.g. 300 mOsm/kg urea and 300 mOsm/kg sucrose are isosmotic, but only sucrose is isotonic for RBCs — urea crosses freely (ineffective osmole;
Remember, not exert osmotic pressure.
A solute that can cross the membrane easily does
Corrected Van’t Hoff:
where reflection coefficient (0 = freely permeable; 1 = totally impermeable).
Normal saline (0.9% NaCl):
154 mmol/L NaCl → theoretical 308 mOsm/L → actual ~290 mOsm/L (93% dissociation) → isotonic solution for medical rehydration.
7. Regulation of Cell Volume
Changes in volume can lead to cell death, so they have developed mechanisms to counteract these changes.
Isotonic Volume Regulation
Most cells are highly permeable to water because of aquaporins.
Even in isotonic ECF1, volume maintenance is active and requires Na⁺/K⁺-ATPase. Intracellular proteins cause ion redistribution (Gibbs-Donnan effect) that would swell the cell if unopposed.
Gibbs-Donnan Effect
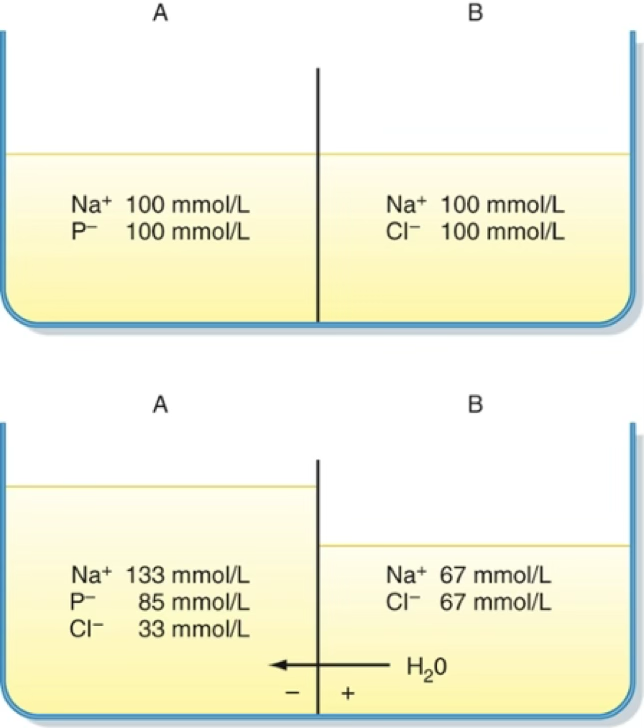
When a membrane is permeable to some but not all solutes (e.g. impermeable to proteins P⁻):
- Cl⁻ diffuses from B → A (down gradient) → A becomes electronegative
- Na⁺ follows into A (electrical gradient)
- ↑ osmolality in A → water flows in → volume increase (or pressure increase in sealed containers) → lower [P⁻]
If the container containing the two solutions were sealed at the top so that water could not move from compartment B to compartment A, the pressure in compartment A would increase as the number of osmotically active particles increases in that compartment.
Note
The same effect operates across capillary walls (plasma vs interstitial fluid). Na⁺/K⁺-ATPase counteracts the Gibbs-Donnan effect.
Non-Isotonic Volume Regulation
Most cells in the body are bathed with isotonic ECF1, but not all of them.
Relevant for cells in kidney medulla, CSF, etc. (with huge concentration gradient (?))
| Situation | Response | Mechanism | |
|---|---|---|---|
| Hypertonic ECF | Water goes out →(then) RVI (Regulatory Volume Increase) | NHE-1, NKCC-1, cation channels → ↑ NaCl in ICF → ↑ Na⁺/K⁺-ATPase → net ↑ KCl + organic osmolytes | 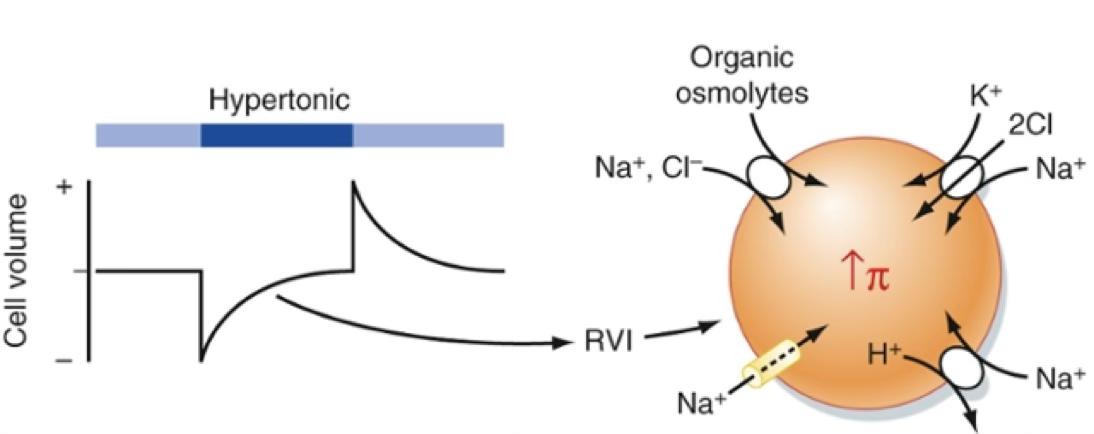 |
| Hypotonic ECF | Water comes in →(then) RVD (Regulatory Volume Decrease) | Loss of KCl from ICF | 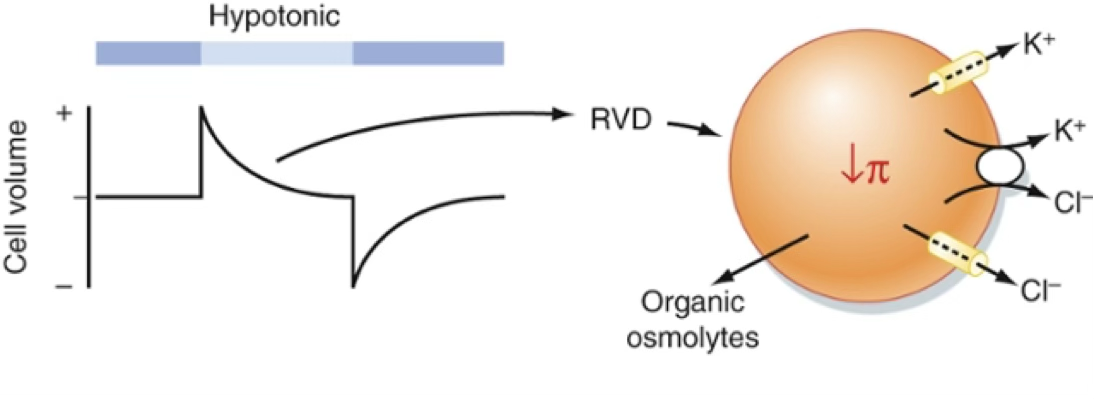 |
Volume changes monitored by: cytoskeleton, macromolecular crowding, ionic strength, stretch-gated channels, second messengers.
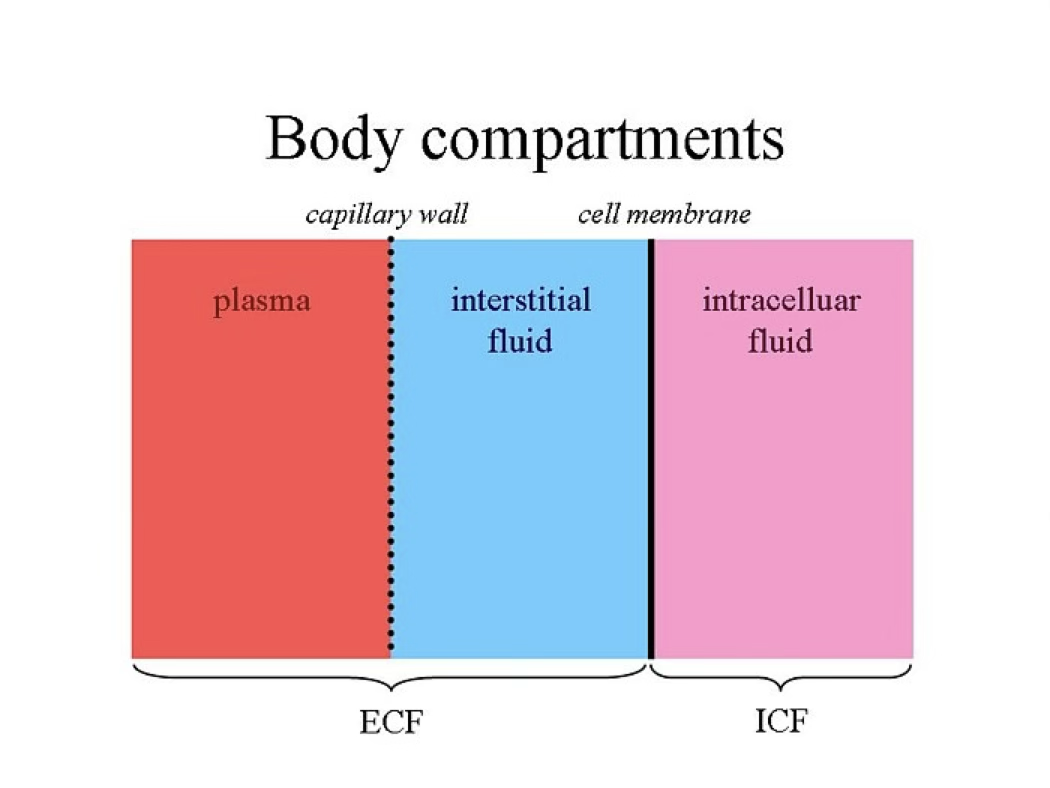
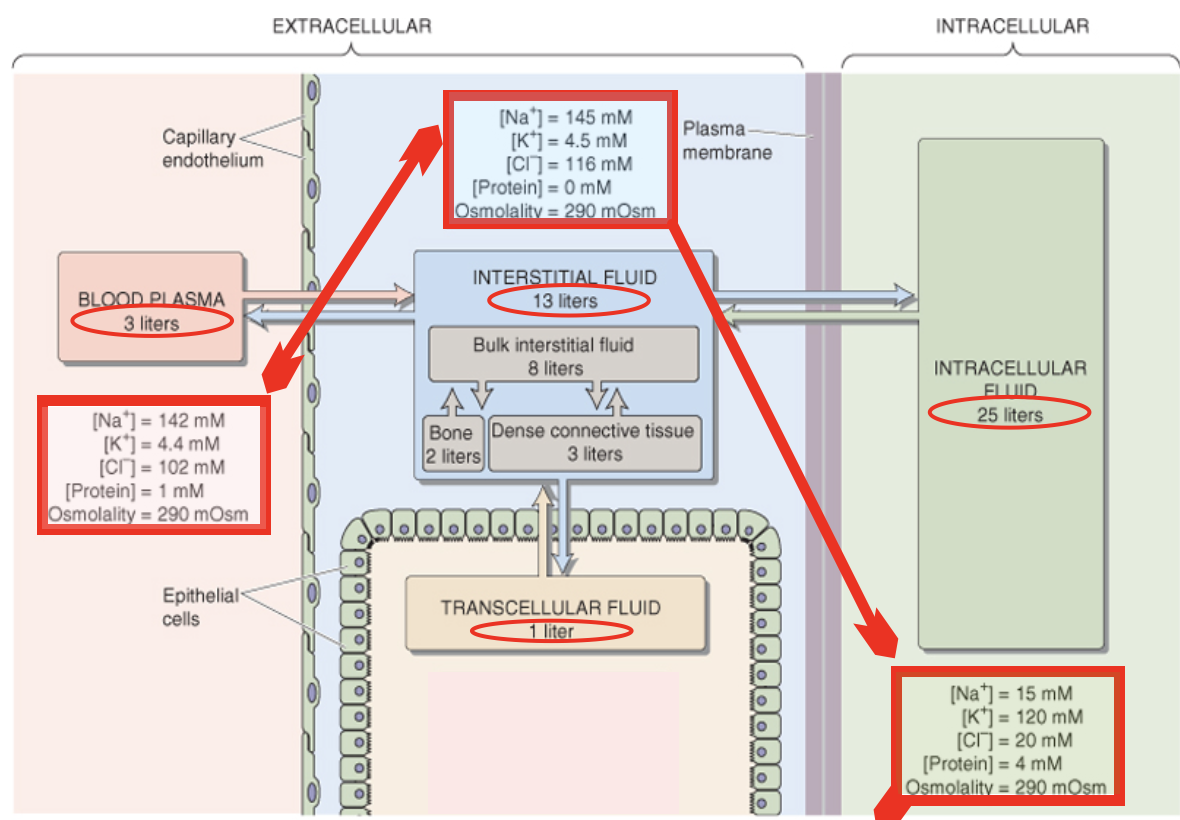
8. Body Fluid Compartments & Homeostasis
Normal cellular function requires that intracellular environment remains constant (in a narrow range).
This is accomplished by transport of many substances / water in&out of the cell to maintain a constant environment → homeostasis.
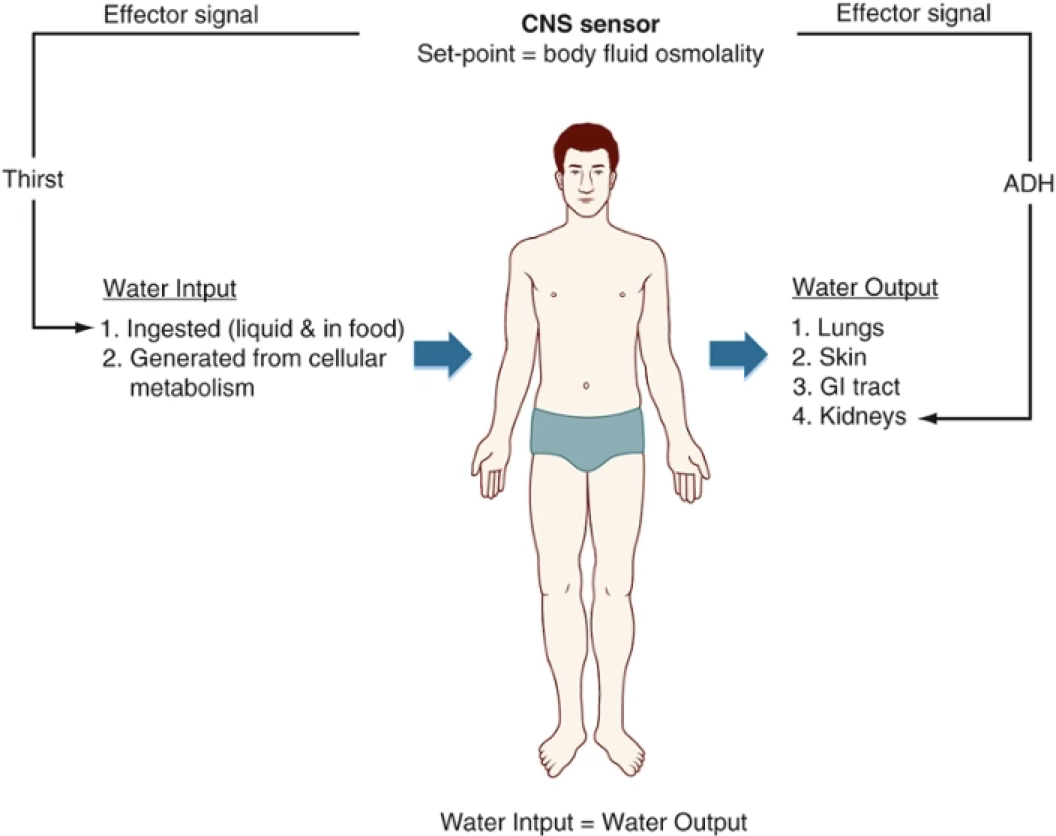
Characteristics of the concept of homeostasis are:
- There must be a ‘set point’ so that deviations from it can be monitored.
- Sensors must generate an ‘effector signal’ when the variable is different from the set point
- Effector organ must respond in the appropriate way to the effector signal
The sensitivity of the system depends on several factors:
- the nature of the sensor,
- time necessary to generate effector signal
- and time necessary for effector organ to respond.
Water is the key element in every bodily fluid:
- High specific heat (): The energy required to raise the temperature of water. This helps the body resist sudden temperature changes, maintaining thermal stability.
- High latent heat of evaporization (): The energy needed to turn liquid water into vapor. This allows for efficient cooling via sweat (evaporative cooling).
- High latent heat of fusion (): The energy required to change ice to liquid water. In physiology, this helps protect cells from freezing by requiring significant energy loss before ice crystals form.
- High surface tension
- High solubilizing capability
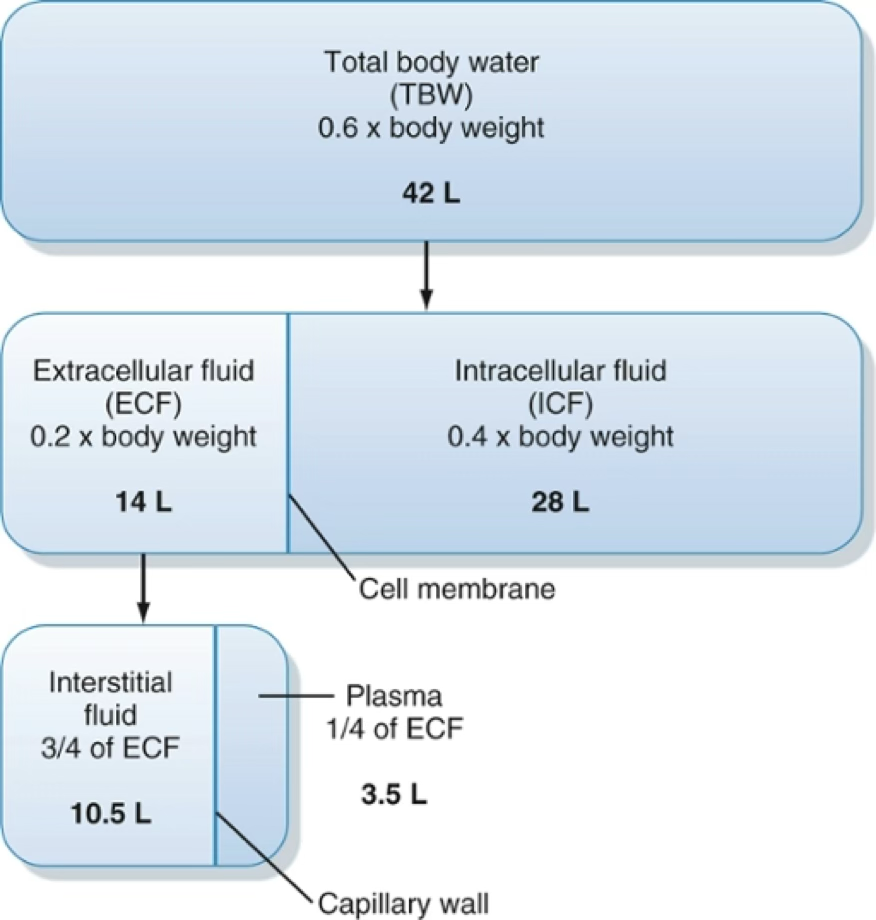
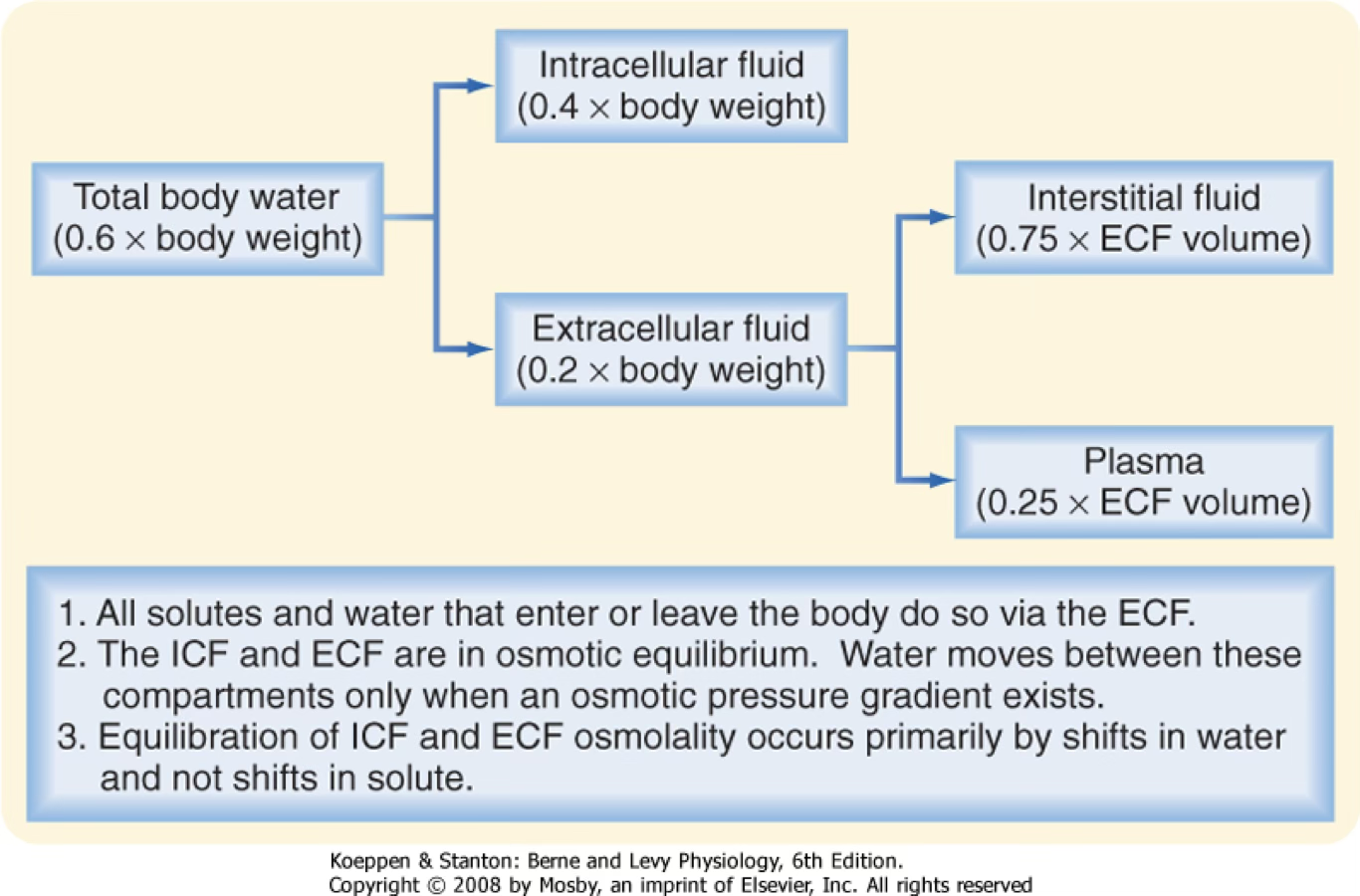
Total body water ≈ 60% of body weight (varies with adipose tissue and age):
Fetus 90% → Newborn 80% → Child 70% → Adult 60% → Elderly 55%
| Compartment | Subdivision |
|---|---|
| ICF | ~2/3 of TBW2 |
| ECF | Plasma + Interstitial fluid (~1/3 of TBW) |
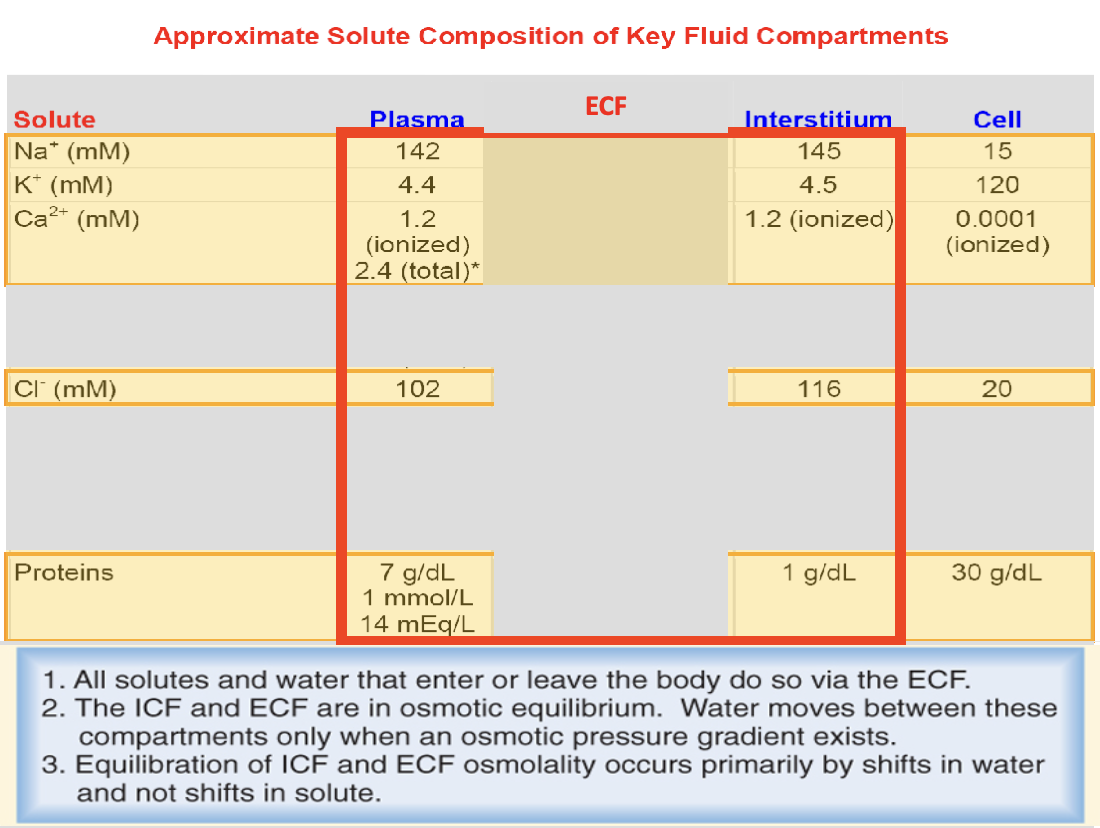
Plasma Osmolality Estimate
Since Na⁺ (with Cl⁻ and HCO₃⁻) dominates ECF osmolality:
Exceptions to uniform ECF osmolality: CSF (higher) and kidney ECF (variable).
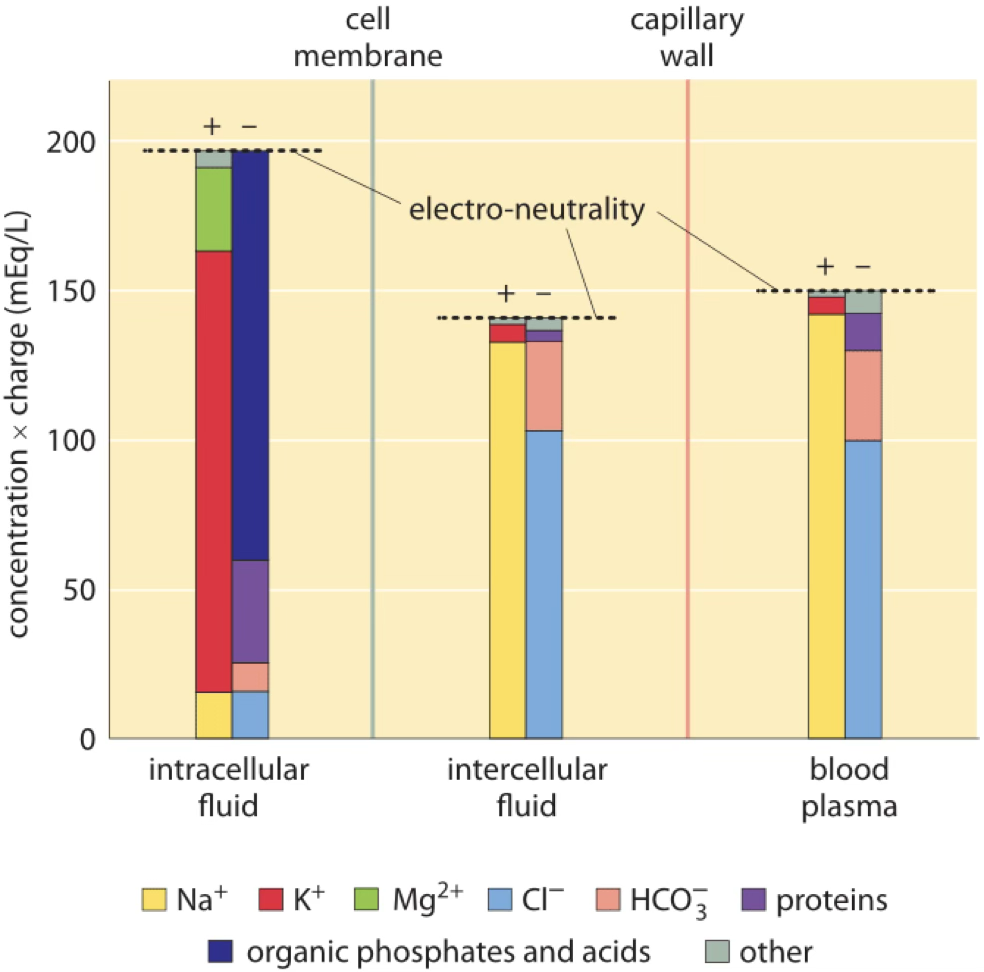
ECF and ICF are different solutions:
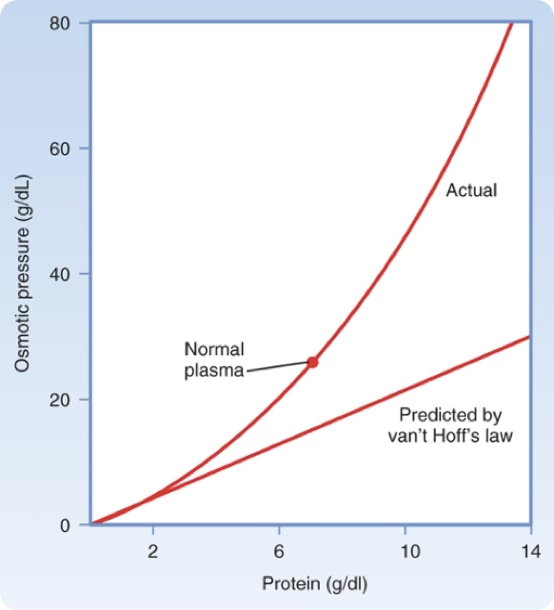
Oncotic Pressure
Osmotic pressure generated by large molecules (mainly proteins).
Does not follow Van’t Hoff’s law precisely.
Plasma oncotic pressure
Human plasma oncotic pressure ≈ 26–28 mmHg (≅ 1.4 mOsm/kg H₂O). Key force for fluid movement across capillaries. Albumin (69 kDa) exerts greater osmotic force than predicted due to its negative charge at physiological pH (Gibbs-Donnan effect).
Specific gravity of normal human plasma: 1.008–1.010
Specific gravity
is defined as the weight of a volume of solution divided by the weight of an equal volume of distilled water.
9. Epithelial Transport
Epithelial cells form sheets at the interface between external world and ECF.
Functions:
- Barrier against microorganisms (lungs, GI, skin)
- Prevention of water loss (skin)
- Maintenance of constant internal environment (lungs, GI, kidneys)
Epithelial cells
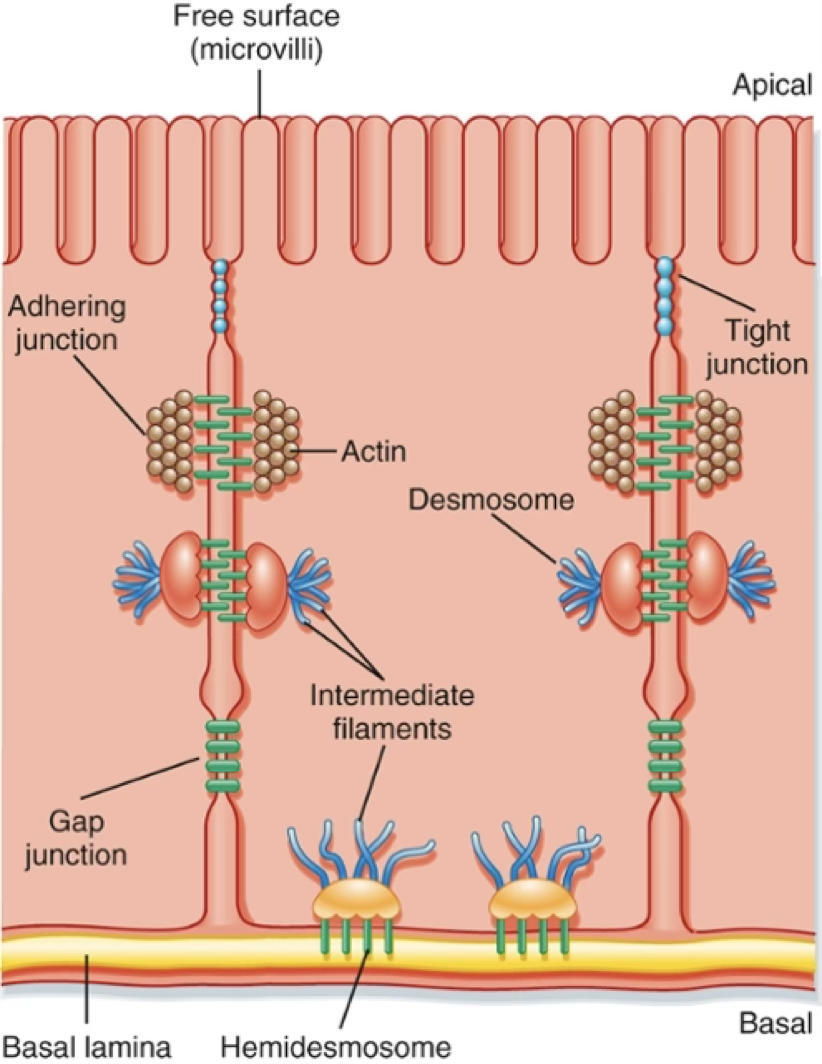
The free surface is the apical mb that is in contact with the external environment or the ECF.
The basal side rests on the basal lamina, secreted by epithelial cells and that is attached to underlying connective tissue.
Junctions
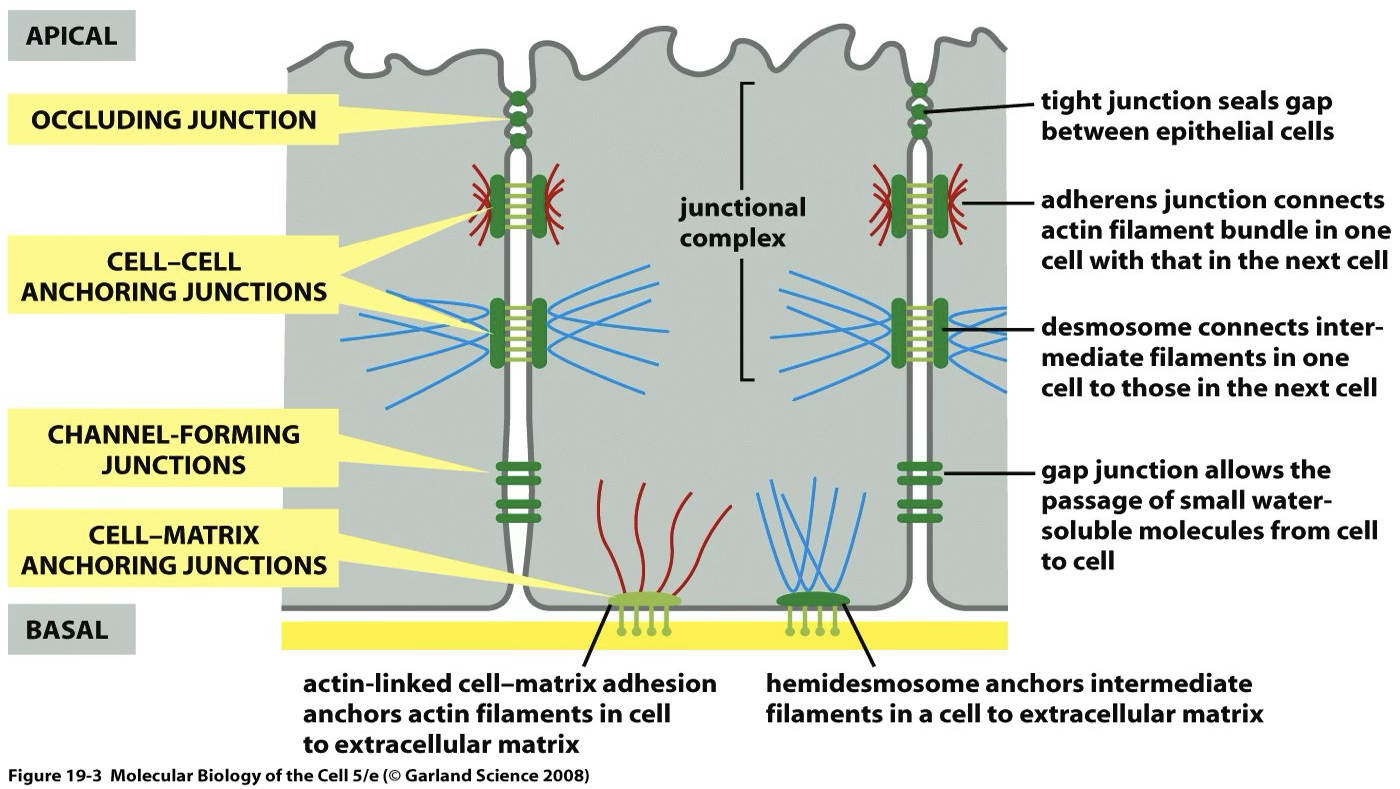
| Junction | Function |
|---|---|
| Tight junctions | Separate apical from basolateral domains; restrict paracellular movement |
| Adherens junctions | Mechanical adhesion |
| Desmosomes | Mechanical resistance |
| Gap junctions | Electrical/metabolic coupling (connexons of 6 connexins) |
| Hemidesmosomes | Anchoring to basal lamina |
Vectorial Transport
Important
Tight junctions enable vectorial transport by segregating different transporters into apical vs basolateral domains.
- Absorption/reabsorption: apical → basolateral (e.g. nutrients from GI, water from nephron)
- Secretion: basolateral → apical
- Na⁺/K⁺-ATPase is segregated to the basolateral membrane in all epithelia except choroid plexus and retinal pigment epithelium.
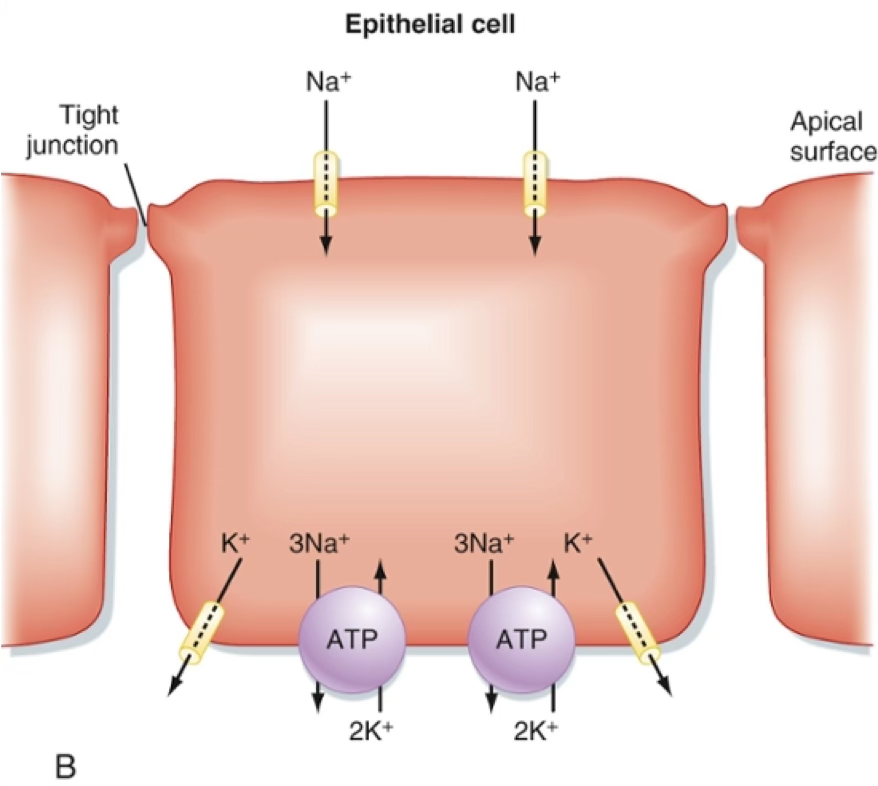 | 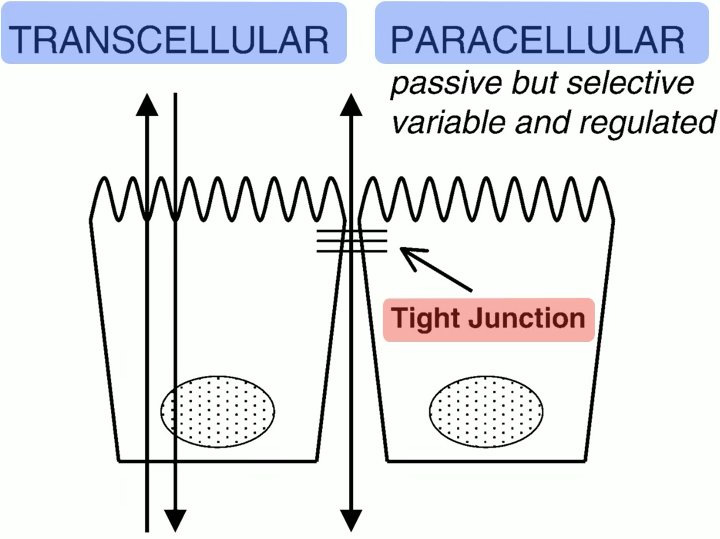 |
|---|---|
| Transcellular transport is a two-step process. | |
| Generally, one of the step is passive and the other active. |
Paracellular transport — passive; driven by transepithelial concentration gradient and transepithelial voltage.
Tight junction permeability varies:
- Leaky: proximal tubule, early intestine
- Tight: collecting duct, urinary bladder, terminal colon
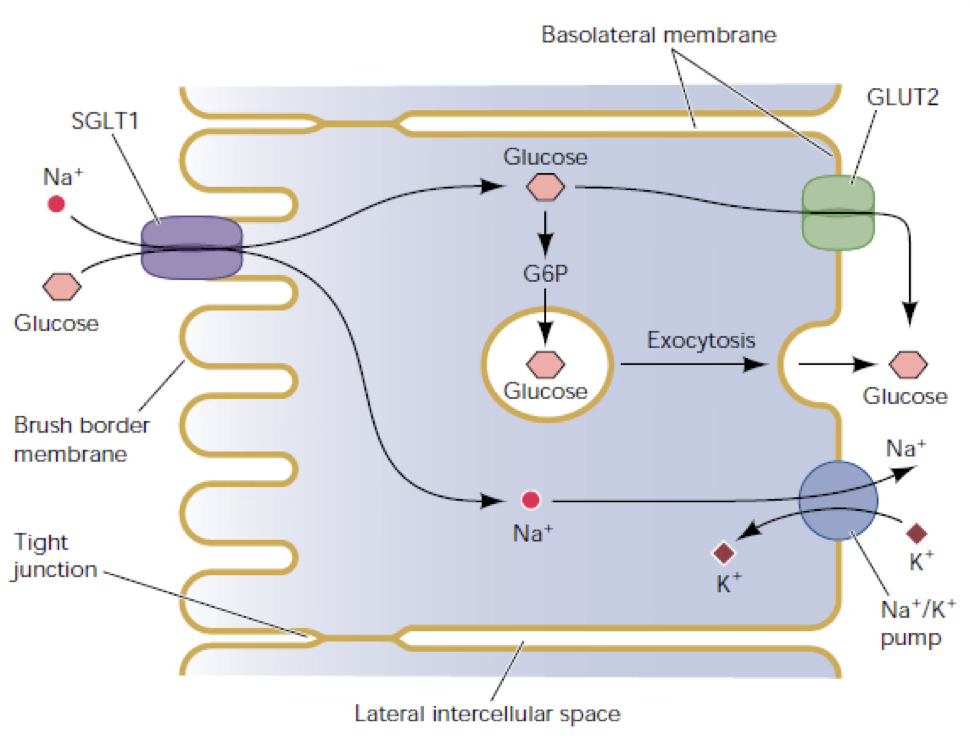
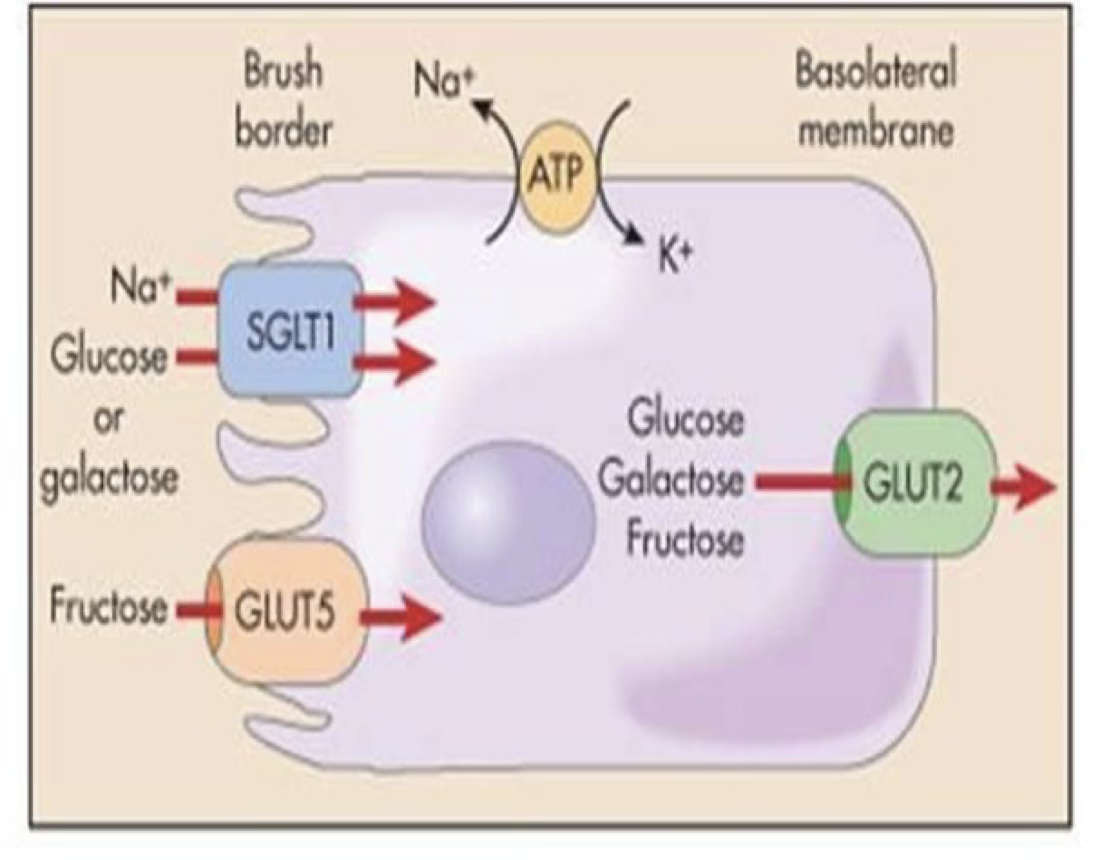
Example — intestinal glucose absorption:
- Apical entry: SGLT1 (Na⁺-glucose co-transport, secondary active; 6 isoforms SGLT1–6)
- Basolateral exit: GLUT2 (facilitative uniporter) or exocytosis
- Na⁺ gradient maintained by basolateral Na⁺/K⁺-ATPase
Galactose uses same mechanism; fructose uses GLUT5 (apical) + GLUT2 (basolateral).
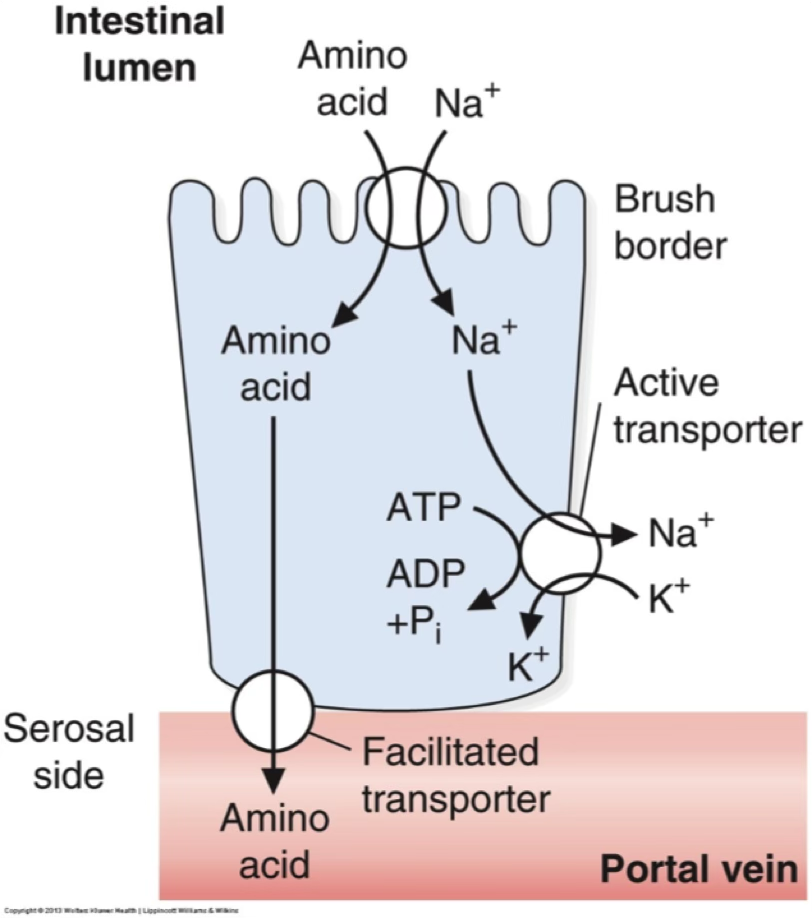
pump creates a gradient that can also be used to transport amino acids:
Transepithelial water movement — passive, driven by osmotic gradients created by solute transport (transcellular + paracellular). Solvent drag: water movement can carry additional solute.
Regulation of epithelial transport:
- hormonal (e.g. aldosterone → NaCl reabsorption in colon, nephron, sweat ducts),
- neural (enteric NS, sympathetic NS),
- paracrine (e.g. histamine → HCl secretion by parietal cells).
Regulation of transport depends on:
- Retrieval of transporters from mb (endocytosis) or insertion in the mb (exocytosis)
- Change in activity of the transporter (gating)
- Sinthesis of specific transporters and insertion into the mb.
First two are rapid, last one is a slow process (minutes/hours/days)
10. Fluid Exchange in Capillaries
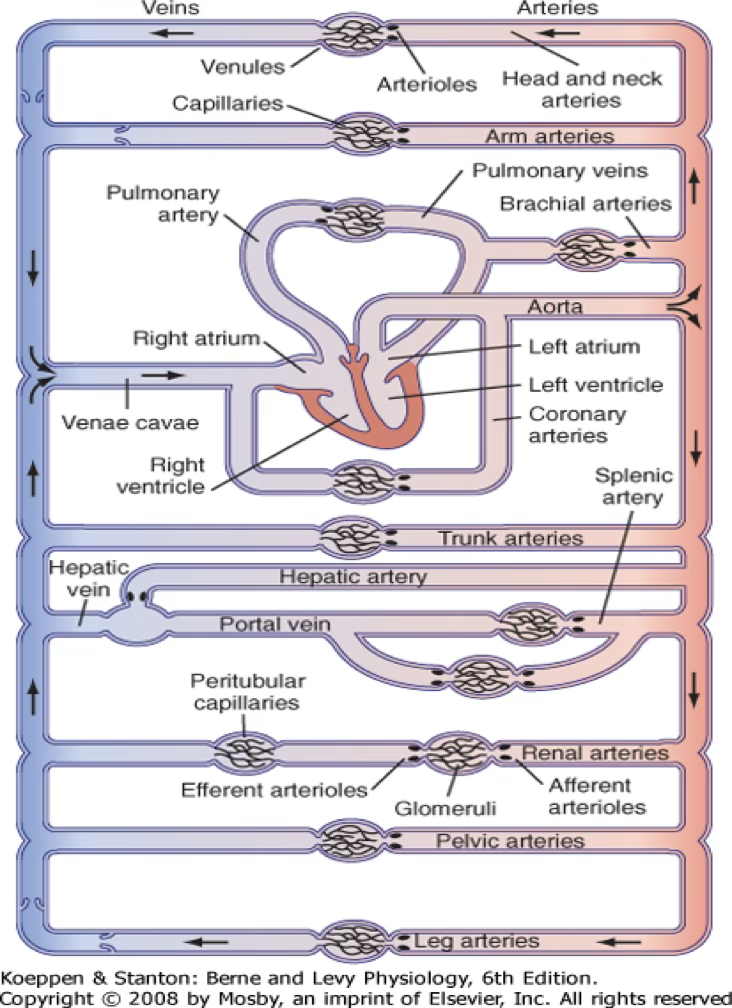
Systemic capillaries:
![]()
Movement of water from interstitial fluid to intravascular environment and viceversa occurs across the capillary wall.
The amount of water that moves depends on the type of capillary:
Capillary Permeability
| Capillary Type | Structure | Examples |
|---|---|---|
| Continuous | Tight barrier | Brain (BBB), muscle |
| Fenestrated | Pores | Kidney, intestine |
| Sinusoidal | Large gaps | Liver |
Transport mechanisms: diffusion, bulk flow (filtration), vesicular transport, active transport.
Fick’s law for capillary diffusion:
- = capillary permeability
- = surface area
- product = index of available capillary surface area
Flow-Limited vs Diffusion-Limited Transport
- Small molecules (H₂O, urea, NaCl, glucose, O₂, CO₂): capillary wall offers low resistance → transport limited by blood flow
- Large molecules (>60 kDa): diffusion across the wall becomes the limiting factor → diffusion-limited
- Lipid-soluble molecules cross directly through the membrane.
Counter-current Exchange
O₂ saturation can decrease to ~80% at capillary entrance because O₂ diffuses from arterioles and arteries into adjacent venules (counter-current exchange). At low blood flow, O₂ delivery to tissue decreases.
Starling Forces
Filtration is driven by hydrostatic pressure () and oncotic pressure () across the capillary wall.
Water permeability / filtration:
Starling equation:
| Parameter | Arteriolar end (mmHg) | Venular end (mmHg) |
|---|---|---|
| (capillary hydrostatic) | +35 | +16 |
| (interstitial hydrostatic) | 0 | 0 |
| (capillary oncotic) | +26 | +26 |
| (interstitial oncotic) | +1 | +1 |
| Net force | +10 (filtration) | −9 (reabsorption) |
Hydrostatic Pressure Determinants
- ↑ arterial/venous pressure → ↑
- ↑ arteriolar resistance → ↓
- ↑ venular resistance → ↑
- Venous pressure changes affect more (~80% transmitted vs ~20% for arterial changes)
Lymphatic System
- Closed-end network without tight junctions between endothelial cells
- Filaments anchor lymphatics to connective tissue → muscle contraction opens large spaces → proteins and water enter
- One-way valve system drives lymph flow
- Present everywhere except brain, bones, cartilage, epithelia
- Returns all filtered proteins to blood (failure → oedema/ascites)
- Transports fat (as chylomicrons) from GI tract
- Volume transported in 24h ≅ total plasma volume
11. Oxygen and Carbon Dioxide Transport
O₂ Transport
O₂ is carried in two forms:
- Dissolved in plasma (measured as ) — minor fraction
- Bound to haemoglobin (Hgb) — major fraction (OxyHb)
Haemoglobin structure: 4 heme groups (Fe²⁺ — must be ferrous, not ferric/Fe³⁺) + 4 globin polypeptide chains.
- Each Hgb binds up to 4 O₂
- Each gram Hgb binds up to 1.34 mL O₂
- ~280 million Hgb molecules per RBC
- Binding/dissociation: milliseconds (RBC spends only 0.75 s in capillary; equilibration in ~0.25 s)
- O₂ transfer is normally perfusion-limited
Oxyhemoglobin Dissociation Curve (S-shaped)
The sigmoid shape reflects cooperative binding: O₂ binding to one heme ↑ affinity of remaining hemes.
Rightward shift (↓ O₂ affinity → ↑ O₂ delivery to tissues):
- ↑ Temperature
- ↑ PCO₂ (Bohr effect)
- ↑ 2,3-DPG
- ↓ pH
Leftward shift (↑ O₂ affinity → ↓ O₂ delivery):
- ↓ Temperature, ↓ PCO₂, ↓ 2,3-DPG, ↑ pH
Bohr Effect
Bohr Effect — O₂ Delivery to Tissues
In metabolically active tissues, CO₂ accumulates → ↓ pH → Hgb releases O₂ more easily.
- CO₂ and H⁺ compete with O₂ for Hgb binding
- Ensures more O₂ reaches cells working harder (e.g. exercising muscle)
Key reactions:
O₂ delivery:
Hgb leaves tissues ~75% saturated → only ~25% O₂ is extracted per pass.
Erythropoiesis — RBC production in bone marrow; controlled by erythropoietin (synthesised by cortical interstitial cells of the kidney in response to hypoxia).
CO₂ Transport
CO₂ is transported in three forms:
- Carbamino-Hgb (Hb-COO⁻)
- Bound to H⁺ as H⁺Hb (after carbonic anhydrase reaction)
- Dissolved in plasma
CO₂ production: ~200 mL/min under resting conditions.
Respiratory exchange ratio = expired CO₂ / O₂ uptake = 0.8 (i.e. 80 CO₂ expired per 100 O₂ entering).
depends solely on alveolar ventilation and CO₂ production (inverse relationship with ventilation).
Chloride Shift
HCO₃⁻ produced in RBCs diffuses out in exchange for Cl⁻ → maintains osmotic equilibrium of RBC.
Haldane Effect
Haldane Effect — CO₂ Removal in the Lungs
Deoxygenated Hgb has greater affinity for CO₂ than oxygenated Hgb.
- In tissues: Hgb releases O₂ → becomes deoxygenated → picks up more CO₂
- In lungs: Hgb binds O₂ → releases CO₂ for exhalation
CO₂ dissociation curve is linear (unlike the sigmoid O₂ curve).
Bohr vs Haldane — Summary
Effect Location Mechanism Outcome Bohr Tissues ↑ CO₂/H⁺ → ↓ O₂ affinity More O₂ delivered to tissues Haldane Lungs O₂ binding → ↓ CO₂ affinity More CO₂ released to alveoli
📚 TLDR — Comprehensive Summary
Cell & Membrane Basics
Eukaryotic cells are bounded by a 5 nm phospholipid bilayer with asymmetric leaflet composition (PC and SM on outer; PE, PS, PI on inner). Membrane fluidity is governed by cis double bond content and cholesterol concentration (cholesterol increases viscosity). Proteins integrate via transmembrane domains, lipid anchors, GPI tails, or non-covalent interactions. Tight junctions polarise epithelia into functionally distinct apical and basolateral domains. Lipid rafts (sphingomyelin + cholesterol microdomains) concentrate signalling proteins.
Diffusion & Fick’s Law
Passive diffusion follows . For membranes, permeability incorporates the partition coefficient . Ions are also driven by the membrane potential (electrochemical gradient ). The Nernst equation gives the equilibrium potential for any ion; at 37°C: . For K⁺, mV; because mV, there is a net outward driving force on K⁺.
Transport Proteins
- Aquaporins: passive water channels (tetramers, 6 TM domains); AQP2 regulated by ADH.
- Ion channels: selective, gated (voltage, ligand, stretch), characterised by conductance (pS).
- SLCs: uniporters (GLUT1), symporters (NKCC2, SGLT1), antiporters (NHE-1) — no ATP.
- P-type ATPases: Na⁺/K⁺-ATPase (3Na⁺ out/2K⁺ in per ATP; blocked by ouabain), Ca²⁺ pumps (SERCA/PMCA/SPCA), H⁺/K⁺ pump.
- V-type H⁺-ATPase: lysosome/endosome/Golgi acidification.
- F-type ATP synthase: mitochondrial ATP synthesis using proton gradient.
- ABC transporters: MDR (multidrug resistance); CFTR (Cl⁻ channel; mutation → Cystic Fibrosis).
- Regulation: fast (endocytosis/exocytosis of carriers, gating changes) or slow (new synthesis).
Vesicular Transport
Endocytosis: pinocytosis (fluid-phase), phagocytosis (large particles), receptor-mediated (clathrin-coated pits + dynamin; or caveolae). Exocytosis: constitutive (plasma cells, fibroblasts) or regulated (triggered by ↑ Ca²⁺, except renin and PTH which are triggered by ↓ Ca²⁺).
Osmosis & Cell Volume
; osmolality ≈ 290 mOsm/kg in body fluids. Tonicity depends on the reflection coefficient : only impermeant solutes (, like sucrose) are effective osmoles — urea () crosses freely and does not exert lasting osmotic effect. Cell volume is actively maintained by Na⁺/K⁺-ATPase (opposing Gibbs-Donnan swelling). RVI uses NHE-1 + NKCC-1; RVD uses KCl loss.
Body Fluids
TBW ≈ 60% of body weight (↓ with age and adiposity). ECF dominated by Na⁺ (with Cl⁻/HCO₃⁻); plasma osmolality ≈ 2[Na⁺]. Oncotic pressure of plasma ≈ 26–28 mmHg (mainly albumin, amplified by Gibbs-Donnan effect).
Epithelial Transport
Tight junctions separate apical from basolateral domains, enabling vectorial transport. Na⁺/K⁺-ATPase on the basolateral side creates the driving gradient for most secondary active transport. Intestinal glucose absorption: SGLT1 (apical, Na⁺-driven) → GLUT2 (basolateral, passive). Paracellular transport is passive and depends on tight junction permeability (leaky in proximal tubule; tight in collecting duct). Regulated by aldosterone, sympathetic/enteric nervous system, and paracrine signals.
Capillary Exchange & Starling Forces
At the arteriolar end: net filtration (+10 mmHg); at the venular end: net reabsorption (−9 mmHg). Small molecules are flow-limited; large molecules are diffusion-limited. The lymphatic system returns all filtered protein and the full plasma volume (~24 h cycle) to prevent oedema.
O₂ / CO₂ Transport
O₂ binds cooperatively to Hgb (sigmoid dissociation curve; 1.34 mL O₂/g Hgb; 4 O₂/Hgb molecule). Bohr effect: ↑ CO₂/↓ pH → rightward curve shift → more O₂ unloaded at tissues. Haldane effect: deoxygenated Hgb has higher CO₂ affinity → more CO₂ loaded at tissues and released at lungs. CO₂ travels as HCO₃⁻ (via carbonic anhydrase + chloride shift), carbamino-Hgb, and dissolved CO₂. Respiratory exchange ratio = 0.8; PaCO₂ is inversely proportional to alveolar ventilation.